A. Medici et al. / Steroids 67 (2002) 51–56
55
2.4. Reduction of 6␣-fluoro-3␣-hydroxy-7-oxo-5-cholan-
24-oic acid methyl ester 12 with X. maltophilia
4.35 (1H, dm, JH-F ϭ 43 Hz, 6-CH); 13C-NMR (CDCl3 ϩ
CD3OD) ␦ 175.66 (CO2H), 98.56 and 95.12 (C-6, JC6,F
ϭ
173.2 Hz), 71.48 (C-3), 69.81 and 69.22 (C-7, JC7,F ϭ 29.6
Hz); HRMS (ESI) calcd for C24H40FO4 [MH]ϩ 411.2905;
found 411.2906; [MH]ϩ major fragmentation observed
[MH –H2O]ϩ.m/z 393. Anal. Calcd for C24H39O4F: C%,
70.21; H%, 9.57; F%, 4.62. Found: C%, 70.64; H%, 9.41;
The reaction is carried out according to method A start-
ing from 0.1 g of 12 dissolved in DMF (1 ml) added to a
grown culture of X. maltophilia (10 ml). Aliquots were
withdrawn periodically and monitored by GLC and TLC.
After 24 h incubation TLC analysis confirmed no more
starting product. Centrifugation, extraction with ethyl ace-
tate, drying with anhydrous Na2SO4, and chromatography
(silica, ethyl acetate/cyclohexane/acetic acid 50:50:1) of the
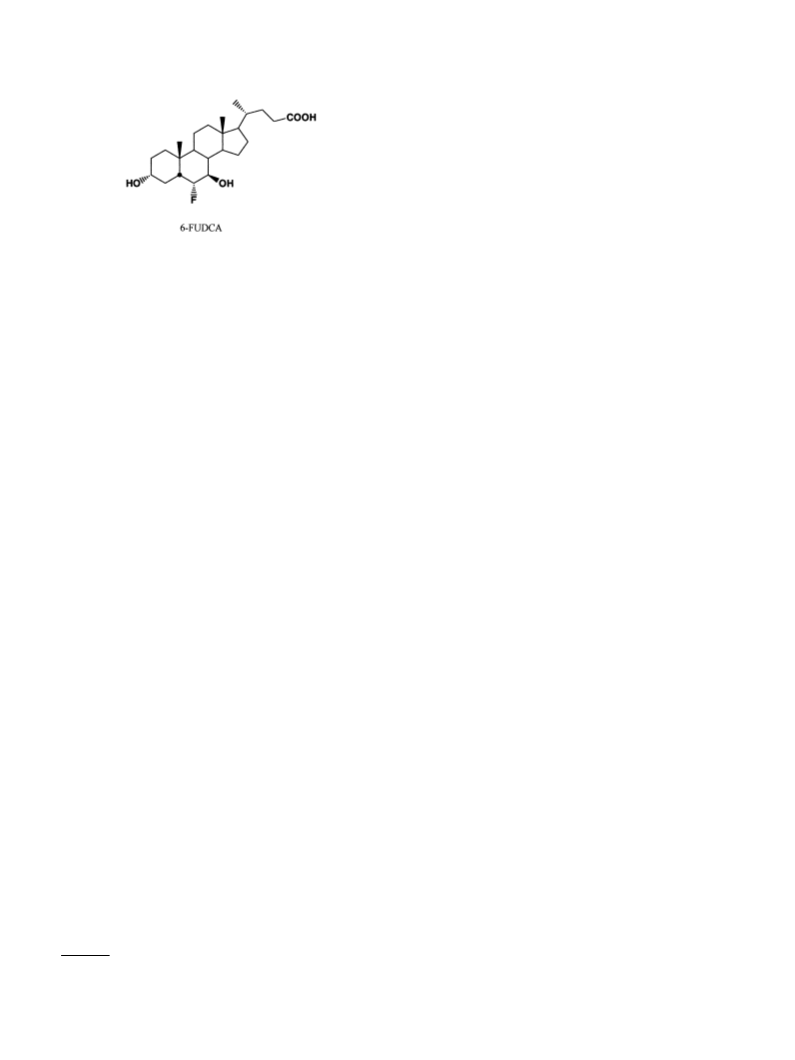
reaction mixture afford the pure 6␣-fluoro-3␣,7␣-dihy-
droxy-5-cholan-24-oic acid (14): 0.092 g (95%): 1H-NMR
(CDCl3 ϩ CD3OD) ␦ 0.55 (3H, s, 18-Me), 0.80 (3H, s,
19-Me), 0.85 (3H, d, 21-Me), 3.30 (1H, m, 3-CH), 3.85 (1H,
m, 7-CH), 4.53 (1H, ddd, JH-F ϭ 45 Hz, JH-H ϭ 3.5, 5.5 Hz,
6-CH); 13C-NMR (CDCl3ϩ CD3OD) ␦ 175.83 (CO2H),
94.20 and 90.66 (C-6, JC6,F ϭ 178.4 Hz), 71.64 (C-3), 70.27
and 69.94 (C-7, JC7,F ϭ 31.5 Hz); HRMS (ESI) calcd for
C24H40FO4 [MH]ϩ 411.2905; found 411.2904; [MH]ϩ ma-
jor fragmentation observed [MH –H2O]ϩm/z 393. Anal.
Calcd for C24H39O4F: C%, 70.21; H%, 9.57; F%, 4.62.
Found: C%, 71.02; H%, 9.39; F%, 4.60. The reaction was
repeated and crude reaction products were monitored by
GLC and TLC until 48 h incubation. At 24 h the quantitative
yield of 14 was confirmed but at 48 h the 7-oxo derivative
17 was quantitatively produced. Compound 17 was trans-
formed with diazomethane in the methyl ester 12 and the
1
F%, 4.66. Compound 16 showed the following: H-NMR
(CDCl3 ϩ CD3OD) ␦ 0.70 (3H, s, 18-Me), 0.95 (3H, d,
21-Me), 1.03 (3H, d, 19-Me), 3.60 (2H, m, 3-CH and
7-CH), 4.73 (1H, dm, JH-F ϭ 36 Hz, 6-CH); 13C-NMR
(CDCl3 ϩ CD3OD) ␦ 177.16 (CO2H), 93.63 and 90.16
(C-6, JC6,F ϭ 177.1 Hz), 78.19 and 77.45 (C-7, JC7,F ϭ 22.6
Hz), 70.54 (C-3); HRMS (ESI) calcd for C24H40FO4 [MH]ϩ
411.2905; found 411.2905. [MH]ϩ major fragmentation ob-
served [MH –H2O]ϩ.m/z 393. Anal. Calcd for C24H39O4F:
C%, 70.21; H%, 9.57; F%, 4.62. Found: C%, 70.83; H%,
9.29; F%, 4.58. The reaction was repeated and crude reac-
tion products were monitored by TLC until 72 h incubation.
At 24 h the 1:1 mixture of 15 and 16 was confirmed but at
72 h only 16 (92%) was recovered.
3. Results and discussion
The incubation of cholic acid (CA, 1) and of 12-oxo-
chenodeoxycholic acid (12-oxo-CDCA, 3) with a grown
and non-stirred culture of Xanthomonas maltophilia CBS
827.97 (method A) affords the corresponding 7-OH deriv-
atives ursocholic acid (UCA, 9) and 12-oxo-ursodeoxy-
cholic acid (12-oxo-UDCA, 11) in 80% yield, after 24 and
96 h respectively (Scheme 1, Table 1).
On the other hand, when the incubation of the BAs 1 and
3 is carried out under vigorous stirring (method B), the
7-oxo derivatives 5 and 7 are obtained in 85% yield, after 24
and 96 h respectively, together with small amount of the
7-OH products 9 and 11 in 10% yield.
1
data are the following: H-NMR (CDCl3) ␦ 0.60 (3H, s,
18-Me), 0.87 (3H, d, 21-Me), 1.18 (3H, s, 19-Me), 3.55 (1H,
m, 3-CH), 3.60 (3H, s, CO2Me), 5.18 (1H, dd, JH-F ϭ 49.7
Hz, JH-H ϭ 6.3 Hz, 6-CH); 13C-NMR (CDCl3 ϩ CD3OD) ␦
205.52 (CϭO), 174.35 (CO2Me), 93.66 and 89.82 (C-6,
JC6,F ϭ 193.5 Hz), 69.72 (C-3); HRMS (ESI) calcd for
C25H40FO4 [MH]ϩ 423.2905; found 423.2906; [MH]ϩ ma-
jor fragmentation observed [MH –H2O]ϩ m/z 405. Anal.
Calcd for C25H39O4F: C%, 71.06; H%, 9.30; F%, 4.49.
Found: C%, 70.95; H%, 9.17; F%, 4.53.
Worst results are obtained in the incubation of chenode-
oxycholic acid (CDCA, 2) with scarsity of oxygen (method
A). After 24 h only 27% of the 7-OH ursodeoxycholic acid
(UDCA, 10) is produced together with 23% of the corre-
sponding 7-keto derivative 6. Surprisingly, prolonged incu-
bation (48 h) of 2 increased the amount of the oxidation
product 6 (44%) and decreased the percentage of UDCA
(10%). In these conditions the 7-OH oxidation is probably
faster than the reduction. On the other hand the incubation
of CDCA 2 under vigorous stirring (method B) affords the
oxidation product 6 in 80% yield after 24 h. The 7-keto
derivative 8 is, however, the only product obtained by
incubation of hyocholic acid 4 with X. maltophilia both with
scarsity of oxygen (method A, 25% yield) and vigorous
stirring (method B, 80% yield). On the other hand the
starting materials were recovered unaltered when the incu-
bation of BAs 1–4 was carried out in anaerobic conditions.
On the basis of these results we can suggest the presence of
two hydroxysteroid dehydrogenases (HSDH) ␣- and -ste-
2.5. Reduction of 6-fluoro-3␣-hydroxy-7-oxo-5-cholan-
24-oic acid methyl ester 13 with X. maltophilia
The reaction is carried out according to method A start-
ing from 0.1 g of 13 dissolved in DMF (1 ml) added to a
grown culture of X. maltophilia (10 ml). Aliquots were
withdrawn periodically and monitored by TLC. After 24 h
incubation TLC analysis confirmed no more starting prod-
uct. Centrifugation, extraction with ethyl acetate, drying
with anhydrous Na2SO4, and chromatography (silica, ethyl
acetate/cyclohexane/acetic acid 50:50:1) of the reaction
mixture afforded the pure 6-fluoro-3␣,7␣-dihydroxy-5-
cholan-24-oic acid 15 (45 mg, 46%) and 6-fluoro-3␣,7-
dihydroxy-5-cholan-24-oic acid 16 (45 mg, 46%). Com-
pound 15 showed the following: 1H-NMR (CDCl3
ϩ
CD3OD) ␦ 0.60 (3H, s, 18-Me), 0.90 (3H, d, 21-Me), 0.95
(3H, d, 19-Me), 3.35 (1H, m, 3-CH), 3.75 (1H, m, 7-CH),

 Medici, Alessandro
Medici, Alessandro
 Pedrini, Paola
Pedrini, Paola
 Bianchini, Ercolina
Bianchini, Ercolina
 Fantin, Giancarlo
Fantin, Giancarlo
 Guerrini, Alessandra
Guerrini, Alessandra
 Natalini, Benedetto
Natalini, Benedetto
 Pellicciari, Roberto
Pellicciari, Roberto