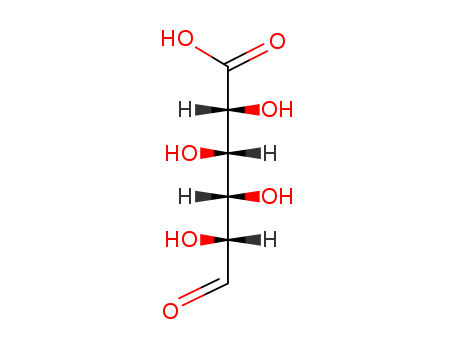
- Chemical Name:D-Guluronic Acid
- CAS No.:15769-56-9
- Molecular Formula:C6H10O7
- Molecular Weight:194.141
- Hs Code.:
- European Community (EC) Number:239-860-9
- Metabolomics Workbench ID:155982
- Wikipedia:Guluronic_acid
- Mol file:15769-56-9.mol
Synonyms:alpha-L-guluronic acid;G2013;G2013 compound;guluronic acid;guluronic acid, (alpha-L)-isomer;guluronic acid, (L)-isomer;guluronic acid, sodium salt, (L)-isomer