B. Drießen-H o¨ lscher, J. Heinen / Journal of Organometallic Chemistry 570 (1998) 141–146
145
Hz, 94.4 (s, Cp–Me ), 61.1 (s, CH OH, J(PC=14 Hz),
solution of 5 g Tris(3-hydroxypropyl)phosphane 15 in 5
ml of methanol was deposited and eluted with a mix-
ture of ethyl acetate/methanol 4:1. The phosphane ox-
ide was obtained first and was detected with UV-light.
The desired phosphane 15 could be detected with
molybdatophosphoric acid solution in ethanol. The
purification by column chromatography took about 12
h and gave the phosphane in a pure form.
5
2
2
5.5 (s, CH CH CH OH), 21.5 (d, CH (CH ) OH,
2 2 2 2 2 2
31
J(PC)=27.2 Hz), 7.76 (s, Cp–Me5). P-NMR (121
MHz, d -acetone) l=30.43 ppm, (free ligand: l= −30
6
−
1
ppm). IR (KBr): w(CO) 1921 cm . Elemental analysis:
C H PO ClRu (507.52 g mol ); Cfound: 47.29% (Ccalc.
−
1
2
0
36
4
47.28%); Hfound 7.11% (Hcalc. 7.09%).
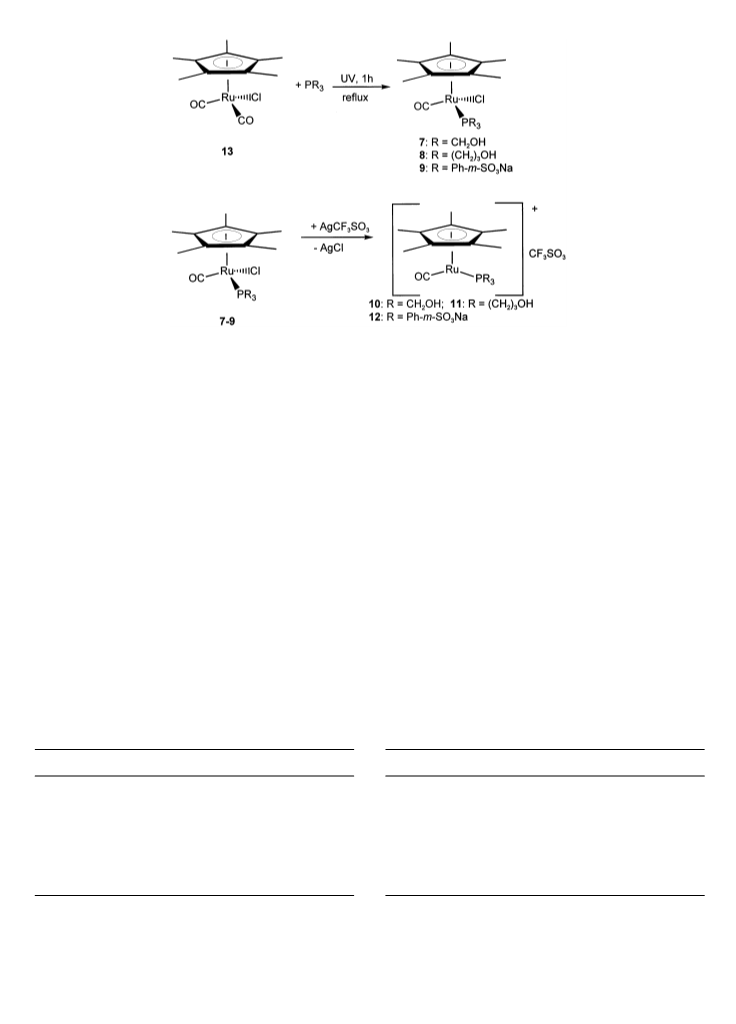
Complex 9 was purified as follows: After the irradia-
tion, the solvents were removed in vacuo. The residue
was suspended twice in 5 ml portions of ether. After
decantation, the solid was dried in vacuo and then
dissolved in 2 ml water. This solution was used for
4
.5. Hydrogenation reaction
The cold catalyst solution was transferred into a 100
®
chromatrography on Sephadex (column dimensions:
ml stainless steel autoclave via syringe with a PTFE
cannula. The n-heptane phase containing the sorbic
acid was added and the autoclave was pressurized with
hydrogen. The autoclave was placed in an oil bath of
diameter 2.5 cm, 45 cm length). The first fraction was
unreacted ligand and the corresponding oxide. Subse-
quently complex 9 was obtained as a deep yellow
fraction. After removal of water the residue was recrys-
tallized from ethanol (yield: 47%).
8
0°C. After the reaction the autoclave was cooled in an
ice bath and was slowly vented. The two phases were
separated. The products were in the n-heptane phase
which was analyzed by GC [CP-Wax-58-(FFAP)-CB
1
H-NMR (300 MHz, D O): l=1.33 (s, 15 H, Cp*),
.61 (s, ca. 6 H), 7.66 (m, 12 H, tppts). C-NMR (75
2
13
4
(
50 m)].
MHz, D O): l=204.2 (d, CO, J(PC, 20 Hz) 141.1 (d)
2
1
33.97 (s) 128.84 (s) 127.5 (d) u.127.73 (d) all tppts, 95.6
31
(
s, Cp–Me ), 6.79 (s, Cp–Me ). P-NMR (121 MHz,
5 5
−
1
CD OD): l=50.69 (s). IR(KBr): w(CO): 1928 cm
.
Acknowledgements
3
Elemental analysis: C H RuClPO S Na (921.96 g
2
9
33
13
3
3
−
1
mol ); Cfound 37.18% (Ccalc. 37.78%); Hfound 3.48%
Hcalc. 3.57%).
This work was supported by the BMBF ‘Homoge-
neous Catalysis in Multiphase Systems’ and by the
(
‘Catalysis Network NRW’. We are grateful to Degussa
4
.3. General procedure for preparation of the cationic
AG which supplied us generously with transition metal
compounds. We thank Professor Dr W. Keim for his
continous interest and support of this work and Dr T.
Prinz of our group for his ideas on ligand purification.
B.D.-H. is grateful for a grant from the state of Nor-
drhein-Westfalen (HSP II).
complexes
Two millimoles of the appropriate neutral complex
(
7–9) were reacted with 2 mmol AgCF SO in 40 ml
3
3
CH Cl in the dark. The suspension was filtered over
2
2
celite. The solvent was removed from the filtrate in
vacuo. The remaining residue was characterized spec-
troscopically and used for the hydrogenation of sorbic
acid.
References
1
Characterization of 11: H-NMR (300 MHz,
[
1] (a) H. Bahrmann, B. Cornils, W. Konkol, W. Lipps (Ruhrchemie
AG), DE-B 3412335, (1984); Chem. Abstr. 104 (1985) 185989m.
CD Cl ): l=3.57 (s, 6H, CH OH), 2.0–1.4 (m, br., 12
2
2
2
3
1
(
b) B. Cornils, W. Konkol, H. Bach, W. Gick, E. Wiebus, H.
H, PCH CH and Cp–Me ). P-NMR (121 MHz,
2
2
5
1
9
Bahrmann, H. Hahn (Ruhrchmie AG), DE-B 3546123, (1985);
Chem. Abstr. 107 (1987) 153922e.
CD Cl ): l=30.05 (s). F-NMR (282 MHz, CD Cl ):
2
2
2
2
−
1
l= −79.24. IR(KBr): 1958 cm
.4. General procedure for purification of ligand 15
A chromatographic purification of 15 was found to
.
[
2] W. Keim, T.M. Shryne, R.S. Bauer, H. Chung, P.W. Glockner,
H. van Zwet, (Shell Int. Res.), DE-P 2054009 (1969).
4
[3] (a) A. Behr, W. Keim, Erd o¨ l, Erdgas, Kohle, 103 (1987) 126. (b)
M. Barton, J.D. Atwood, J. Coord. Chem. 24 (1991) 43. (c) P.
Kalck, F. Monteil, Adv. Organomet. Chem. 34 (1992) 219. (d)
W.A. Herrmann, C.W. Kohlpaintner, Angew. Chem. 105 (1993)
be the best. The stationary phase was silica 60 (Merck)
which was evacuated several times and set under argon.
The glass column which can be handled under argon
1
524. (e) I.T. Horvath, F. Joo (Eds.), Nato ASI Ser. 3 (1995) 5. (f)
D.M. Roundhill, Adv. Organometal. Chem. 38 (1995) 155. (g) B.
Cornils, E. Wiebus, CHEMTECH, (1995) 33. (h) G. Papado-
gianakis, R.A. Sheldon, New J. Chem. 20 (1996) 175. (i) B.
Cornils, W.A. Herrmann, Applied Homogeneous Catalysis with
Organometallic Compounds, VCH, New York, 1996. (j) F. Joo,
A. Katho, J. Mol. Catal. 116 (1997) 3. (k) B. Drießen-H o¨ lscher,
Multiphase Homogeneous Catalysis, Advances in Catalysis, vol.
42, Academic Press, New York, 1998, p. 473.
(
¥3.5 cm, length 45 cm) was filled with 500 ml silica
which was suspended in 160 ml methanol and 640 ml
ethyl acetate. A 250 ml aliquot of the solvent mixture
was run through the column to homogenate the
column. The column was left to settle overnight. A

 Driessen-Hoelscher, Birgit
Driessen-Hoelscher, Birgit
 Heinen, Joerg
Heinen, Joerg