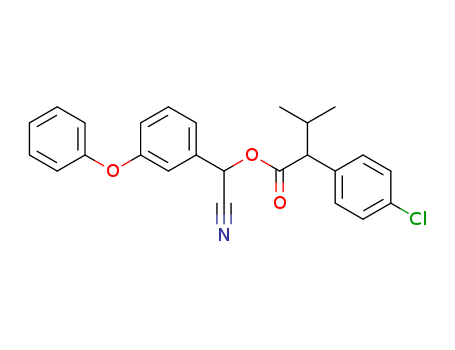
- Chemical Name:Fenvalerate
- CAS No.:51630-58-1
- Deprecated CAS:131641-62-8
- Molecular Formula:C25H22ClNO3
- Molecular Weight:419.908
- Hs Code.:2926909036
- European Community (EC) Number:257-326-3
- ICSC Number:0273
- UN Number:2811,3352,2765
- UNII:Z6MXZ39302
- DSSTox Substance ID:DTXSID101017940
- Nikkaji Number:J3.515G
- Wikipedia:Fenvalerate
- Wikidata:Q412010
- NCI Thesaurus Code:C65672
- ChEMBL ID:CHEMBL492491
- Mol file:51630-58-1.mol
Synonyms:alpha-cyano-3-phenoxybenzyl 2-(4-chlorophenyl)-3- methylbutyrate;alpha-cyano-3-phenoxybenzyl alpha-isopropyl-4-chlorophenyl acetate;Belmark;esfenvalerate;fenvalarate;fenvalerate;Pydrin;pydrin, (R-(R*,R*))-isomer;pydrin, (R-(R*,S*))-isomer;pydrin, (S-(R*,R*))-isomer;pydrin, (S-(R*,S*))-isomer;S-5602;Sumi-alpha;Sumicidin;WL 43775


 T,
T,  N
N




