J . Org. Chem. 1999, 64, 4665-4668
4665
A Sim p le Solu tion to th e Age Old P r oblem of Regioselective
F u n ction a liza tion of Gu a n in e: F ir st P r a ctica l Syn th esis of Acyclic
9
7
N - a n d /or N -Gu a n in e Nu cleosid es Sta r tin g fr om
2
9
N ,N -Dia cetylgu a n in e
Dharmendra Singh, Mukesh J . Wani, and Ashok Kumar*
Lupin Laboratories Ltd. 202, New Industrial Area No. 2,
Mandideep-462 046, District-Raisen (M.P.), India
Received November 23, 1998
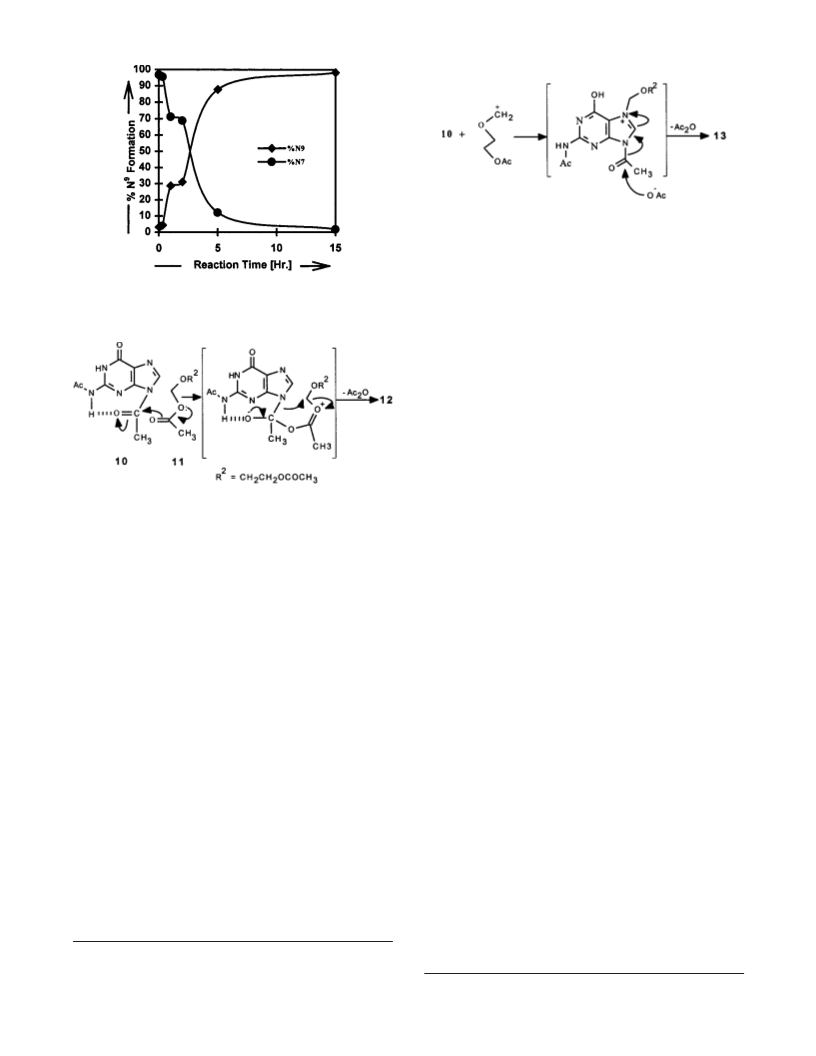
Regioselective alkylation of guanine, a long-lasting challenge, has been overcome by understanding
the role of acids as catalyst in the coupling reaction of DAG (10) with OBDDA (11). The acid-
7
9
catalyzed and noncatalyzed reactions of 10 with OBDDA which mainly give N and N isomers,
respectively, appear to follow different mechanisms. The practical utility of the noncatalyzed
9
reaction, which gives almost quantitative yields of N derivatives, is demonstrated by synthesizing
acylovir/gancyclovir in high yields.
Guanine is one of the five important bases of life (DNA
and RNA). However, because of its polyfunctionality and
amphoteric nature, which is accompanied by its ex-
tremely poor solubility in most solvents, this molecule is
difficult to functionalize. Direct alkylation or glycosyla-
2
2
9
tion of N - or N ,N -protected guanines always produce
9
7
1-7
N /N isomeric mixtures that are difficult to separate.
Since regioselective N alkylation of guanine precursors
is the primary route to clinically effective antiviral drugs
such as 9-[2-(hydroxyethoxy)methyl]guanine (acyclovir,
) and 9-[(1,3-dihydroxy-2-propoxy)methyl]guanine (gan-
9
2
8
1
cyclovir, 2), which are active against viral diseases caused
by herpes simplex virus-1 (HSV-1), herpes simplex
virus-2 (HSV-2), varicella zoster virus (VZV), and the
F igu r e 1.
dates. For example, 2-amino-7-[(1,3-dihydroxy-2-propoxy)-
methyl]purine (3), the 6-deoxy derivative of the N
regioisomer of gancyclovir, has recently been found to
display excellent antiherpes action.
Several multistep conversions which include alkylation
of 2-amino-6-chloropurine (4), 2,6-dichloropurine (5),
and 6-enolate derivatives of guanine (6) followed by
hydrolytic cleavage of intermediates to give the corre-
sponding 6-oxo derivatives, have been employed to give
9
human cytomagalo virus, studies toward achieving high
7
regioselectivity in this transformation have been the
subject of considerable interest and intensive investiga-
10
7
tion in recent years. Selective N alkylation of guanine
is also important for understanding the factors respon-
sible for achieving high regioselectivity as well as for
11
2d
12
7
synthesizing N analogues for evaluating as drug candi-
(
1) (a) Kjellberg, J .; J ohansson, N. G. Tetrahedron 1986, 42, 6541.
b) Kjellberg, J .; J ohansson, N. G. Nucleosides Nucleotides 1989, 8,
25 and references therein.
2) (a) Ellion, G. B.; Furman, P. A.; Fyfe, J . A.; de Miranda, P.;
Beauchamp, L. M. Schaeffer, H. J . Proc. Natl. Acad. Sci. U.S.A. 1977,
4, 5716. (b) Schaeffer, H. J .; Beauchamp, L. M.; de Miranda, P.; Elion,
9
7
(
enhancement in N /N isomer ratios (Figure 1). Even
better results are reported by using persilylated 6-eno-
2
(
2
13
lates of N -acetylguanine (7) and 2-amino-6-chloro-
purine (4), and its derivatives.1
0,14
Vorbruggen’s group
7
G. B.; Bauer, D. J .; Collins, P. Nature (London) 1978, 272, 583. (c)
Beauchamp, L. M.; Dolmatch, B. L.; Schaeffer, H. J .; Collins, P.; Bauer,
D. J .; Kelter, P. M.; Fyfe, J . A. J . Med. Chem. 1985, 28, 982. (d)
Schaeffer, H. J . Ger. Patent 2539963, 1974.
demonstrated reasonably high regioselectivity in the
glycosylation15 reaction of per(trimethylsilyl)-N -acetylgua-
nine (8) with 1-O-acetyl-2,3,5-tri-O-benzoyl-â-D-ribofura-
nose (9) in the presence of TMSOTf as catalyst at
elevated temperature. The alkylated product after depro-
tection and crystallization afforded guanosine in 66%
2
(
3) Hua, M.; Korkowski, P. M.; Vince, R. J . Med. Chem. 1987, 30,
1
98.
(
(
4) Lin, T.-S.; Liu, M.-C. Tetrahedron Lett. 1984, 25, 611.
5) Robins, M. J .; Zou, R.; Hanske, F.; Madej, D.; Tyrrell, D. L. J .
Nucleosides Nucleotides 1989, 8, 725.
6) Boryski, J .; Manikowski, A. Nucleosides Nucleotides 1995, 14,
87.
7) Matsumoto, H.; Kaneko, C.; Yamada, K.; Takeuchi, T.; Mori, T.;
Mizuno, Y. Chem. Pharm. Bull. 1988, 36, 1153.
8) (a) Ogilvie, K. K.; Cheriyan, U. O.; Radatus, B. K.; Smith, K. O.;
(
2
(10) J ahne, G.; Kroha, H.; Muller, A.; Helsberg, M.; Winkler, I.;
Gross, G.; Scholl, T. Angew. Chem., Int. Ed. Engl. 1994, 33, 562.
(11) Harnden, M. R.; J arvest, R. L.; Bacon, T. H.; Boyd, M. R. J .
Med. Chem. 1987, 30, 1636 and references therein.
(12) Geen, G. R.; Grinter, T.; Kincey, P. M.; J arvest, R. L. Tetrahe-
dron 1990, 46, 6903.
(
(
Galloway, K. S.; Kennell, W. L. Can. J . Chem. 1982, 60, 3005. (b)
Ashton, W. T.; Karkas, J . D.; Field, A. K.; Tolman, R. L. Biochem.
Biophys. Res. Commun. 1982, 108, 1716. (c) Martin, J . C.; Dvorak, C.
A.; Smee, D. F.; Mathews, T. R.; Verheyden, J . P. H. J . Med. Chem.
(13) Zou, R.; Robins, M. J . Can. J . Chem. 1987, 65, 1436 and
references therein.
1
983, 26, 759.
9) Hirsch, M. S.; Kaplan J . C.In Virology, Vol. 1; Fields, B. N.,
(14) MacCoss, M.; Chen, A.; Tolman, R. L. Tetrahedron Lett. 1985,
26, 4287.
(
Knipe, D. M., Chanock, R. M., Hirsch, M. S., Melnick, J . L., Monath,
T. P., Roizman, B., Eds.; Raven: New York, 1990; p 441.
(15) (a) Vorbruggen, H.; Krolikiewicz, K.; Bennua, B. Chem. Ber.
1981, 114, 1234. (b) Vorbruggen, H. Acta Biochim. Pol. 1996, 43, 25.
1
0.1021/jo982304y CCC: $18.00 © 1999 American Chemical Society
Published on Web 06/02/1999

 Singh, Dharmendra
Singh, Dharmendra
 Wani, Mukesh J.
Wani, Mukesh J.
 Kumar, Ashok
Kumar, Ashok