Products Categories
| CAS No.: | 104-98-3 |
|---|---|
| Name: | Urocanic acid |
| Molecular Structure: | |
|
|
|
| Formula: | C6H6N2O2 |
| Molecular Weight: | 138.126 |
| Synonyms: | 4-Imidazolylacrylic acid;5-Imidazoleacrylicacid;NSC 66357;Urocaninic acid;3-(1H-Imidazol-4-yl)acrylic acid;3-(4-Imidazolyl)acrylic acid;3-(1H-Imidazol-4-yl)-2-propenoic acid;Imidazole-4-acrylic acid (8CI);2-Propenoicacid, 3-(1H-imidazol-4-yl)- (9CI); |
| EINECS: | 203-258-4 |
| Density: | 1.43 g/cm3 |
| Melting Point: | 226-228 °C(lit.) |
| Boiling Point: | 456.9 °C at 760 mmHg |
| Flash Point: | 230.1 °C |
| Solubility: | SLIGHTLY SOLUBLE |
| Appearance: | white to beige fine powder |
| Safety: | 22-24/25 |
| PSA: | 65.98000 |
| LogP: | 0.50750 |
- 144851-82-1METHYL2-AMINO-3-FLUOROBENZOATE
- 483366-12-7(2S,4R)-1-Boc-2-cyano-4-hydroxypyrrolidine
- 173606-50-3BOC-10-AMINODECANOIC ACID
- 361456-36-2METHYL (R)-(+)-ISOCYANATO-3-PHENYLPROPI&
- 5156-58-1N-(1-Benzyl-4-pipperidinyl)-N-phenylpropanamide HCl
- 81281-59-67-Benzylideneaminotheophylline
- 50288-62-5threo-Phenyl-2-piperidyl acetamide
- 82993-81-5D-threo-Ritalinic acid hydrochloride
- 47087-37-6Z-D-Glu-OMe
- 1219080-61-1IMIDAZOLE-2-BORONIC ACID

| Conditions | Yield |
|---|---|
| With histidase EC 4.3.1.3; Tris buffer In water at 37℃; for 24h; pH=9.0; | |
| With histidase Enzymatic reaction; |

- 104-98-3
urocanic acid


- 49549-75-9
4-(3-hydroxypropyl)-1H-imidazole

| Conditions | Yield |
|---|---|
| With lithium aluminium tetrahydride | 100% |

| Conditions | Yield |
|---|---|
| With thionyl chloride | 98% |
| With hydrogenchloride Reflux; | 94% |
| With sulfuric acid; magnesium sulfate Fischer esterification; Reflux; | 93% |

- 67-56-1
methanol


- 104-98-3
urocanic acid


- 53958-94-4
3-(1H-imidazol-4-yl)propionic acid methyl ester hydrochloride

| Conditions | Yield |
|---|---|
| Stage #1: urocanic acid With hydrogenchloride; palladium 10% on activated carbon; hydrogen In methanol; water at 45℃; under 760.051 Torr; for 96h; Large scale; Stage #2: methanol With hydrogenchloride at 45℃; Large scale; | 98% |

| Conditions | Yield |
|---|---|
| at 250℃; | 93% |
| at 110℃; | 71% |
| at 240℃; under 0.0165017 Torr; for 6h; | 66% |

- 67-56-1
methanol


- 104-98-3
urocanic acid


- 52838-22-9
methyl 3-[1H-imidazol-4-yl]propenoate hydrochloride salt

| Conditions | Yield |
|---|---|
| With hydrogenchloride at 0℃; for 2h; | 87% |

- 57260-73-8
N-BOC-1,2-diaminoethane


- 104-98-3
urocanic acid

| Conditions | Yield |
|---|---|
| With benzotriazol-1-ol; 1-ethyl-(3-(3-dimethylamino)propyl)-carbodiimide hydrochloride In N,N-dimethyl-formamide at 0 - 20℃; | 86% |

| Conditions | Yield |
|---|---|
| Stage #1: urocanic acid With palladium on activated charcoal; hydrogen In methanol at 25℃; Stage #2: ethanol With sulfuric acid In methanol Reflux; Inert atmosphere; | 82% |
| Stage #1: urocanic acid With palladium on activated charcoal; hydrogen In methanol at 25℃; Stage #2: ethanol With sulfuric acid Reflux; Inert atmosphere; | 82% |

| Conditions | Yield |
|---|---|
| Stage #1: methanol; urocanic acid With thionyl chloride for 16h; Reflux; Stage #2: With sodium carbonate In water; ethyl acetate | 81% |

- 70-34-8
2,4-Dinitrofluorobenzene


- 104-98-3
urocanic acid


- 507444-19-1
3-[1-(2,4-dinitrophenyl)-1H-imidazol-4-yl]propenoic acid

| Conditions | Yield |
|---|---|
| With sodium hydrogencarbonate In methanol; water at 20℃; for 16h; Substitution; | 76% |
Chemistry
Chemical Name: Urocanic acid
Molecular Formula: C6H6N2O2
molecular structure:
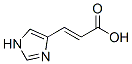
Formula Weight: 138.12
CAS No.: 104-98-3
CBNumber: CB8750635
Synonyms:3-(1h-imidazol-4-yl)-2-propenoicaci;3-(1h-imidazol-4-yl)-2-propenoicacid;3-imidazol-4-ylacrylic;3-imidazol-4-ylacrylicacid;5-imidazoleacrylicacid;imidazoleacrylicacid;3-(1H-IMIDAZOL-4-YL)ACRYLIC ACID;3-(4-IMIDAZOLY)ACRYLIC ACID
storage temp:2-8°C
mp :226-228 °C
slightly soluble in water
Urocanic acid is an intermediate in the catabolism of L-histidine.
Urocanic acid was detected in animal sweat and skin where, among other possible functions, it acts as an endogenous sunscreen or photoprotectant against UVB-induced DNA damage. When exposed to UVB irradiation, trans-urocanic acid is converted in vitro and in vivo to the cis isomer. The cis form is known to activate suppressor T cellsUrocanic acid is found predominantly in the stratum corneum of the skin and it is likely that most of it is derived from filaggrin catabolism (a histidine-rich protein).
History
Toxicity Data With Reference
| 1. | dni-hmn:oth 1 mmol/L | JIDEAE Journal of Investigative Dermatology. 65 (1975),400. | ||
| 2. | ivn-mus LD50:100 mg/kg | 85GDA2 CRC Handbook of Antibiotic Compounds. 5 (1981),81. |

