Preparation of 2,6-di-(t-Butyl)-4-methylphenol
239
radical. Therefore in order to prevent this occurrence, 1% (by weight) of thiourea was
added as an anti-oxidant immediately after completion of the reaction and prior to work-
up. The mixture was then cooled with stirring and neutralized, the product was collected,
washed and recrystallized. As illustrated in the Table (Entry 3), when TsOH and thiourea
were applied, the content of the major impurity in the crude and purified BHT were lower
than that of Entries 1 and 2. Adoption of these measures led to BHT with a purity above
9
9.7% in an 81.1% overall yield, in particular, the content of major impurity was
decreased to 0.16% and the total amount of all the other impurities was less than 0.1%,
this will be of great importance when BHT is used in food and pharmaceutical industries.
To evaluate the possibility of the improved process to be scaled up, 1.2 kg of p-cresol
was fed in a 5L glass jacketed autoclave, the other operations being the same except that
ꢀ
ꢀ
the temperature at which isobutylene was introduced was lowered from 62 C to 56 C and
the reaction time was extended to 9 h. The purity of final product was a little higher and
the content of the major impurity was a little lower than that in Entry 3 of Table.
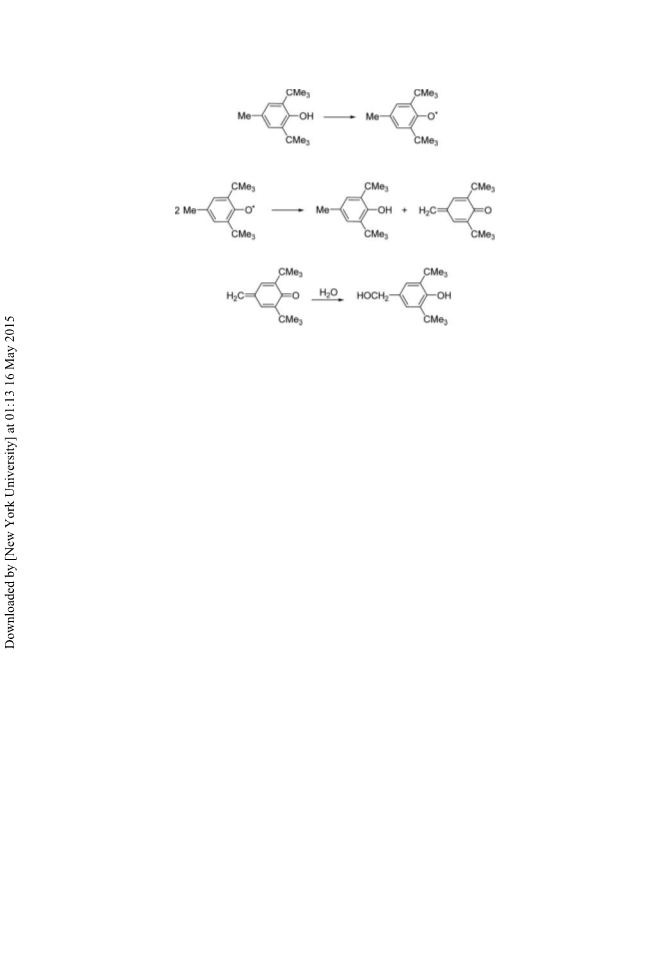
In conclusion, an improved preparation of BHT which minimizes the formation of
2
,6-di-(t-butyl)-4-(hydroxymethyl)phenol, the major impurity and increases the purity of
BHT has been designed. Our calculations – based on the manufacture of one ton of BHT
indicates that the higher yield of purer BHT obtained using p-toluenesufonic acid
–
requires a lesser amount of p-cresol thus compensating for the higher cost of the catalyst.
Thus our procedure may constitute a valuable alternative in the preparation of pure BHT
for the food and pharmaceutical industries. Moreover, when the improved process was
scaled up to 1.2 kg in a 5L glass jacketed autoclave, the results were as good as those per-
formed on a laboratory scale.
Experimental Section
Reagents and solvents were purchased from commercial suppliers and used without fur-
ther purification. Mps were measured in open capillaries and are uncorrected. The purity
1
of product was determined using a Tianmei GC7900 GC chromatograph. H NMR spec-
tra were recorded in CDCl on a Bruker Avance (Varian Unity Inova) 400 MHz spec-
3
trometer with TMS as the internal standard. MS spectra were performed on a Waters
Quattro Premier XE triple quadrupole mass spectrometer. Because the butylation of p-
cresol with isobutylene is a typical liquid-gas reaction process, a custom-made glass reac-
tor with a flat bottom, a cylindrical body, and three necks to accommodate a condenser, a
gas inlet tube, and a thermometer respectively, was utilized in the laboratory-scale experi-
ments. The isobutylene was bubbled through a sintered glass plate just above the bottom
thus resulting in a good dispersion of isobutylene and a comparatively long contact time
for the isobutylene and p-cresol. The gas chromatographic conditions utilized to monitor
and analyze the reaction were as follows: GC column: TM-1701 capillary column, the
ꢀ
ꢀ
inlet temperature: 280 C, the column temperature: 200 C, FID detector temperature:
ꢀ
2
80 C, nitrogen flow rate: 45mL/min, hydrogen flow rate: 30mL/min, and injection vol-
ume: 1mL. The concentration of sample was about 30mg/mL with dichloromethane as
the solvent. The retention time of BHT in the gas chromatogram was about 5.7 min and
that of the major impurity was about 5.4 min.
Preparation of BHT using conc. Sulfuric Acid as the Catalyst
The three-necked glass reactor fitted with a condenser, a gas inlet tube, and a thermometer
was immersed in a water-bath and melted p-cresol (80.0g, 0.74 mol) was added followed

 Xie
Xie
 Liu
Liu
 Zhang
Zhang
 Li
Li
 Li
Li
 Huang
Huang