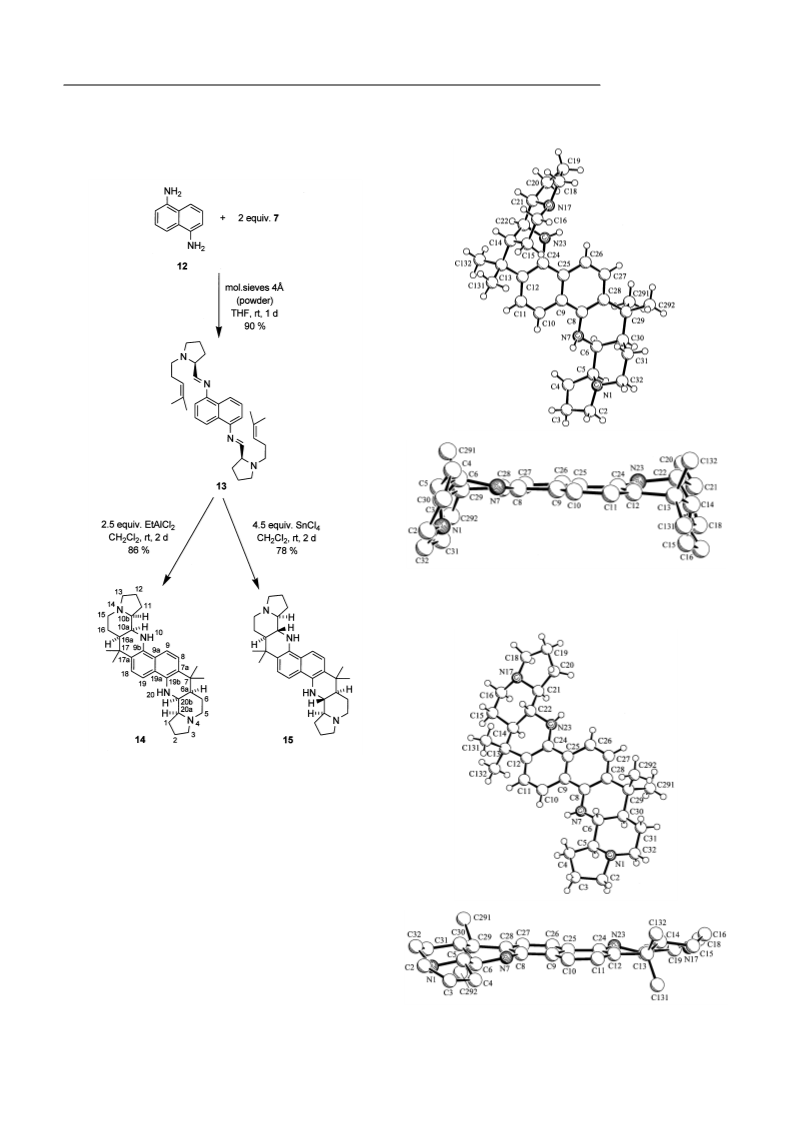
O. Temme, T. Dickner, S. Laschat, R. Fröhlich, S. Kotila, K. Bergander
FULL PAPER
NMR (50 MHz, CDCl3): δ ϭ 143.9, 140.3, 131.9, 129.9, 129.2,
the procedure described for 9, cyclization of 7 (6.55 g, 36.0 mmol)
129.1, 128.5, 126.7, 115.0, 114.9 (C-1Ј, C-2Ј, C-3Ј, C-4Ј, C-5Ј, C- and 12 (5.75 g, 36.0 mmol) gave after work-up and flash chroma-
6Ј, C-8, C-8a, C-9, C-10, C-11, C-11a), 66.9 (C-12b), 53.9, 52.3 (C-
tography on SiO2 (hexane/CHCl3/triethylamine, 5:3:1) 1.74 g (3.60
3, C-5), 46.6 (C-12a), 44.8 (C-6a), 40.2 (CH2), 35.1 (C-7), 33.6, 26.0
mmol, 10%) of a violet solid (14) as the first fraction and 1.80 g
(C-13, C-14), 24.7, 22.8, 20.8 (C-1, C-2, C-6). Ϫ MS (70 eV); m/z (5.60 mmol, 16%) of a red solid (31) as the second fraction; m.p.
22
(%): 361 (22) [Mϩ], 346 (10) [Mϩ Ϫ CH3], 277 (15), 249 (14), 106 217°C; [α]D ϭ ϩ93.0 (c ϭ 0.10 in CH2Cl2). Ϫ IR (KBr): ν˜ ϭ
(36), 84 (100) [C5H10Nϩ], 69 (18). Ϫ C24H31N3: calcd. 361.2518; 3343 cmϪ1, 2989, 2928, 2792, 1622, 1517, 1432, 1401, 786, 734. Ϫ
found 361.2525 (MS); calcd. C 79.25, H 9.05, N 11.70; found C 1H NMR (600 MHz, C6D6): δ ϭ 7.39 (d, J ϭ 9.0 Hz, 1 H, 9-H),
79.15, H 9.01, N 11.84.
7.36 (d, J ϭ 8.4 Hz, 1 H, 11-H), 7.23 (dd, J ϭ 7.2/8.4 Hz, 1 H, 12-
H), 7.19 (d, J ϭ 9.0 Hz, 1 H, 8-H), 6.70 (d, J ϭ 7.2 Hz, 1 H, 13-
H), 4.36 (d, J ϭ 4.2 Hz, 1 H, NH), 4.07 (br. s, 2 H, NH2), 3.77 (s,
1 H, 14a-H), 3.10 (dd, J ϭ 9.0/8.4 Hz, 1 H, 3-Heq), 3.05 (ddd, J ϭ
12.0/2.2/2.4 Hz, 1 H, 5-Heq), 2.21Ϫ2.18 (m, 1 H, 14b-H), 2.13Ϫ2.06
(m, 2 H, 3-Hax, 2-Heq), 1.98 (ddd, J ϭ 12.0/12.0/3.0 Hz, 1 H, 5-
Hax), 1.95Ϫ1.87 (m, 2 H, 2-Hax, 1-Heq), 1.84Ϫ1.80 (m, 1 H, 1-Hax),
1.55 (ddd, J ϭ 12.0/3.0/2.4 Hz, 1 H, 6-Heq), 1.41 (s, 3 H, 15-H),
1.38Ϫ1.37 (m, 1 H, 6a-H), 1.36 (s, 3 H, 16-H), 1.24 (ddd, J ϭ 12.0/
12.0/2.2 Hz, 1 H, 6-Hax). Ϫ 13C NMR (150 MHz, C6D6): δ ϭ 142.3
(C-10), 137.3 (C-13b), 125.3 (C-7a), 125.0 (C-12), 124 (C-9a), 123.8
(C-11), 122.6 (C-13a), 111.7, 110.3 (C-8, C-9), 109.2 (C-13), 67.1
(C-14b), 54.0 (C-3), 52.3 (C-5), 47.2 (C-14a), 44.2 (C-6a), 35.6 (C-
7), 33.6, 26.5 (C-15, C-16), 25.2 (C-2), 22.8 (C-6), 21.3 (C-1). Ϫ
MS (70 eV); m/z (%): 321 (62) [Mϩ], 306 (23) [Mϩ Ϫ CH3], 237
(14) [Mϩ Ϫ C5H10Nϩ], 209 (18), 97 (32), 84 (100) [C5H10Nϩ], 69
(27), 57 (31). Ϫ C21H27N3: calcd. 321.2205; found 321.2196 (MS).
1,3,5-Tris{(6aS,12aS,12bS)-4-[9-(1,2,3,5,6,6a,7,12,12a,12b-
decahydro-7,7-dimethylindolizino[8,7-b]quinolyl)methyl]phenyl-
aminomethylene}benzene (28): A solution of 1,3,5-triformylbenzene
(27, 16.2 mg, 0.10 mmol) and amine 25 (108 mg, 0.30 mmol) in
toluene (10 ml) was treated with 4-A molecular-sieve beads (1.00
g) and the mixture was refluxed for 20 d. After filtration through
Celite, the solvent was removed in vacuo to give 83 mg (70%) of a
˚
22
yellow amorphous solid; [α]D ϭ ϩ114.3 (c ϭ 1.00 in CH2Cl2). Ϫ
IR (KBr): ν˜ ϭ 2957 cmϪ1, 2921, 2870, 1622, 1501. Ϫ 1H NMR
(200 MHz, C6D6): δ ϭ 8.50 (s, 3 H, 2-H), 8.19 (s, 3 H, HCϭN),
7.30Ϫ7.10 (m, 15 H, 8Љ-H, 2Ј-H, 3Ј-H, 5Ј-H, 6Ј-H), 6.92 (d, J ϭ
8.1 Hz, 3 H, 10Љ-H), 6.68 (d, J ϭ 8.1 Hz, 3 H, 11Љ-H), 3.91Ϫ3.85
(m, 9 H, CH2, NH), 3.53 (s, 3 H, 12aЉ-H), 2.90Ϫ2.75 (m, 6 H, 3Љ-
Heq, 5Љ-Heq), 1.80Ϫ1.10 (m, 30 H, 1Љ-H, 2Љ-H, 3Љ-Hax, 5Љ-Hax, 6Љ-
H, 6aЉ-H, 12bЉ-H), 1.25, 1.22 (s, 18 H, 13Љ-H, 14Љ-H). Ϫ 13C NMR
(50 MHz, C6D6): δ ϭ 158.0 (HCϭN), 149.9 (C-11aЉ), 141.6, 141.2,
137.9, 131.4, 130.0, 129.4, 127.5, 126.8, 121.6, 116.3, 115.3 (C-1,
C-2, C-1Ј, C-2Ј, C-3Ј, C-4Ј, C-5Ј, C-6Ј, C-8Љ, C-7aЉ, C-9Љ, C-10Љ, C-
11Љ), 67.3 (C-12bЉ), 54.3, 52.6 (C-3Љ, C-5Љ), 47.4 (C-12aЉ), 44.8 (C-
6aЉ), 41.6 (CH2), 35.7 (C-7Љ), 33.4, 26.5 (C-13Љ, C-14Љ), 25.2, 23.3,
21.5 (C-1Љ, C-2Љ, C-6Љ). Ϫ TOF MS (LDI); m/z (%): 1191 [Mϩ]. Ϫ
C81H93N9 (1192.7): calcd. C 81.57, H 7.86, N 10.57; found C 81.38,
H 8.02, N 10.60.
(6aS,14aS,14bS)-10-(4-Formylbenzylidene)amino-1,2,3,5,6,6a,
7,14,14a,14b-decahydro-7,7-dimethylbenzo[h]indolizino[8,7-b]-
quinoline (32): According to the procedure described for 30, con-
densation of 29 (201 mg, 1.50 mmol) and 31 (1.29 g, 4.00 mmol)
gave after work-up and flash chromatography (isohexane/CHCl3/
triethylamine, 8:3:1) 219 mg (0.30 mmol, 20%) of a red solid (33)
as the first fraction and 128 mg (0.29 mmol, 19%) of a red solid
22
(32) as the second fraction; m.p. 110°C; [α]D ϭ ϩ66.0 (c ϭ 0.10
in CH2Cl2). Ϫ IR (KBr): ν˜ ϭ 3015 cmϪ1, 2985, 2779, 2748, 2729,
1701, 1621, 1605, 1511, 1502, 1466Ϫ1442, 1381Ϫ1366, 829, 800,
745. Ϫ 1H NMR (300 MHz, C6D6): δ ϭ 9.65 (s, 1 H, HCϭO),
8.13 (s, 1 H, HCϭN), 8.12 (d, J ϭ 8.8 Hz, 1 H, 8-H), 8.04 (d, J ϭ
8.3 Hz, 1 H, 11-H), 7.75 (d, J ϭ 8.1 Hz, 2 H, 2Ј-H, 6Ј-H), 7.55 (d,
J ϭ 8.1 Hz, 2 H, 3Ј-H, 5Ј-H), 7.51 (d, J ϭ 8.8 Hz, 1 H, 9-H), 7.26
(dd, J ϭ 8.3/7.1 Hz, 1 H, 12-H), 6.79 (d, J ϭ 7.1 Hz, 1 H, 13-H),
4.38 (d, J ϭ 5.5 Hz, 1 H, NH), 3.59 (s, 1 H, 14a-H), 2.89Ϫ2.80 (m,
2 H, 3-Heq, 5-Heq), 2.15Ϫ2.05 (m, 1 H, 14b-H), 1.91Ϫ1.88 (m, 2
H, 3-Hax, 2-Heq), 1.80Ϫ1.45 (m, 5 H, 1-H, 2-Hax, 5-Hax, 6-Heq),
1.28 (s, 3 H, 15-H), 1.27 (s, 3 H, 16-H), 1.20Ϫ1.00 (m, 2 H, 6-Hax,
6a-H). Ϫ 13C NMR (75 MHz, C6D6): δ ϭ 191.3 (HCϭO), 158.7
(HCϭN), 149.9 (C-4Ј), 142.0 (C-10), 138.8 (C-13b), 138.0 (C-1Ј),
129.8, 129.3 (C-2Ј, C-6Ј, C-3Ј, C-5Ј), 129.3, 127.1, 126.3 (C-7a, C-
9a, C-13a), 125.8, 125.3, 120.7 (C-11, C-12, C-13), 115.3, 112.9 (C-
8, C-9), 67.6 (C-14b), 54.7, 52.9 (C-3, C-5), 48.1 (C-14a), 44.8 (C-
6a), 36.4 (C-7), 34.0 (C-15), 27.2 (C-16), 26.0, 23.6, 22.2 (C-2, C-
1,4-Bis{(6aS,12aS,12bS)-4-[9-(1,2,3,5,6,6a,7,12,12a,12b-de-
cahydro-7,7-dimethylindolizino[8,7-b]quinolyl)methyl]phenylamino-
methylene}benzene (30): To a solution of terephthalaldehyde (29,
67 mg, 0.50 mmol) and indolizino[3,4-b]quinoline (25, 321 mg, 1.00
˚
mmol) in CH2Cl2 was added 4-A molecular-sieve beads (10 g) and
the mixture was stirred for 14 d at room temp. After filtration
through Celite and evaporation of the solvent, the crude product
was purified by flash chromatography on SiO2 (eluent: isohexane/
CHCl3/triethylamine, 5:3:1) to give 90 mg (22%) of a pale-yellow
22
solid; m.p. Ͼ300°C (dec.); [α]D ϭ ϩ170.0 (c ϭ 0.10 in CH2Cl2).
Ϫ IR (KBr): ν˜ ϭ 3036 cmϪ1, 3017, 2989Ϫ2787, 1622, 1602, 1501,
1472, 1384, 1299, 837, 810, 736. Ϫ 1H NMR (300 MHz, C6D6):
δ ϭ 8.14 (s, 2 H, HCϭN), 7.82 (s, 4 H, 2-H, 3-H, 5-H, 6-H), 7.18
(m, 10 H, 8-H, 2Ј-H, 3Ј-H, 5Ј-H, 6Ј-H), 6.91 (dd, J ϭ 8.1/2.0 Hz,
2 H, 8Љ-H), 6.54 (d, J ϭ 8.1 Hz, 2 H, 10Љ-H), 4.27 (s, 2 H, NH),
3.91 (s, 4 H, CH2), 3.75 (s, 2 H, 12aЉ-H), 3.54 (s, 2 H, 12bЉ-H),
2.87Ϫ2.80 (m, 4 H, 3Љ-Heq, 5Љ-Heq), 1.90Ϫ1.42 (m, 12 H, 3Љ-Hax,
2Љ-H, 1Љ-H, 5Љ-Hax), 1.25, 1.22 (s, 12 H, 13Љ-H, 14Љ-H), 1.15Ϫ0.98
(m, 2 H, 6aЉ-H). Ϫ 13C NMR (75 MHz, C6D6): δ ϭ 176.0 (HCϭ
N), 158.3 (C-3Ј, C-5Ј), 141.6, 141.0, 140.9, 134.2 (C-4Ј, C-11aЉ, C-
1, C-7aЉ), 130.1, 126.8, 116.5 (C-8Љ, C-9Љ, C-10Љ), 129.9 (C-1Ј, C-
7aЉ), 129.2, 121.4 (C-2, C-6, C-2Ј, C-6Ј), 67.2 (C-12bЉ), 54.3 (C-3Љ),
52.6 (C-5Љ), 47.4 (C-12aЉ), 44.9 (C-6aЉ), 41.5 (CH2), 35.7 (C-7Љ),
33.3 (C-13Љ), 26.5 (C-14Љ), 25.2 (C-2Љ), 23.3 (C-6Љ), 21.6 (C-1Љ). Ϫ
MS (70 eV); m/z (%): 820 (20) [Mϩ], 736 (6) [Mϩ Ϫ C5H10Nϩ],
111 (17), 97 (36), 84 (100) [C5H10Nϩ], 57 (65). Ϫ C56H64N6: calcd.
820.5192; found 820.5210 (MS); calcd. C 81.91, H 7.86, N 10.23;
found C 81.73, H. 7.90, N 9.86.
6, C-1). Ϫ MS (70 eV); m/z (%): 437 (58) [Mϩ], 422 (11) [Mϩ
Ϫ
CH3], 353 (6) [Mϩ Ϫ C5H10Nϩ], 325 (12), 218 (6), 149 (19), 111
(23), 97 (48), 84 (92) [C5H10Nϩ], 71 (49), 69 (55), 60 (100). Ϫ
C29H31N3O: calcd. 437.2467; found 437.2459 (MS).
1,4-Bis{(6aS,14aS,14bS)-10-(1,2,3,5,6,6a,7,14,14a,14b-deca-
hydro-7,7-dimethylbenzo[h]indolizino[8,7-b]quinolyl)amino-
methylene}benzene (33): Red solid, which was obtained during
22
preparation of 32 (see above); m.p. > 300°C (dec.); [α]D
ϭ
ϩ170.0° (c ϭ 0.10 in CH2Cl2). Ϫ IR (KBr): ν˜ ϭ 3015 cmϪ1, 2973,
2786, 1620, 1568, 1514, 1508, 1471, 1440, 1393, 1365, 813, 801,
1
748. Ϫ H NMR (300 MHz, C6D6): δ ϭ 8.27 (d, J ϭ 8.3 Hz, 2 H,
(6aS,14aS,14bS)-10-Amino-1,2,3,5,6,6a,7,14,14a,14b-decahydro- 8-H), 8.25 (s, 2 H, HCϭN), 8.10 (d, J ϭ 8.5 Hz, 2 H, 11-H), 7.95
7,7-dimethylbenzo[h]indolizino[8,7-b]quinoline (31): According to
(s, 4 H, 2Ј-H, 3Ј-H), 7.31 (dd, J ϭ 8.5/7.3 Hz, 2 H, 12-H), 6.86 (d,
Eur. J. Org. Chem. 1998, 651Ϫ659
658

 Temme, Oliver
Temme, Oliver
 Dickner, Tim
Dickner, Tim
 Laschat, Sabine
Laschat, Sabine
 Froehlich, Roland
Froehlich, Roland
 Kotila, Sirpa
Kotila, Sirpa
 Bergander, Klaus
Bergander, Klaus