Chemical Property of 1,2-Diphenylhydrazine
Chemical Property:
- Appearance/Colour:yellow crystalline powder
- Vapor Pressure:0.07mmHg at 25°C
- Melting Point:123-126 °C(lit.)
- Refractive Index:1.7
- Boiling Point:229.3 °C at 760 mmHg
- PKA:3.02±0.70(Predicted)
- Flash Point:87.7 °C
- PSA:24.06000
- Density:1.179 g/cm3
- LogP:3.27160
- Storage Temp.:Refrigerator
- Solubility.:Soluble in ethanol (Weast, 1986).
- Water Solubility.:221 mg/L at 25 °C (U.S. EPA, 1980a)
- XLogP3:2.9
- Hydrogen Bond Donor Count:2
- Hydrogen Bond Acceptor Count:2
- Rotatable Bond Count:3
- Exact Mass:184.100048391
- Heavy Atom Count:14
- Complexity:128
- Transport DOT Label:Poison
- Purity/Quality:
-
98% *data from raw suppliers
1,2-DiphenylHydrazine *data from reagent suppliers
Safty Information:
- Pictogram(s):
 T,
T,  N
N
- Hazard Codes:T,N
- Statements:
45-22-50/53
- Safety Statements:
53-45-60-61
- MSDS Files:
-
SDS file from LookChem
Useful:
- Chemical Classes:Nitrogen Compounds -> Hydrazines
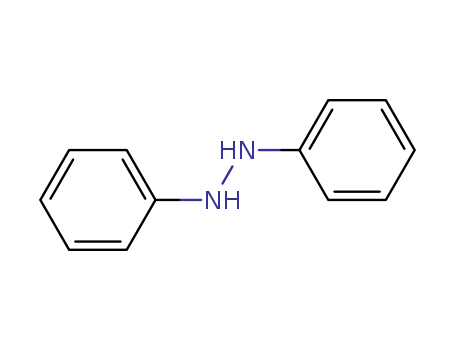
- Canonical SMILES:C1=CC=C(C=C1)NNC2=CC=CC=C2
- Inhalation Risk:A harmful concentration of airborne particles can be reached quickly when dispersed.
- Effects of Short Term Exposure:May cause mechanical irritation.
- Effects of Long Term Exposure:This substance is probably carcinogenic to humans.
-
General Description
1,2-Diphenylhydrazine (also known as hydrazobenzene) is an intermediate formed during the catalytic hydrogenation of azobenzene, as demonstrated in the presence of a cuboidal Mo3S4 cluster. The reaction proceeds via a sulfur-based H2 activation mechanism, where 1,2-diphenylhydrazine appears and disappears at similar rates, suggesting its transient role in the pathway toward aniline formation. This intermediate is part of two interconnected catalytic cycles, supported by DFT calculations and experimental kinetics. Additionally, diarylhydrazines like 1,2-diphenylhydrazine can undergo photocatalytic acceptorless dehydrogenation to form aromatic azo compounds, highlighting their utility in reversible hydrogen storage applications.


 T,
T,  N
N




