Full Paper
clooctene (95 %), norbonene (95 %), trans-stilbene (96 %), styrene
(99 %), and 1-octene (98 %) were obtained from TCI, Fluka, Merck,
and Aldrich, respectively. Deionized water, used in all experiments,
was obtained from a Nanopure® Analytical Deionization Water Sys-
tem with an electronic resistance of ≥ 18.2 Ω. All other solvents
were analytical (AR) grade and were purchased from RCI Labscan.
Acknowledgments
Financial support from the Center of Excellence for Innovation
in Chemistry (PERCH-CIC), the Office of the Higher Education
Commission, the Ministry of Education, and Mahidol University
through the New Researcher Grants are acknowledged. The au-
thors are also grateful to Dr. Decha Dechtrirat from Kasetsart
University for assistance with XRD measurements.
Preparation of MIL-53(Al)-NH2 and MIL-53[Cu]
The preparation of MIL-53(Al)-NH2 was adapted from the litera-
ture.[12b] H2N-H2BDC (0.24 g) was dispersed in water (56 mL) in a
Keywords: Metal–organic frameworks · Olefins ·
Postsynthetic modification · Oxidation · Copper
Teflon™ cup. Then, NaOH (1.12 mL, 0.4
M) and AlCl3·6H2O (2.2 mL,
0.4 ) were added. The Teflon™ cup was sealed in a stainless-steel
M
reactor that was placed in an oven at 110 °C for 48 h. After that,
the resulting product was isolated by filtration and washed with
water. This as-synthesized product was then activated in DMF at
130 °C for 18 h. Finally, pale-yellow powder was obtained after filtra-
tion and was dried at 130 °C overnight.
[1] a) S. S. Stahl, Angew. Chem. Int. Ed. 2004, 43, 3400–3420; Angew. Chem.
2004, 116, 3480; b) T. Punniyamurthy, L. Rout, Coord. Chem. Rev. 2008,
252, 134–154; c) S. Caron, R. W. Dugger, S. G. Ruggeri, J. A. Ragan, D. H. B.
Ripin, Chem. Rev. 2006, 106, 2943–2989.
[2] a) T. Punniyamurthy, S. Velusamy, J. Iqbal, Chem. Rev. 2005, 105, 2329–
2364; b) B. Huang, C. Fan, C. Pan, A. Zheng, X. Ma, Y. Li, J. Zhang, Y. Sun,
Powder Technol. 2017, 315, 258–269; c) M. Sankaralingam, Y.-M. Lee, W.
Nam, S. Fukuzumi, Inorg. Chem. 2017, 56, 5096–5104; d) G. Tuci, G. Giam-
bastiani, S. Kwon, P. C. Stair, R. Q. Snurr, A. Rossin, ACS Catal. 2014, 4,
1032–1039; e) D. T. Bregante, D. W. Flaherty, J. Am. Chem. Soc. 2017, 139,
6888–6898; f) W. Wu, H. Jiang, Acc. Chem. Res. 2012, 45, 1736–1748; g)
C. Parmeggiani, F. Cardona, Green Chem. 2012, 14, 547–564.
[3] a) S. D. McCann, S. S. Stahl, Acc. Chem. Res. 2015, 48, 1756–1766; b) S. E.
Allen, R. R. Walvoord, R. Padilla-Salinas, M. C. Kozlowski, Chem. Rev. 2013,
113, 6234–6458.
Copper-functionalized MIL-53 or MIL-53[Cu] was prepared by micro-
wave-assisted one-step synthesis. In a microwave reaction vessel,
MIL-53(Al)-NH2 (0.1 g) was added to solution of Cu(acac)2 (5 mL,
0.089 g, 0.34 mmol) in CHCl3. The vessel was placed in the micro-
wave reactor (CEM, Discover S) and heated to 60 °C, at 2 bar and
100 W for 1 h. The solid product was filtered out, washed with
CHCl3 (5 × 10 mL), and dried at 80 °C overnight to afford the light-
green powder of MIL-53[Cu] (0.0724 g).
[4] Y. Li, T. B. Lee, T. Wang, A. V. Gamble, A. E. V. Gorden, J. Org. Chem. 2012,
77, 4628–4633.
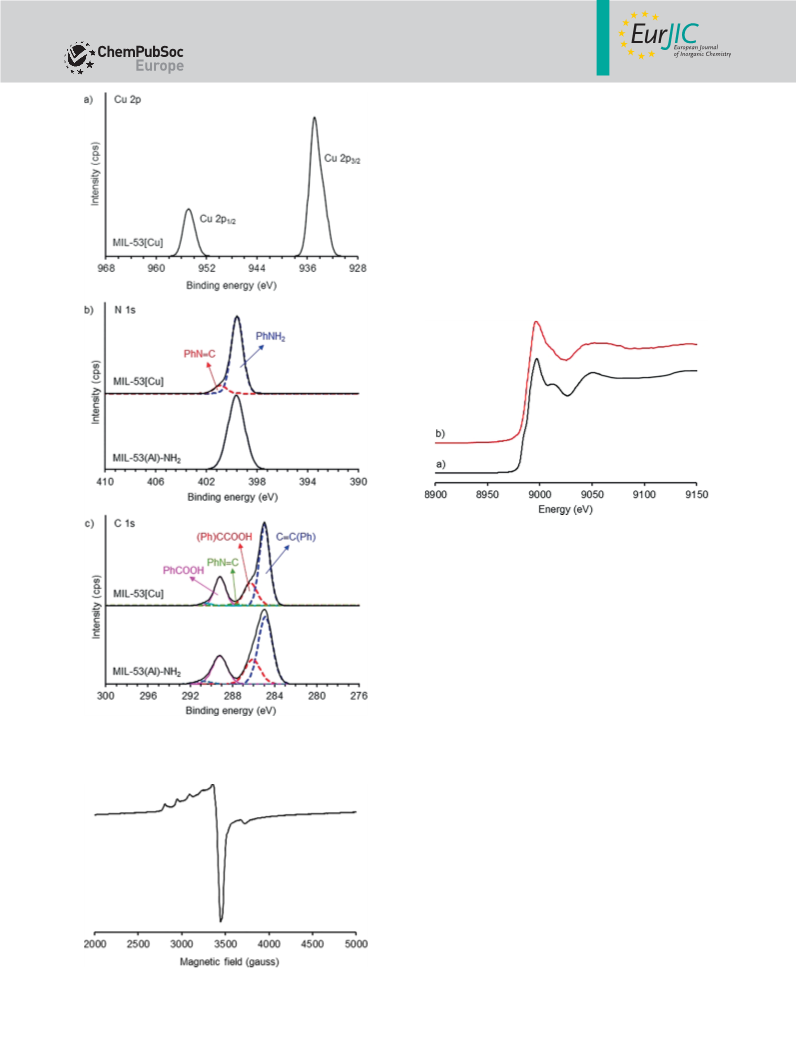
Characterization: XRD data were collected with a Bruker D8 Ad-
vance diffractometer using Cu-Kα radiation with a Ge crystal and
Johansson type monochromator (λ = 1.540619 Å); the measure-
ments were conducted at 40 kV and 40 mA, with a step size of
0.075° and a counting time of 1 s/step. EPR spectra were collected
with a Bruker e500 spectrometer at room temperature, with a fre-
quency of 9.86 GHz and microwave power of 2.02 W. Copper con-
tent in the samples was analyzed by ICP-OES (Spectro CIROSCCD),
with an emission line at 324.754 nm. XPS measurements were car-
ried out with an AXIS ULTRADLD instrument equipped with a
monochromated 150 W Al-Kα; binding-energy (BE) values were ob-
tained by curve fitting and were referenced to the C 1s band at
284.6 eV. X-ray absorption spectroscopy (XAS) studies were carried
out at Beamline-8 of the Synchrotron Light Research Institute (SLRI),
Nakhon Ratchasima, Thailand.[35] The storage ring was operated at
1.2 GeV, with a beam current of 80–150 mA. The energy was cali-
brated by using copper foil as a reference at 8979 eV before XAS
data were collected. The experiments were conducted at room tem-
perature in the fluorescence mode, with a 13-element germanium
detector (Canberra). The synchrotron beam was monochromated
using a Ge(220) double-crystal monochromator with an energy res-
olution (ΔE/E) of 2 × 10–4. The XAS spectra were averaged for four
scans. The resulting data were baseline-corrected and edge-normal-
ized using ARTENA software.[36]
[5] a) M. Nandi, P. Roy, H. Uyama, A. Bhaumik, Dalton Trans. 2011, 40, 12510–
12518; b) S. Jana, B. Dutta, R. Bera, S. Koner, Langmuir 2007, 23, 2492–
2496; c) M. Selvaraj, V. Narayanan, S. Kawi, Microporous Mesoporous Ma-
ter. 2010, 132, 494–500; d) P. Roy, M. Nandi, M. Manassero, M. Ricco, M.
Mazzani, A. Bhaumik, P. Banerjee, Dalton Trans. 2009, 9543–9554.
[6] S. Natarajan, P. Mahata, Chem. Soc. Rev. 2009, 38, 2304–2318.
[7] a) C. Janiak, Dalton Trans. 2003, 2781–2804; b) L. J. Murray, M. Dincă,
J. R. Long, Chem. Soc. Rev. 2009, 38, 1294–1314; c) M. Dincă, J. R. Long,
Angew. Chem. Int. Ed. 2008, 47, 6766–6779; Angew. Chem. 2008, 120,
6870; d) R. E. Morris, P. S. Wheatley, Angew. Chem. Int. Ed. 2008, 47, 4966–
4981; Angew. Chem. 2008, 120, 5044; e) X. Lin, J. Jia, P. Hubberstey, M.
Schroder, N. R. Champness, CrystEngComm 2007, 9, 438–448; f) J. L. C.
Rowsell, O. M. Yaghi, Angew. Chem. Int. Ed. 2005, 44, 4670–4679; Angew.
Chem. 2005, 117, 4748.
[8] a) G. J. Halder, C. J. Kepert, B. Moubaraki, K. S. Murray, J. D. Cashion,
Science 2002, 298, 1762–1765; b) L. G. Beauvais, M. P. Shores, J. R. Long,
J. Am. Chem. Soc. 2000, 122, 2763–2772; c) J. A. Real, E. Andrés, M. C.
Muñoz, M. Julve, T. Granier, A. Bousseksou, F. Varret, Science 1995, 268,
265–267.
[9] S. Ma, D. Sun, M. Ambrogio, J. A. Fillinger, S. Parkin, H.-C. Zhou, J. Am.
Chem. Soc. 2007, 129, 1858–1859.
[10] a) P. Horcajada, C. Serre, M. Vallet-Regí, M. Sebban, F. Taulelle, G. Férey,
Angew. Chem. Int. Ed. 2006, 45, 5974–5978; Angew. Chem. 2006, 118,
6120; b) M. Vallet-Regí, F. Balas, D. Arcos, Angew. Chem. Int. Ed. 2007, 46,
7548–7558; Angew. Chem. 2007, 119, 7692; c) G. Ferey, Chem. Soc. Rev.
2008, 37, 191–214.
Oxidation of Cyclohexene: Cyclohexene (2.4 mmol), TBHP or iso-
butyraldehyde, the MIL-53[Cu] catalyst, and n-nonane (0.24 mmol,
internal standard) were added into a stainless-steel reactor contain-
ing solvent (24 mL). The reactor was purged with an appropriate
gas for five cycles and then pressurized to the desired pressure. The
reaction mixture was stirred and heated at 50 °C. After a certain
time, the reaction was quickly cooled to room temperature, the
catalyst was filtered out, and the substrate conversion and the prod-
uct selectivity were determined by GCMS (Agilent 7890A with 5975
MSD), equipped with an HP-5 capillary column (polydimethylsilox-
ane with 5 % phenyl group, 20 m length, 0.25 mm i.d., 0.25 μm film
thickness).
[11] a) A. Corma, H. García, F. X. Llabrés i Xamena, Chem. Rev. 2010, 110,
4606–4655; b) J. Lee, O. K. Farha, J. Roberts, K. A. Scheidt, S. T. Nguyen,
J. T. Hupp, Chem. Soc. Rev. 2009, 38, 1450–1459; c) D. Farrusseng, S.
Aguado, C. Pinel, Angew. Chem. Int. Ed. 2009, 48, 7502–7513; Angew.
Chem. 2009, 121, 7638; d) Q. Yang, W. Liu, B. Wang, W. Zhang, X. Zeng,
C. Zhang, Y. Qin, X. Sun, T. Wu, J. Liu, F. Huo, J. Lu, Nat. Commun. 2017,
8, 14429; e) W. Liu, J. Huang, Q. Yang, S. Wang, X. Sun, W. Zhang, J. Liu,
F. Huo, Angew. Chem. Int. Ed. 2017, 56, 5512–5516; Angew. Chem. 2017,
129, 5604–5608; f) B. Wang, W. Liu, W. Zhang, J. Liu, Nano Res. 2017, 10,
3826–3835; g) T. Zhang, W. Liu, G. Meng, Q. Yang, X. Sun, J. Liu, Chem-
CatChem 2017, 9, 1771–1775.
[12] a) S. M. Cohen, Chem. Rev. 2012, 112, 970–1000; b) T. Ahnfeldt, D. Gunzel-
mann, T. Loiseau, D. Hirsemann, J. Senker, G. Férey, N. Stock, Inorg. Chem.
Eur. J. Inorg. Chem. 2018, 703–712
711
© 2018 Wiley-VCH Verlag GmbH & Co. KGaA, Weinheim

 Chotmongkolsap, Pannapat
Chotmongkolsap, Pannapat
 Bunchuay, Thanthapatra
Bunchuay, Thanthapatra
 Klysubun, Wantana
Klysubun, Wantana
 Tantirungrotechai, Jonggol
Tantirungrotechai, Jonggol