Green Chemistry
Paper
1
8
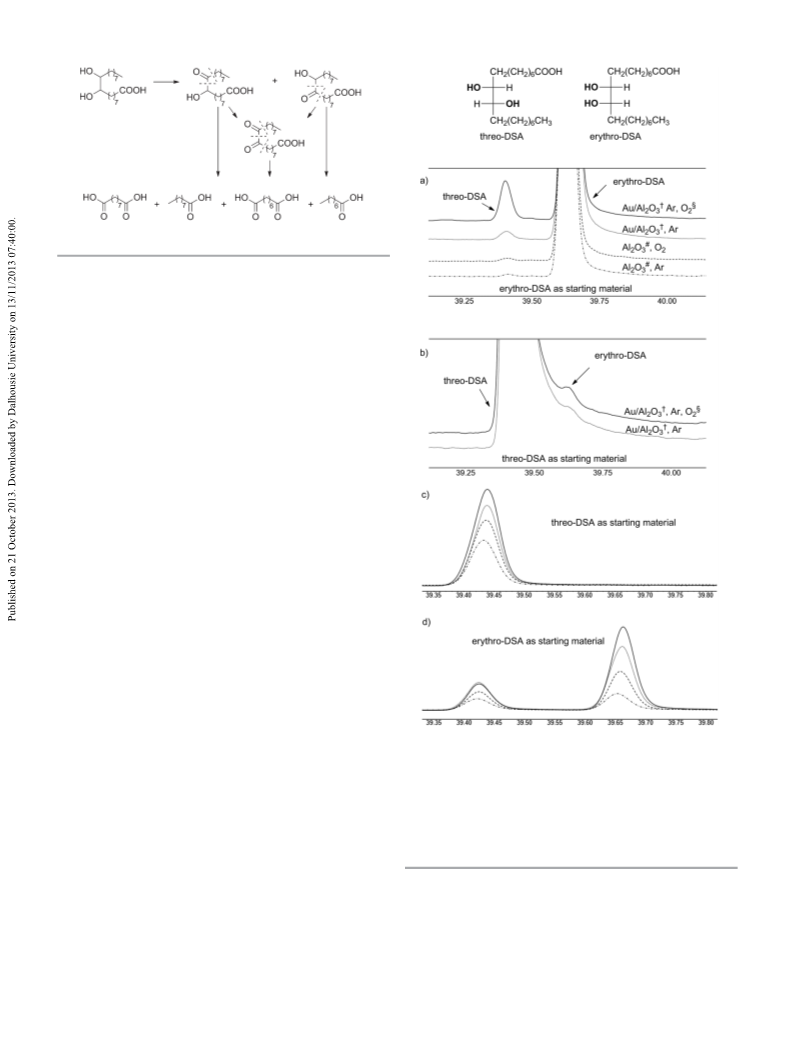
show different reactivity. Probably the cleavage of erythro-DSA 0.25 M Na OH solution were placed in a 5 mL glass vial
in a mixture of both diastereomers goes along with a prelimi- (equipped with a screw cap, cannula and stirrer), which was
nary isomerization of threo to erythro diastereomer. The put into the Parr autoclave. The autoclave was flushed three
problem of aging of the gold catalysts used was also studied. times with argon and the suspension was heated to a reaction
Storage conditions (such as the atmosphere and the presence temperature of 80 °C using a heating bath. The autoclave was
2
or absence of light) have a strong influence on the gold particle pressurized with 5 bar O for a reaction time of 260 min (YAA =
growth and in consequence on the catalyst activity. The clea- 58%). After the reaction was complete, the samples were
vage of DSA to AA and PA with the aged catalysts applied as a treated as described above.
test reaction was repeated every three months within one year.
Cleavage of HK and DK: both these experiments were
All catalysts lost activity; the highest yield of AA after a catalyst carried out in 5 mL glass vials using 0.1 mmol substrate (HK
storage time of 1 year was received with a catalyst which was or DK) 0.2 mol% gold (1 wt% Au/Al O ) and 2 mL of 0.25 M
2
3
stored under argon in the dark. The reason for the loss was the NaOH solution. The continuing implementation was per-
disappearance of very small gold particles of ≤1 nm size due formed as already described in the preceding paragraph. The
to agglomeration. Comparing all catalysts used in the oxidative yields of AA were 26% (cleavage of HK) and 28% (cleavage of
cleavage of DSA, the best results after a storage time of DK).
3
−
months were obtained using the catalyst stored in vacuum at
15 °C in the dark. A conclusion from this behavior for
further investigations may be that the catalysts have to be pre-
pared freshly before use. Probably, also a higher activity of
Acknowledgements
gold catalysts on other oxidic supports as described in ref. 9 Anna Kulik gratefully acknowledges the financial support from
might be observed using freshly synthesized catalysts. LIKAT.
Methods of stabilization of gold particles, possibly by the for-
mation of bimetallic catalysts, should be explored.
References
1
B. Cornils and P. Lappe, in Ullmann’s Encyclopedia of Indus-
trial Chemistry, Electronic Release, Wiley-VCH, Weinheim,
Germany, 7th edn, 2010.
Experimental
9
18
1
2 3
wt% Au/Al O catalyst, 9(10)-hydroxy-10(9)-oxostearic acid
1
9
and 9,10-dioxostearic acid were prepared according to the
improved literature procedures (see ESI†).
A typical reaction procedure for the oxidative cleavage of
DSA to AA and PA is as follows: in typical experiments, 1 mmol
DSA, a 0.2 mol% gold catalyst (1 wt% Au/Al
NaOH and 20 mL H O were placed in a 100 mL Parr autoclave.
2 http://www.matweb.com (September 30, 2013).
3 T. Roloff, U. Nagorny and U. Erkens, Kunststoffe, 2004, 5,
104–106.
4 Saechtling Kunststoff Taschenbuch, ed. E. Baur, S. Brink-
mann, T. A. Osswald and E. Schmachtenberg, Hanser,
Munich, 2007.
2 3
O ), 5 mmol
2
The autoclave was flushed three times with argon and the sus-
pension was heated to a reaction temperature of 80 °C. The
5 Plastics Additive Handbook, ed. H. Zweifel, Hanser, Munich,
2009.
autoclave was pressurized with 5 bar O
2
for the reaction time
6 A. Köckritz and A. Martin, Eur. J. Lipid Sci. Technol., 2011,
113, 83–91.
7 A. S. K. Hashmi and G. J. Hutchings, Angew. Chem., Int. Ed.,
2006, 45, 7896–7936.
of 260 min. After the reaction was finished, the catalyst was
separated from the liquid phase by filtration. The conversion
of DSA and the yields of products were determined by GC-MS
after derivatization. Experiments using isotope labelled
sodium hydroxide and oxygen were measured by MS-ESI.
8 For selected examples of homogeneous and heterogeneous
gold-catalysed oxidation reactions, see: (a) D. I. Enache,
J. K. Edwards, P. Landon, B. Solsona-Espriu, A. F. Carley,
A. A. Herzing, M. Watanabe, C. J. Kiely, D. W. Knight and
G. J. Hutchings, Science, 2006, 311, 362–365; (b) A. Abad,
P. Concepción, A. Corma and H. García, Angew. Chem., Int.
Ed., 2005, 44, 4066–4069; (c) B. Guan, D. Xing, G. Cai,
X. Wan, N. Yu, Z. Fang, L. Yang and Z. Shi, J. Am. Chem.
Soc., 2005, 127, 18004–18005; (d) A. S. K. Hashmi,
C. Lothschütz, M. Ackermann, R. Doepp, S. Anantharaman,
B. Marchetti, H. Bertagnolli and F. Rominger, Chem.–Eur.
J., 2010, 16, 8012–8019.
1
8
16
Reaction of DSA using
O
2
: in order to ensure that no
O
2
is contained in the reaction mixture all arrangements of the
experiments were carried out in the glovebox under an Ar
atmosphere. In this experiment 2 mmol DSA, 0.2 mol% gold
(
1 wt% Au/Al O ), and 40 mL of 0.25 M NaOH (refluxed under
2 3
1
6
Ar for removal of
2
O ) solution were placed in a 100 mL Parr
autoclave. The suspension was heated to a reaction tempera-
ture of 80 °C. The autoclave was pressurized with 0.5 bar O .
For temporal tracking experiments samples of 1 mL were
taken after 1 h (YAA = 4%), 2 h (YAA = 8%), 4 h (YAA = 22%), 6 h
1
8
2
(
YAA = 35%), and 24 h (YAA = 63%). After the reaction was com-
9 A. Kulik, A. Janz, M.-M. Pohl, A. Martin and A. Köckritz,
Eur. J. Lipid Sci. Technol., 2012, 114, 1327–1332.
2
O: in this study, 10 S. Warwel and M. Rüsch gen Klaas, Lipid Technol., 1997, 9,
plete, the samples were treated as described above.
1
8
18
Reaction of DSA using Na OH/H
0
.1 mmol DSA, 0.4 mol% gold (1 wt% Au/Al O ), and 2 mL of
10–14.
2
3
This journal is © The Royal Society of Chemistry 2013
Green Chem.

 Kulik, Anna
Kulik, Anna
 Martin, Andreas
Martin, Andreas
 Pohl, Marga-Martina
Pohl, Marga-Martina
 Fischer, Christine
Fischer, Christine
 Koeckritz, Angela
Koeckritz, Angela