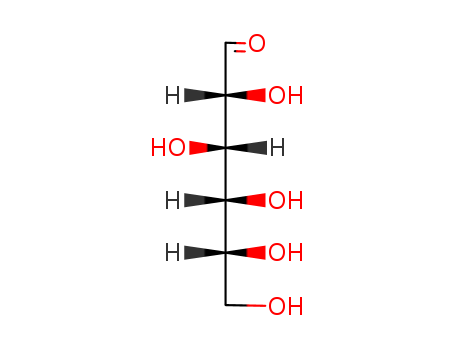
- Chemical Name:(2R,3S,4R,5R)-2,3,4,5,6-pentahydroxyhexanal
- CAS No.:50-99-7
- Deprecated CAS:162222-91-5,165659-51-8,50933-92-1,8012-24-6,80206-31-1,8030-23-7,111688-73-4,111688-73-4,165659-51-8,2280-44-6,8012-24-6,80206-31-1,8030-23-7
- Molecular Formula:C6H12O6
- Molecular Weight:180.158
- Hs Code.:17023051
- UNII:5SL0G7R0OK
- DSSTox Substance ID:DTXSID7022910,DTXSID4048729
- Nikkaji Number:J4.109B
- Wikidata:Q21036645
- Metabolomics Workbench ID:52322
- ChEMBL ID:CHEMBL448805
- Mol file:50-99-7.mol
Synonyms:Anhydrous Dextrose;D Glucose;D-Glucose;Dextrose;Dextrose, Anhydrous;Glucose;Glucose Monohydrate;Glucose, (alpha-D)-Isomer;Glucose, (beta-D)-Isomer;Glucose, (DL)-Isomer;Glucose, (L)-Isomer;L Glucose;L-Glucose;Monohydrate, Glucose


 Xn,
Xn,  Xi
Xi




