10.1002/cbic.201300187
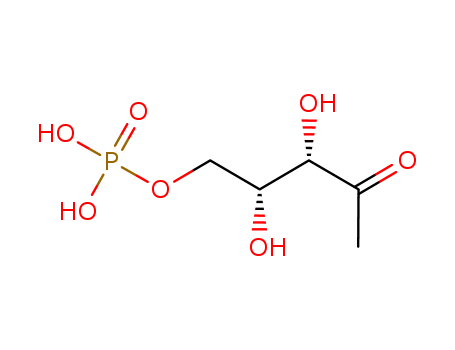
The research study on DXP Synthase-Catalyzed C-N Bond Formation, which is crucial for the selective inhibitor design targeting the enzyme DXP synthase. The purpose of the study was to understand the substrate specificity of DXP synthase, particularly its affinity for aromatic nitroso substrates, and to explore its potential as a drug target for anti-infective agents. The researchers discovered that DXP synthase has a high affinity for aromatic nitroso substrates, which are more reactive than their aldehyde counterparts, and that it can catalyze the formation of C-N bonds to generate aromatic hydroxamic acids or amides. They also found that DXP synthase has a larger active site compared to related enzymes like pyruvate dehydrogenase (PDH), which allows it to accommodate sterically demanding substrates. The study concluded that incorporating aryl acceptor substrate mimics into unnatural bisubstrate analogues could lead to selective inhibitors of DXP synthase. Key chemicals used in the process include 1-deoxy-d-xylulose 5-phosphate (DXP), pyruvate, d-glyceraldehyde 3-phosphate (d-GAP), thiamin diphosphate (ThDP), and various aromatic nitroso substrates such as nitrosobenzene and nitrosonaphthols. The researchers also synthesized benzylacetylphosphonate (BnAP) as a potential selective inhibitor of DXP synthase, demonstrating its effectiveness in inhibiting the enzyme with higher selectivity compared to PDH.