Chemical Property of Nepafenac
Chemical Property:
- Vapor Pressure:1.11E-12mmHg at 25°C
- Melting Point:177-181oC
- Refractive Index:1.641
- Boiling Point:562.5 °C at 760 mmHg
- Flash Point:294 °C
- PSA:86.18000
- Density:1.249 g/cm3
- LogP:2.80910
- Storage Temp.:Refrigerator
- Solubility.:DMSO: ≥5mg/mL
- Water Solubility.:0.014 mg/mL in water
- XLogP3:1.9
- Hydrogen Bond Donor Count:2
- Hydrogen Bond Acceptor Count:3
- Rotatable Bond Count:4
- Exact Mass:254.105527694
- Heavy Atom Count:19
- Complexity:337
- Purity/Quality:
-
99.5% *data from raw suppliers
Nepafenac *data from reagent suppliers
Safty Information:
- Pictogram(s):
N
- Hazard Codes:N
- Statements:
50/53
- Safety Statements:
60-61
- MSDS Files:
-
SDS file from LookChem
Useful:
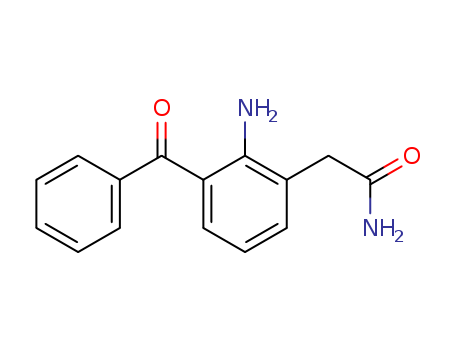
- Canonical SMILES:C1=CC=C(C=C1)C(=O)C2=CC=CC(=C2N)CC(=O)N
- Recent ClinicalTrials:Macular Edema Nepafenac vs. Difluprednate Uveitis Trial
- Recent EU Clinical Trials:Non-steroidal anti-inflammatory medication in cataract surgery
- Recent NIPH Clinical Trials:Factors related to dry eye after ocular surgery and its treatment.
-
Description
Nepafenac, launched by Alcon Laboratories, is a topical ophthalmic medication indicated for the treatment of ocular pain and inflammation associated with cataract surgery. Nepafenac is a prodrug of amfenac, which is an NSAID and a potent non-selective inhibitor of COX-1 (IC50=0.25 μM)) and COX-2 (IC50=0.15μM). Nepefenac itself exhibits only weak activity against COX-1 (IC50=64.3μM). Amfenac (Fenazox) has been marketed in Japan since 1986 for the treatment of rheumatoid arthritis, post-surgical pain, and inflammation. With most NSAIDs that are currently being used as topical ophthalmic agents, the maximum drug concentration is achieved on the ocular surface, with progressively lower concentrations in the cornea, aqueous humor, vitreous, and retina. Nepafenac has been found to have a penetration coefficient that is 4-28 times greater than that achieved with conventional NSAIDs such as diclofenac, bromofenac, and ketorolac. In addition, the bioconversion of nepefenac to amfenac is primarily mediated by ocular tissue hydrolases, specifically in the iris, ciliary body, retina, and choroid. The enhanced permeability of nepefenac combined with rapid bioactivation in the ocular tissue translates into superior anti-inflammatory efficacy at the target sites.
-
Uses
Nepafenac is a non-steroidal anti-inflammatory drug (NSAID), usually sold as a prescription eye drop. It reduces pain and inflammation in the eyes. Nepafenac ophthalmic is used to reduce pain and swelling after cataract surgery.
-
Clinical Use
Nepafenac is a novel ophthalmic non-steroidal anti-inflammatory drug (NSAID), for the treatment of eye pain and inflammation caused by cataract surgery, compared with traditional NSAIDs, chemical structure of Nepafenac is conducive to make it rapidly penetrate the cornea and distribute to its target site, which is helpful to reduce the accumulation of the drug in the corneal surface and to reduce the incidence of complications of the eye surface, it has many advantages such as infiltration, targeting strong, little toxic side effects and so on. August 19, 2005 ,the US Food and Drug Administration (FDA) approved nepafenac ophthalmic suspension for the treatment of cataract surgery-related pain and inflammation, it is the first ophthalmic NSAID prodrug formulation approved for marketing. Nepafenac after ocular administration, can rapidly pass through the cornea , and under the action of eye tissue hydrolytic enzymes,it can become into ammonia diclofenac (a kind of NSAID); and ammonia diclofenac by inhibiting prostaglandin H synthase ( cyclooxygenase), can block prostaglandin synthesis to play its role as an anti-inflammatory analgesic. As is known, prostaglandin is one of the media causing ocular inflammation it can lead to blood-aqueous barrier crash, vasodilatation, increased vascular permeability and leukocyte chemotaxis, etc. In addition, prostaglandins can also control contraction of the iris sphincter through non-cholinergic mechanism which can trigger the miosis reaction during eye surgery and after surgery. After ocular administration of NSAIDs, it can inhibit prostaglandins synthesis in the iris, ciliary body and conjunctiva, so people can prevent eye inflammation, and reduce the associated pain.The above information is edited by the lookchem of Tian Ye.