Green Chemistry
Paper
Table 3 Observations of the substrate scope (% yield is given in terms
of % of the theoretically expected yield of the specific product)
Notes and references
1
M. Baumann and I. R. Baxendale, Beilstein J. Org. Chem.,
015, 11, 1194–1219.
2 R. Porta, M. Benaglia and A. Puglisi, Org. Process Res. Dev.,
015, 20, 2–25.
J. Britton and C. L. Raston, Chem. Soc. Rev., 2017, 46, 1250–
1271.
Sr. no.
1
Substrate
Oleic acid
Products (% yield)
2
Azelaic acid (89%)
Pelargonic acid (74%)
Azelaic acid (87%)
Pelargonic acid (76%)
Azelaic acid (86%)
2
2
3
4
5
Elaidic acid
3
Ricinoleic acid
Petroselinic acid
Erucic acid
3-Hydroxy pelargonic acid (70%)
4
B. Gutmann, D. Cantillo and C. O. Kappe, Angew. Chem.,
Int. Ed., 2015, 54, 6688–6728.
Adipic acid (72%)
Dodecanoic acid (76%)
Tridecanedioic acid (88%)
Pelargonic acid (74%)
5 D. L. Sparks, R. Hernandez, L. A. Estévez, N. Meyer and
T. French, J. Chem. Eng. Data, 2007, 52, 1246–1249.
6
7
8
9
D. L. Sparks, L. A. Estévez, R. Hernandez, K. Barlow and
T. French, J. Chem. Eng. Data, 2008, 53, 407–410.
S. R. Warwel and M. Rüsch gen. Klaas, Lipid Technol., 1997,
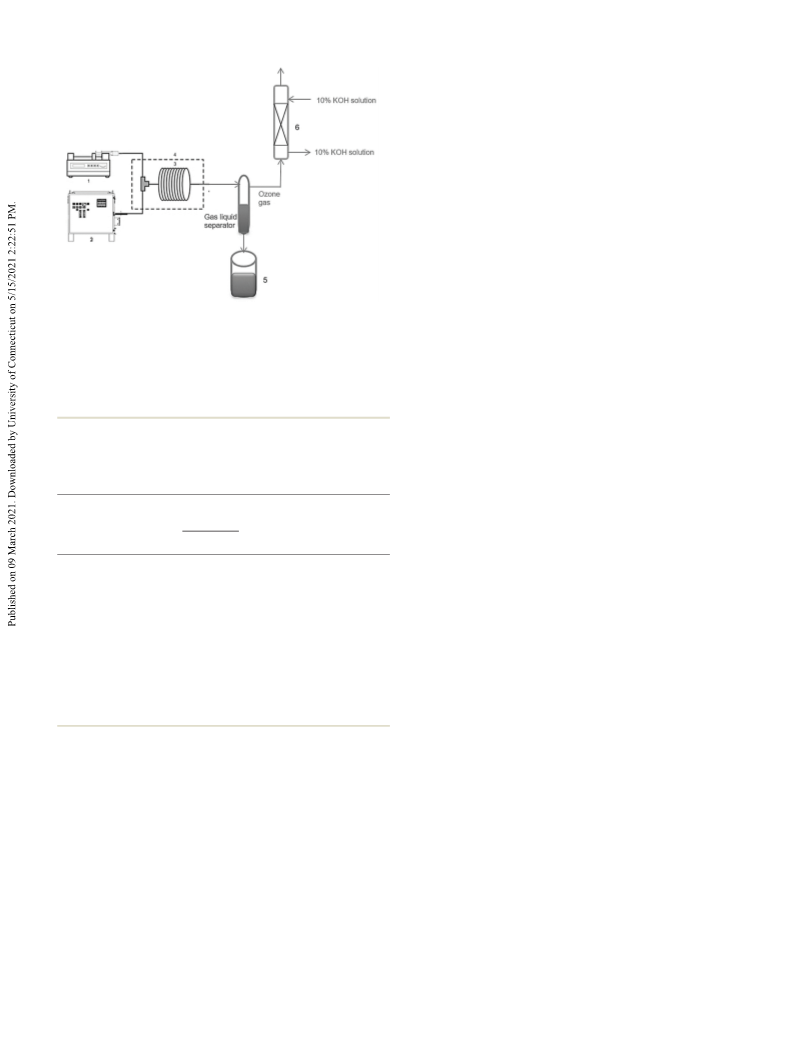
vidual substrates are given in the ESI†). Thus, a catalyst-free
continuous ozonolysis of fatty acids that results in 100% con-
version of the substrate with an almost stoichiometric quantity
of ozone in oxygen and complete recovery and recycling of sol-
vents makes this process a scalable green approach.
9
, 5.
X. Li, J. C. P. Syong and Y. Zhang, Green Chem., 2018, 20,
619–3624.
A. S. K. Hashmi and G. J. Hutchings, Angew. Chem., Int. Ed.,
006, 45, 7896–7936.
3
2
1
1
1
1
1
1
1
1
1
0 A. Kulik, A. Janz, M. M. Pohl, A. Martin and A. Köckritz,
Eur. J. Lipid Sci. Technol., 2012, 114, 1327–1332.
1 A. Kulik, A. Martin, M.-M. Pohl, C. Fischer and A. Köckritz,
Green Chem., 2014, 16, 1799–1806.
2 Z. Masyithah, L. V. Sitohang and M. P. Sihombing, ARPN
J. Eng. Appl. Sci., 2017, 12, 7031–7038.
Conclusions
Here we report an efficient catalyst-free green synthesis of
azelaic acid in a one-pot reaction by batch and continuous flow
methods. The method does not require any terminal oxidant or
metal/metal oxide catalyst. Excellent mixing, mass transfer and
large heat transfer area offered by a pinched tube reactor help
facilitate continuous ozonolysis that needs less than 5 s at 0 °C
with complete conversion of the substrate and almost stoichio-
metric quantity of ozone. This method has several advantages
over traditional methods in terms of time, cost, safety and sus-
tainability. Finally, the optimized protocol was demonstrated
with 20 g h− of azelaic acid in the laboratory. Complete re-
cycling and reuse of the acetone–water mixture as the solvent
have been established and the reused solvent mixture was used
at least 3 times with no detectable change in the yield, making
it a much greener process. The present approach is also shown
to be useful for the oxidative cleavage of other unsaturated fatty
acids and thus a single continuous platform at any scale can be
used for different fatty acids. A pilot plant that produces a few
kilograms of azelaic acid per day is under operation and a
detailed work based on the steady-state analysis of the pilot
plant will be reported separately.
3 D. L. Sparks, L. A. Estévez and R. Hernandez, Green Chem.,
2
009, 11, 986–993.
4 A. Köckritz and A. Martin, Eur. J. Lipid Sci. Technol., 2011,
13, 83–91.
1
5 H.-Y. Jang, K. Singha, H.-H. Kim, Y.-U. Kwon and J.-B. Park,
Green Chem., 2016, 18, 1089–1095.
6 P. Wu, J. Yan-Xia, H.-L. Yu and J.-He Xu, J. Biotechnol.: X,
1
2019, 2, 100008.
7 K. W. Anderson, J. D. Wenzel, R. G. Fayter and K. R. McVay,
Process for making polycarboxylic acids, US596228, 1999.
8 B. Hocevar, A. Prasnikar, M. Hus, M. Grilc and B. Likozar,
Angew. Chem., 2021, 60, 1244–1253.
1
2
9 B. Hocevar, M. Grilc and B. Likozar, Catalysts, 2019, 9, 286.
0 M. M. Cajnko, U. Novak, M. Grilc and B. Likozar,
Biotechnol. Biofuels, 2020, 13, 66.
2
1 F. O. Herman and G. R. Herbert, Process for the production
of a purified grade of azelaic acid by treatment with ozone
during purification, US3402108, 1968.
22 G. G. Charles, C. B. Alexander, F. O. Herman and P. R. Richard,
Mathod of making azelaic acid, US2813113, 1957.
Conflicts of interest
2
3 M. D. Lundin, A. M. Danby, G. R. Akien, T. P. Binder,
D. H. Busch and B. Subramaniam, ACS Sustainable Chem.
Eng., 2015, 3, 3307–3314.
A patent has been filed (INV-2018-66/83NF2019). The techno-
logy will be licensed to the industry.
2
2
4 H. Baumann, M. Bühler, H. Fochem, F. Hirsinger,
H. Zoebelein and J. Falbe, Angew. Chem., Int. Ed. Engl.,
1988, 27, 41–62.
Acknowledgements
5 M. Irfan, T. N. Glasnov and C. O. Kappe, Org. Lett., 2011,
13, 984–987.
The authors acknowledge the funding from the CSIR through
the INPROTICS (HCP0011) Project. R. S. A. and P. R. A. 26 M. O’Brien, I. R. Baxendale and S. V. Ley, Org. Lett., 2010,
acknowledge the CSIR for senior research fellowship.
12, 1596–1598.
This journal is © The Royal Society of Chemistry 2021
Green Chem., 2021, 23, 2391–2396 | 2395

 Atapalkar, Ranjit S.
Atapalkar, Ranjit S.
 Athawale, Paresh R.
Athawale, Paresh R.
 Srinivasa Reddy
Srinivasa Reddy
 Kulkarni, Amol A.
Kulkarni, Amol A.