Mohammad & Mohammad
FULL PAPER
is more polar than the solvent n-heptan or toluene.
Similarly oxides of manganese could be easily oxidized
or reduced at a relatively lower temperature than ZrO2.
Conclusions
The results of the present study uncovered the fact
that Pt/ZrO2 is a better catalyst for catalytic oxidation of
toluene in aqueous solution. It is free from additives,
promoters, and co-catalysts and can easily be separated
from reaction mixture by simple filtration and can be
reused. This gives us reasons to conclude that it is a
possible alternative for the purification of wastewater
containing toluene under mild conditions. Literature
suggests that toluene concentration in industrial waste-
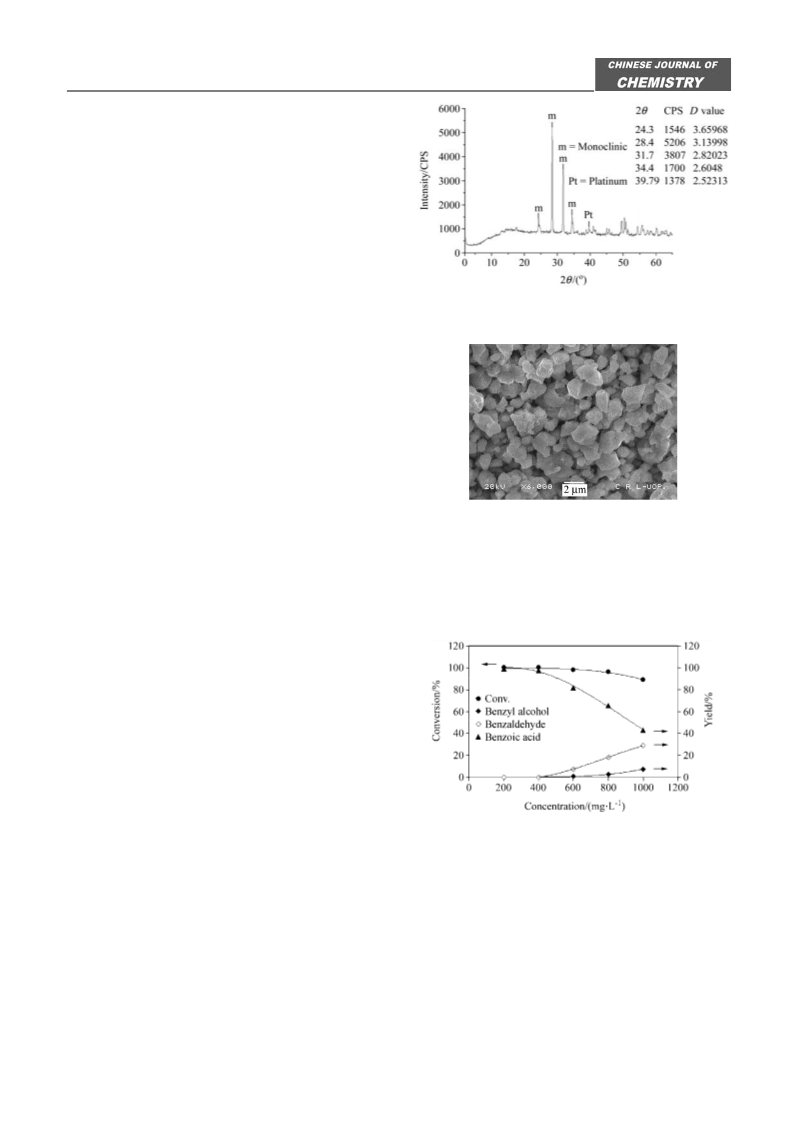
Figure 8 Effect of oxygen partial pressure on the conversion of
toluene in aqueous medium catalyzed by Pt/ZrO2 at 333 K. Reac-
tion conditions: catalyst (100 mg), solution volume (10 mL),
toluene concentration (200— 1000 mg•L ), stirring (900 r/min),
time (30 min).
-
1
water varies between 7—753 mg•L- depending on the
1
type of industry. Optimizing conditions for complete-
oxidation of toluene to benzoic acid in the above
men-tioned range are: time 30 min, temperature 333 K,
agitation 900 r/min, p(O2) ca. 101 kPa, catalyst amount
100 mg. The main advantage of the above optimal con-
dition allows the treatment of wastewater at a lower
temperature (333 K). Among the various models for the
kinetics of surface catalyzed reactions the Eley-Rideal
mechanisms gave a better fit for experimental results.
References
1
2
3
Enright, A. M.; Collins, G.; Flaherty, V. O. Water Res. 2007,
41, 1465.
eco-usa.net/toxics/toluene.shtml.
Drinking Water Contaminants Toluene. Free Drinking Water.
taminant/toluene-contaminants-removal-water.htm
Langwaldt, J. H.; Puhakka, J. Environ. Pollut. 2000, 107,
187.
De Nardi, I. R.; Varesche, M. B.; Zaiat, M.; Foresti, E. Wa-
ter Sci. Technol. 2002, 45, 175.
De Nardi, I. R.; Ribeiro, R.; Zaiat, M.; Foresti, E. Proc.
Biochem. 2005, 40, 587.
Christoskova, S. T.; Stoyanova, M. Water Res. 2002, 36,
2297.
Christoskova, S.; Danova, N.; Georgieva, M.; Argirov, O.;
Mehandjiev, D. Appl. Catal. A 1995, 128, 219.
Stenstrom, M. K.; Cardinal, L.; Libra, L. Environ. Prog.
1989, 8, 107.
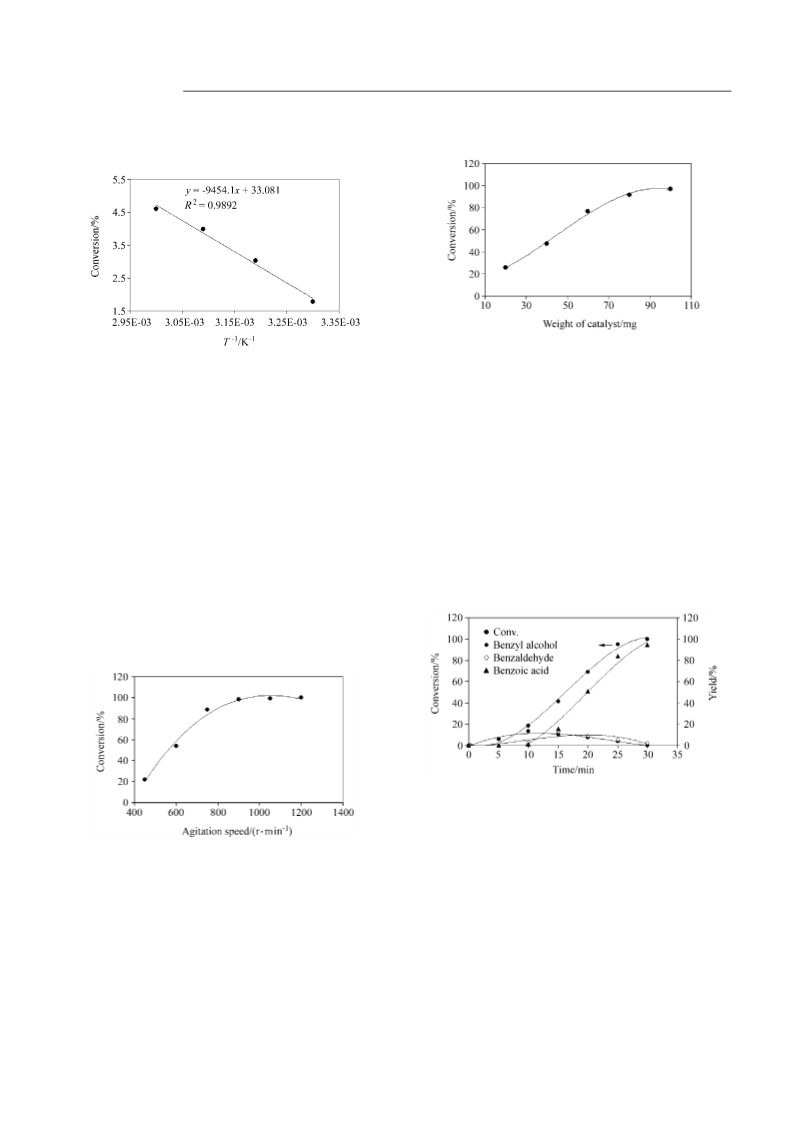
Figure 9 Rate of toluene conversion vs. toluene concentration.
Data for toluene conversion from Figure 3 was used.
4
5
6
7
8
9
Figure 10 Plot of calculated conversion vs. experimental con-
version. Data (from Figure 8) for the effect of oxygen partial
pressure effect on conversion of toluene was analyzed according
to E-R mechanism using Eq. (3b).
10 Mantzavinos, D.; Sahibzada, M.; Livingston, A.; Metcalfe,
I.; Hellgardt, K. Catal. Today 1999, 53, 93.
11 Vigneron, S.; Deprelle, P.; Hermia, J. Catal. Today. 1996, 27,
229.
12 Sharma, R. K.; Zhou, B.; Toung, S.; Chuang, K. Ind. Eng.
Chem. Res. 1995, 34, 4310.
13 Ilyas, M.; Sadiq, M. Chem. Eng. Technol. 2007, 30, 1391.
14 Makwana, V. D.; Son, Y. C.; Howell, A. R.; Suib, S. L. J.
Catal. 2002, 210, 46.
15 Ordóñez, S.; Bello, L.; Sastre, H.; Rosal, R.; Diez, F. V.
Appl. Catal. B 2002, 38, 139.
surface could be envisaged. Ordóñez et al.15 have re-
ported the Mars-van Krevelen mechanism for the deep
oxidation of toluene, benzene and n-hexane catalyzed
by platinum on γ-alumina. However, in that case the
reaction was taking place in the gas phase at a higher
temperature and higher gas phase concentration of
toluene. Similarly Makwana et al.14 have observed
Mars-van Krevelen mechanism for benzyl alcohol oxi-
dation in toluene catalyzed by OMS-2 type manganese
oxide at 383 K. In both the above cases benzyl alcohol
(E1001265 Sun, H.; Fan, Y.)
2220
© 2010 SIOC, CAS, Shanghai, & WILEY-VCH Verlag GmbH & Co. KGaA, Weinheim
Chin. J. Chem. 2010, 28, 2216— 2220

 Mohammad, Sadiq
Mohammad, Sadiq
 Mohammad, Ilyas
Mohammad, Ilyas