18
Chem. Pharm. Bull. 63, 18–24 (2015)
Vol. 63, No. 1
Regular Article
Stability Order of Caffeine Co-crystals Determined by Co-crystal
Former Exchange Reaction and Its Application
for the Validation of in Silico Models
,a,b
Makoto Mukaida,* Kiyohiko Sugano,a,b and Katsuhide Teradab
b
a Asahi Kasei Pharma Corporation; 632–1 Mifuku Izunokuni, Shizuoka 410–2321, Japan: and Department of
Pharmaceutics, Faculty of Pharmaceutical Sciences, Toho University; 2–2–1 Miyama, Funabashi, Chiba 274–8510,
Japan.
Received June 30, 2014; accepted October 29, 2014
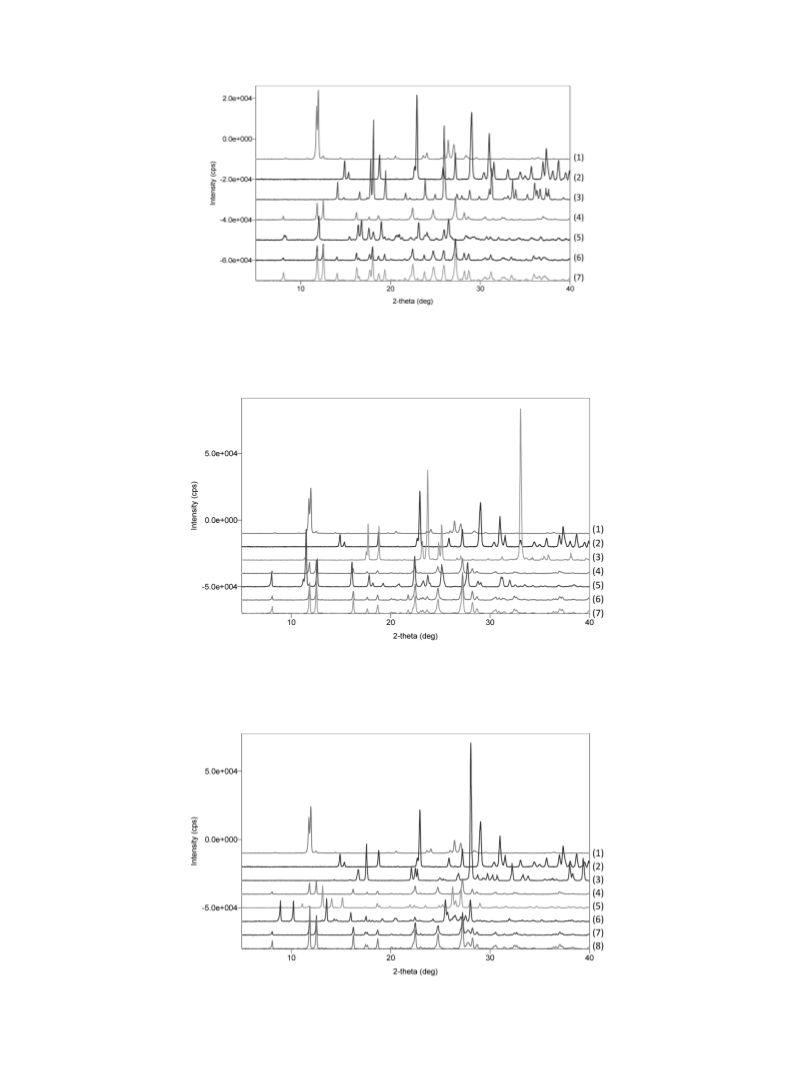
The purpose of the present study was to determine the thermodynamic stability orders of co-crystals
by co-crystal former (CCF) exchange reactions. Caffeine (CA) was employed as a model drug. The CCF ex-
change reaction was performed by liquid-assisted grinding using ethanol. When oxalic acid (OX) was added
to CA–citric acid co-crystal (CA–CI), CA–CI converted to CA–OX, suggesting that CA–OX is more stable
than CA–CI. The stability orders of other co-crystals were determined in the same manner. The stability
order of CA co-crystals was determined as CA–OX≈CA–p-hydroxybenzoic acid (HY)>CA–CI>CA–malonic
acid>CA–maleic acid. The stability order correlated with the difference in hydrogen bond energy estimated
in silico, except for CA–HY. The π–π stacking in CA–HY was suggested as a reason for this discrepancy. The
CCF exchange reaction was demonstrated as a useful method to determine the stability order of co-crystals,
which can be used for the validation of in silico parameters to predict co-crystal formation.
Key words co-crystal; co-crystal former; exchange reaction; caffeine; hydrogen bond energy
Co-crystals have recently received much attention as a
means of improving physicochemical properties of active
Experimental
Materials p-Hydroxybenzoic acid was purchased from
pharmaceutical ingredients.1–10) The number of potential Tokyo Chemical Industry Co., Ltd. (Tokyo, Japan). Other
co-crystal formers (CCFs) can reach up to several hundred; agents were purchased from Wako Pure Chemical Industries,
therefore, an efficient strategy for CCF screening in drug Ltd. (Osaka, Japan).
discovery and development is required. High throughput
Preparation of CA Co-crystals CA–p-hydroxybenzoic
screening (HTS) technologies have been employed for CCF acid co-crystal (CA–HY), CA–oxalic acid co-crystal (CA–
screening.10–15) However, CCF selection by HTS remains a OX), CA–citric acid co-crystal (CA–CI), CA–malonic acid
time- and resource-consuming task. Therefore, an effective co-crystal (CA–MO), and CA–maleic acid co-crystal (CA–
pre-screening method for CCF selection would be valuable for MA) were prepared by liquid-assisted grinding. CA (25mg,
drug discovery and development. In the case of salt selection, 0.13mmol) was mixed with each CCF and chloroform (25µL)
the difference in pKa values between a drug and a counter- in a 1-mL glass vial with 2.4-mm tungsten balls. The mixture
ion (ΔpKa) has been used as a criterion for the selection of was shaken in a BMS-TMS 200 shaker at 1800rpm at ambient
potential counter-ion candidates.16) Similarly, some in silico temperature. Chloroform was then evaporated under nitrogen
physicochemical parameters for CCF selection were proposed gas flow for more than 1h. The amount of CCF, the molar
by several research groups.17–20) For example, the difference ratio of the co-crystal components, and the shaking duration
in hydrogen bond energy between a co-crystal and each of are shown in Table 1.
the sole components (ΔE) was proposed by Musumeci et al.,
CCF Exchange Reaction Equimolar amounts of CCF
based on the hypothesis that the formation of a co-crystal (1–7mg) were added to each co-crystal in a 1-mL glass vial
becomes more probable as ΔE becomes larger.21–23) How- with 2.4-mm tungsten balls. Ethanol (5µL) was then added
ever, ΔE has not been rigorously validated due to the lack to the vial and the vial was shaken for 2–48h (in most cases
of information about the stability order of co-crystals. The 22h). Ethanol was then evaporated under nitrogen gas flow for
stability order of co-crystals can be determined by CCF ex- more than 1h.
change reactions. In the literature, there were a few studies on
the CCF exchange reactions (a sulfonamide derivative24) and
Table 1. Preparation Conditions for Each Co-crystal
carbamazepine25)). However, in these studies, only a few CCFs
Molar ratio
of the components
(CA:CCF)
were employed so that the data is insufficient for validating in
silico models.
Amount of CCF
(mg)
Reaction time
(h)
Co-crystal
The purpose of the present study was to determine the
stability orders of co-crystals by CCF exchange reaction.
Caffeine (CA) was employed as a model drug. The stability
order of five CA co-crystals was determined using the CCF
exchange reaction. The stability order was then compared with
the ΔE estimated in silico.
CA–HY
CA–OX
CA–CI
35
6
1:2
2:1
1:1
2:1
1:1
44
22
22
6
25
7
CA–MO
CA–MA
15
26
*To whom correspondence should be addressed. e-mail: mukaida.mb@om.asahi-kasei.co.jp
© 2015 The Pharmaceutical Society of Japan

 Mukaida, Makoto
Mukaida, Makoto
 Sugano, Kiyohiko
Sugano, Kiyohiko
 Terada, Katsuhide
Terada, Katsuhide