- 10241-05-1Molybdenum chloride(MoCl5)
- 102410-65-1Benzeneacetic acid, a-[[(9H-fluoren-9-ylmethoxy)carbonyl]amino]-,(aS)-
- 102417-95-85-DTAF [5-(4,6-Dichlorotriazinyl)aminofluorescein]
- 102418-16-6Thaimycin B (9CI)
- 10241-97-11H-Indole-2-carboxylicacid, 5-methyl-
- 10242-01-01H-Indole-3-carboxylicacid, 5-methoxy-
- 10242-02-11H-Indole-3-carboxylicacid, 5-methyl-
- 10242-03-21H-Indole-3-carboxylicacid, 6-nitro-
- 10242-08-72-Benzofurancarboxylicacid, 5-methoxy-
- 10242-10-12-Benzofurancarboxylicacid, 5-chloro-
Hot Products
- 104987-11-3Tacrolimus
- 141-53-7Sodium formate
- 8001-54-5Quaternary ammonium compounds, alkylbenzyldimethyl, chlorides
- 9003-39-8Povidone
- 10161-34-9Trenbolone acetate
- 402957-28-2Telaprevir
- 68-19-9Cyanocobalamin
- 7631-86-9Silicon dioxide
- 302-79-4Tretinoin
- 77-92-9Citric acid

|
Basic Information |

|
Post buying leads |

|
Suppliers |

| Name |
Trichlorocobalt |
EINECS | 233-574-8 |
| CAS No. | 10241-04-0 | Density | g/cm3 |
| PSA | 0.00000 | LogP | -8.98800 |
| Solubility | N/A | Melting Point |
N/A |
| Formula | Cl3Co | Boiling Point | °Cat760mmHg |
| Molecular Weight | 165.352 | Flash Point | °C |
| Transport Information | N/A | Appearance | N/A |
| Safety | Risk Codes | N/A | |
| Molecular Structure |
|
Hazard Symbols | N/A |
| Synonyms |
Cobalttrichloride; Cobaltic chloride |
Trichlorocobalt Synthetic route
- 7646-79-9
cobalt(II) chloride

- 10241-04-0
cobalt(III) chloride

| Conditions | Yield |
|---|---|
| With Cl2 In ethanol oxidation at -60°C;; | |
| With HCl In ethanol Electrochem. Process; anodic oxidation of CoCl2, dissolved in cold alcoholic HCl, formation of a green soln.;; | |
| With HCl; Cl2 In ethanol oxidation of CoCl2, dissolved in cold alcoholic HCl, by chlorination, formation of a green soln.;; |

| Conditions | Yield |
|---|---|
| In neat (no solvent) calcination with NH4Cl;; | |
| In neat (no solvent) calcination with NH4Cl;; |

| Conditions | Yield |
|---|---|
| In diethyl ether suspension of freshly prepared Co2O3 is treated at -5°C in the dark with dry HCl-gas;; | |
| In diethyl ether suspension of Co2O3 in ether is treated in the dark with HCl-gas;; | |
| In diethyl ether suspension of freshly prepared Co2O3 is treated at -5°C in the dark with dry HCl-gas;; | |
| In diethyl ether suspension of Co2O3 in ether is treated in the dark with HCl-gas;; |

| Conditions | Yield |
|---|---|
| In ethanol gaseous HCl is passed into a suspension of Co2O3*xH2O in abs. alcohol;; solution is concentrated in vacuum at -15 - -20°C to a small volume and stored over CaCl2, P2O5 and NaOH at low temperatures;; |

| Conditions | Yield |
|---|---|
| In diethyl ether Electrolysis; solution of HCl in ether is electrolyzed with Co-anode and Cu-cathode at 0°C in the dark;; | |
| In diethyl ether Electrolysis; solution of HCl in ether is electrolyzed with Co-anode and Cu-cathode at 0°C in the dark;; |

| Conditions | Yield |
|---|---|
| With CaCl2 In ethanol -70°C;; |

| Conditions | Yield |
|---|---|
| In neat (no solvent) CoCl2 is treated with Cl2 at ambient temperatures;; | |
| In neat (no solvent) at higher temperatures;; | |
| In neat (no solvent) at higher temperatures;; | |
| In neat (no solvent) CoCl2 is treated with Cl2 at ambient temperatures;; | |
| In ethanol -60°C;; |
- 7291-22-7
perdeuteriopyridine

- 39091-63-9
2-bromo-[1,1,1,2,3,3,3-2H7]-propane

- 10241-04-0
cobalt(III) chloride

| Conditions | Yield |
|---|---|
| With deuteriated sodium hydroxide In d(4)-methanol CoCl3 and glyoxime were dissolved in MeOD containing NaOD and pyridine-d5 under N2, then isopropyl bromide was added under D2 and stirred; neutralized with DCl, the soln. was diluted with D2O and filtered, the crystals were washed with D2O, petroleum ether and ether, then dissolved in CDCl3/MeOD, and pptd. by D2O, drying; | 86% |
- 10241-04-0
cobalt(III) chloride

- 624746-31-2
4-(2-(1,5-dimethyl-3-oxo-2-phenyl-2,3-dihydro-1H-pyrazol-4-yl)hydrazono)-3-methyl-5-oxo-4,5-dihydro-1H-pyrazole-1-carbothioamide

- 624746-09-4
[Co(N-(3-methyl 1-thiocarbamyl-5-oxo-2-pyrazolin-4-ylene)-N'-(4'-antipyrine)hydrazine)Cl3]

| Conditions | Yield |
|---|---|
| In ethanol; water refluxing soln. metal salt in aq. EtOH and soln. ligand in EtOH for 1 h; react. mixt. was cooled, ppt. was filtered, washed with EtOH and benzene, and dried over P4O10; elem. anal.; | 65% |
- 10241-04-0
cobalt(III) chloride

- 142557-68-4
1,2-dicyano-4,5-diheptylbenzene

- 165285-38-1
(2,3,9,10,16,17,23,24-octaheptylphthalocyaninato)cobalt(II)

| Conditions | Yield |
|---|---|
| In ethylene glycol refluxing (24 h); cooling, pouring into MeOH (pptn.), centrifugation, washing (acetone), drying, chromy. (silica gel/toluene), collection of blue fraction, evapn.; elem. anal.; | 63% |
Trichlorocobalt Chemical Properties
IUPAC Name: Trichlorocobalt
CAS: 10241-04-0
The Molecular formula of Trichlorocobalt ( CAS NO.10241-04-0): Cl3Co
The Molecular Weight of Trichlorocobalt ( CAS NO.10241-04-0): 165.2922
Molecular Structure of Trichlorocobalt ( CAS NO.10241-04-0):
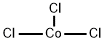
Enthalpy of Vaporization: 16.15 kJ/mol
Vapour Pressure: 33900 mmHg at 25°C
Water Solubility: 6616(mg/L) at 25 °C
Trichlorocobalt Specification
The chemical synonyms of Trichlorocobalt ( CAS NO.10241-04-0) are Cobalt trichloride ; Cobaltic Chloride ; Cobalt(III) trichloride ; Trichlorocobalt(III) .

