Chemical Property of Iodine pentoxide
Chemical Property:
- Appearance/Colour:needle shaped crystals
- Melting Point:300-350 °C (dec.)
- Boiling Point:184.3°C at 760 mmHg
- PSA:77.51000
- Density:5.08 g/mL at 25 °C
- LogP:1.22780
- Storage Temp.:Store at +5°C to +30°C.
- Sensitive.:Light Sensitive & Hygroscopic
- Water Solubility.:soluble
- XLogP3:-0.2
- Hydrogen Bond Donor Count:0
- Hydrogen Bond Acceptor Count:5
- Rotatable Bond Count:0
- Exact Mass:333.78352
- Heavy Atom Count:7
- Complexity:124
- Purity/Quality:
-
99% *data from raw suppliers
Iodine(V) oxide, 99% *data from reagent suppliers
Safty Information:
- Pictogram(s):
 O,
O, C
C
- Hazard Codes:O,C
- Statements:
8-34
- Safety Statements:
17-26-27-36/37/39-45
- MSDS Files:
-
SDS file from LookChem
Useful:
- Chemical Classes:Other Classes -> Other Inorganic Compounds
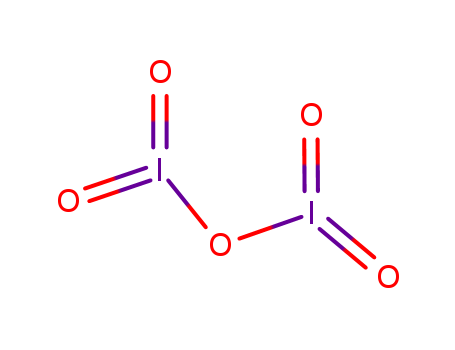
- Canonical SMILES:O=I(=O)OI(=O)=O
-
Physical Properties
White crystals; hygroscopic; density 4.98 g/cm3; decomposes around 300°C; highly soluble in water; soluble in nitric acid; insoluble in ethanol, ether and carbon disulfide.
-
Uses
Iodine pentoxide is used for analysis of carbon monoxide and for CO removal from air. It also is used as an oxidizing agent in other oxidation reactions. Iodine (V) oxide is used as an oxidizing agent to remove carbon monoxide from gas. Oxidizes carbon monoxide to carbon dioxide with the formn of iodine (Ditte's reaction, which proceeds rapidly at 65° or above, but slowly at room temp). This reaction is used in gas analysis and for removing carbon monoxide from the air (in respirators preferably in presence of some H2SO4).
-
Physical properties
White crystals; hygroscopic; density 4.98 g/cm3; decomposes around 300°C;highly soluble in water; soluble in nitric acid; insoluble in ethanol, ether andcarbon disulfide.


 O,
O, C
C




