10.1016/S0040-4020(01)00917-6
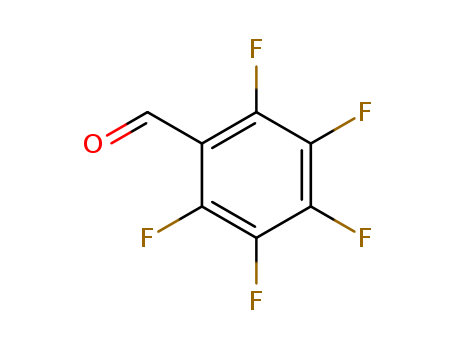
The research focuses on the reactions between 5-amino-1,2-azoles and aromatic or heterocyclic o-chloroaldehydes, which yield pyrazolo[3,4-b;2,3-b]quinolines, bispyrazolo[3,4-b;4',3'-e]pyridines, and their heterocyclic analogs as [1+1] or [2+1] cyclocondensation products. The study aims to explore the fluorescent properties of these synthesized compounds, which are potentially useful in photophysical processes, devices, and as electroluminescent compounds and polymers. The researchers found that the reactions can lead to different types of products depending on the reactivity of the starting materials, with yields ranging from 52-87%. The chemicals used in the process include various 5-amino-1,2-azoles, pentafluorobenzaldehyde, and other aromatic and heterocyclic aldehydes, which upon reaction, resulted in the formation of fluorescent condensed products with large conjugated π-systems. The conclusions drawn from the research highlight the potential applications of these compounds in areas such as luminescent dyes, scintillators, and fluorescent probes, due to their strong fluorescent properties and the ability to maintain these properties across different solvents and conditions.


 Xn,
Xn, F,
F, Xi
Xi




