10.1021/acs.orglett.0c03990
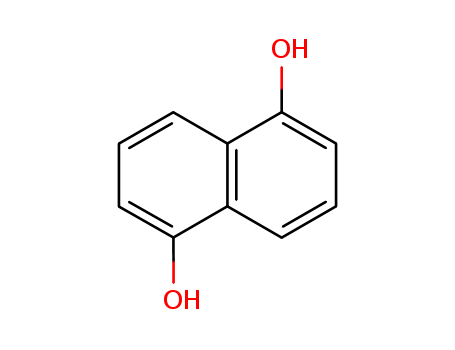
The study presents an asymmetric total synthesis of (+)-PD-116740, an angucyclinone with potent anticancer activity. The synthesis involved a convergent approach with naphthalene-1,5-diol as the starting material, which was converted into quinone derivatives and then reduced to hydroquinone. Selective protection and bromination steps led to the formation of coupling precursors. The D ring fragment was prepared from benzyl alcohol, with TBS protection, lithium/bromide exchange, and conversion to an alkyne. A Pd-catalyzed Suzuki-Miyama coupling reaction combined the A/B and D ring fragments. Asymmetric dihydroxylation using osmium tetroxide and a chiral ligand introduced chirality. The core trans-9,10-dihydrophenanthrene-9,10-diol unit was constructed via oxidative cyclization using Cu(OH)OTf·NMI2 as the oxidant, which also facilitated phenol oxidation. Finally, global deprotection yielded PD-116740.


 Xn,
Xn,  N,
N,  Xi
Xi




