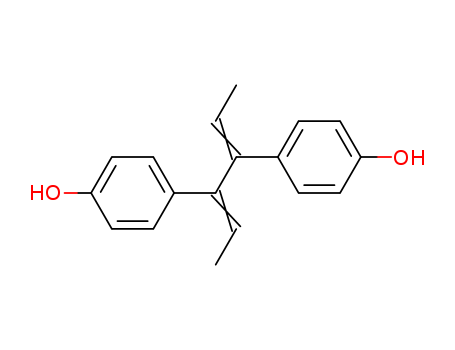
- Chemical Name:Phenol, 4,4'-(1,2-diethylidene-1,2-ethanediyl)bis-
- CAS No.:84-17-3
- Molecular Formula:C18H18O2
- Molecular Weight:266.34
- Hs Code.:
- European Community (EC) Number:201-519-7
- NSC Number:59809
- Nikkaji Number:J2.197.959G
- Pharos Ligand ID:G6CJ8NCGNM47
- ChEMBL ID:CHEMBL1450936
Synonyms:84-17-3;4-[(2Z,4E)-4-(4-hydroxyphenyl)hexa-2,4-dien-3-yl]phenol;Isodienestrol;Phenol, 4,4'-(1,2-diethylidene-1,2-ethanediyl)bis-;Dienestrol USP;CAS-84-17-3;NCGC00016335-01;(Z,E)-Dienestrol;BIDD:ER0215;CHEMBL1450936;2, 3,4-bis(4-hydroxyphenyl)-;NSC59809;Phenol,4'-(diethylideneethylene)di-;NCGC00018153-01;NCGC00018153-02;2,3,4-TRIMETHOXYBENZALDEHYDEOXIME;Phenol,4'-(1,2-diethylidene-1,2-ethanediyl)bis-;76156-28-0


 Xn
Xn



