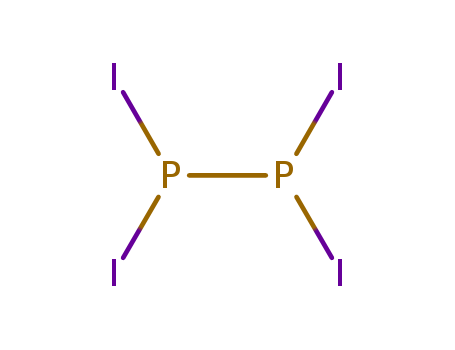
- Chemical Name:Hypodiphosphorous tetraiodide
- CAS No.:13455-00-0
- Molecular Formula:I4P2
- Molecular Weight:569.566
- Hs Code.:
- European Community (EC) Number:236-646-7
- DSSTox Substance ID:DTXSID6065474
- Nikkaji Number:J166.100K
- Wikipedia:Diphosphorus tetraiodide
- Wikidata:Q412973
- Mol file:13455-00-0.mol
Synonyms:Diphosphorus tetraiodide;13455-00-0;Hypodiphosphorous tetraiodide;diiodophosphanyl(diiodo)phosphane;Diphosphorous tetraiodide;Phosphorus iodide;Diphosphorus tetraiodide, 95%;EINECS 236-646-7;tetraiododiphosphane;periododiphosphine;Tetraiododiphosphine;Diphosphine, tetraiodo-;I4P2;P2I4;DTXSID6065474;I4-P2;MFCD00011526;AKOS015916035;Q412973;J-006558


 C
C




