Products Categories
| CAS No.: | 113852-37-2 |
|---|---|
| Name: | Cidofovir |
| Article Data: | 10 |
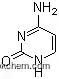
| Molecular Structure: | |
|
|
|
| Formula: | C8H14N3O6P |
| Molecular Weight: | 279.189 |
| Synonyms: | Phosphonicacid, [[(1S)-2-(4-amino-2-oxo-1(2H)-pyrimidinyl)-1-(hydroxymethyl)ethoxy]methyl]-(9CI);Phosphonic acid,[[2-(4-amino-2-oxo-1(2H)-pyrimidinyl)-1-(hydroxymethyl)ethoxy]methyl]-, (S)-;(S)-1-(3-hydroxy-2-phosphonomethoxypropyl)cytosine;(S)-HPMPC;1-(S)-(3-Hydroxy-2-phosphonylmethoxypropyl)cytosine;1-[(S)-3-Hydroxy-2-(phosphonomethoxy)propyl]cytosine;Cidovir;GS 0504;HPMPC;Vistide; |
| EINECS: | 638-807-0 |
| Density: | 1.76 g/cm3 |
| Melting Point: | 260 °C |
| Boiling Point: | 609.5 °C at 760 mmHg |
| Flash Point: | 322.4 °C |
| Appearance: | fluffy white powder |
| Hazard Symbols: | T |
| Risk Codes: | 25-38 |
| Safety: | 36-37-45 |
| PSA: | 157.71000 |
| LogP: | -1.08060 |
- 144851-82-1METHYL2-AMINO-3-FLUOROBENZOATE
- 483366-12-7(2S,4R)-1-Boc-2-cyano-4-hydroxypyrrolidine
- 173606-50-3BOC-10-AMINODECANOIC ACID
- 361456-36-2METHYL (R)-(+)-ISOCYANATO-3-PHENYLPROPI&
- 5156-58-1N-(1-Benzyl-4-pipperidinyl)-N-phenylpropanamide HCl
- 81281-59-67-Benzylideneaminotheophylline
- 50288-62-5threo-Phenyl-2-piperidyl acetamide
- 82993-81-5D-threo-Ritalinic acid hydrochloride
- 47087-37-6Z-D-Glu-OMe
- 1219080-61-1IMIDAZOLE-2-BORONIC ACID
- 120362-35-8
(S)-1-<3-Hydroxy-2-<(diethylphosphonyl)methoxy>propyl>cytosine

- 113852-37-2
cidofovir

| Conditions | Yield |
|---|---|
| With trimethylsilyl bromide In acetonitrile for 14h; Ambient temperature; | 95% |
| With trimethylsilyl bromide | 80% |
- 132336-37-9
N4-benzoyl-1-(S)-(3-hydroxy-2-phosphonomethoxypropyl)cytosine

- 113852-37-2
cidofovir

| Conditions | Yield |
|---|---|
| With ammonium hydroxide at 20℃; for 3h; | 80% |
| With ammonium hydroxide for 4h; Ambient temperature; Yield given; |

- 113852-37-2
cidofovir

| Conditions | Yield |
|---|---|
| With ammonium hydroxide at 20℃; for 3h; | 71% |
- 18002-25-0
4-methoxy-2-pyrimidone

- 141110-74-9
(R)-2--3-ytimethylacetyl-1,2,3-propanetriol 1-p-toluenesulfonate

- 113852-37-2
cidofovir

| Conditions | Yield |
|---|---|
| Yield given. Multistep reaction; |
- 71-30-7
Cytosine

- 141110-74-9
(R)-2--3-ytimethylacetyl-1,2,3-propanetriol 1-p-toluenesulfonate

- 113852-37-2
cidofovir

| Conditions | Yield |
|---|---|
| With trimethylsilyl bromide; caesium carbonate 1.) DMFA, 100 deg C, 72 h, 2.) CH3CN, RT, 24 h; Yield given. Multistep reaction; |
- 148873-52-3
bis(2-propyl) N4-benzoyl-1-(S)-(2-phosphonomethoxy-3-triphenylmethoxypropyl)cytosine

- 113852-37-2
cidofovir

| Conditions | Yield |
|---|---|
| With trimethylsilyl bromide; sodium methylate 1.) CH3CN, overnight, 2.) CH3OH, RT, overnight; Yield given. Multistep reaction; |
- 148873-53-4
bis(2-propyl) 1-(S)-(3-hydroxy-2-phosphonomethoxypropyl)cytosine

- 113852-37-2
cidofovir

| Conditions | Yield |
|---|---|
| With trimethylsilyl bromide; water; triethylamine 1.) CH3CN, overnight, 2.) 20 min; Multistep reaction; |
- 132336-34-6
(S)-N1-[2-hydroxy-3-(triphenylmethoxy)propyl]-N4-benzoylcytosine

- 113852-37-2
cidofovir

| Conditions | Yield |
|---|---|
| Multi-step reaction with 4 steps 1: NaH / dimethylformamide / 6 h / 0 °C 2: HCl(g) / CH2Cl2 / 0.17 h / 0 - 5 °C 3: TMSBr / CH2Cl2 / 18 h / Ambient temperature 4: conc. NH4OH / 4 h / Ambient temperature View Scheme | |
| Multi-step reaction with 4 steps 1: potassium carbonate / acetonitrile / 12 h / 60 °C 2: trifluoroacetic acid / dichloromethane / 6 h / 20 °C 3: trimethylsilyl bromide / dichloromethane / 18 h / 20 °C 4: ammonium hydroxide / 3 h / 20 °C View Scheme |
- 132336-36-8
diethyl (S)-4-N-benzoyl-1-[3-hydroxy-2-(phosphonomethoxy)propyl]cytosine

- 113852-37-2
cidofovir

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: TMSBr / CH2Cl2 / 18 h / Ambient temperature 2: conc. NH4OH / 4 h / Ambient temperature View Scheme |
- 132336-35-7
[(S)-1-(4-Benzoylamino-2-oxo-2H-pyrimidin-1-ylmethyl)-2-trityloxy-ethoxymethyl]-phosphonic acid diethyl ester

- 113852-37-2
cidofovir

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: HCl(g) / CH2Cl2 / 0.17 h / 0 - 5 °C 2: TMSBr / CH2Cl2 / 18 h / Ambient temperature 3: conc. NH4OH / 4 h / Ambient temperature View Scheme | |
| Multi-step reaction with 3 steps 1: trifluoroacetic acid / dichloromethane / 6 h / 20 °C 2: trimethylsilyl bromide / dichloromethane / 18 h / 20 °C 3: ammonium hydroxide / 3 h / 20 °C View Scheme |
Analytical Methods
Antineoplastic Agents : substances that inhibit or prevent the proliferation of NEOPLASMS
Specification
The Phosphonic acid,P-[[(1S)-2-(4-amino-2-oxo-1(2H)-pyrimidinyl)-1-(hydroxymethyl)ethoxy]methyl]-, with the CAS registry number 113852-37-2, is also known as 1-(S)-(3-Hydroxy-2-phosphonylmethoxypropyl)cytosine. This chemical's molecular formula is C8H14N3O6P and molecular weight is 279.19. What's more, its systematic name is ({[(2S)-1-(4-amino-2-oxopyrimidin-1(2H)-yl)-3-hydroxypropan-2-yl]oxy}methyl)phosphonic acid. Its classification codes are: (1)Anti-HIV agents; (2)Anti-Infective Agents; (3)Anti-Retroviral Agents; (4)Antineoplastic Agents; (5)Antiviral; (6)Antiviral agents; (7)Radiation-Sensitizing Agents. It is a potent, selective inhibitor of human cytomegalovirus replication. It is an injectable antiviral medication for the treatment of cytomegalovirus (CMV) retinitis in patients with AIDS.
Physical properties of Phosphonic acid,P-[[(1S)-2-(4-amino-2-oxo-1(2H)-pyrimidinyl)-1-(hydroxymethyl)ethoxy]methyl]- are: (1)ACD/LogP: -3.37; (2)# of Rule of 5 Violations: 1; (3)ACD/LogD (pH 5.5): -6.65; (4)ACD/LogD (pH 7.4): -7.31; (5)ACD/BCF (pH 5.5): 1; (6)ACD/BCF (pH 7.4): 1; (7)ACD/KOC (pH 5.5): 1; (8)ACD/KOC (pH 7.4): 1; (9)#H bond acceptors: 9; (10)#H bond donors: 5; (11)#Freely Rotating Bonds: 7; (12)Polar Surface Area: 99.71 Å2; (13)Index of Refraction: 1.656; (14)Molar Refractivity: 58.29 cm3; (15)Molar Volume: 158.6 cm3; (16)Polarizability: 23.1×10-24cm3; (17)Surface Tension: 90.5 dyne/cm; (18)Density: 1.76 g/cm3; (19)Flash Point: 322.4 °C; (20)Enthalpy of Vaporization: 103.84 kJ/mol; (21)Boiling Point: 609.5 °C at 760 mmHg; (22)Vapour Pressure: 2.11E-17 mmHg at 25°C.
Preparation: first dissolving 1-(2, 3-dihydroxypropyl)cytosine in triethyl phosphate, then adding in ClCH2P(O)Cl2 by stiring, and then mixing and rejoining the ether, filtering and collecting the precipitates which are washed with ether and then dried in the vacuum. Then dissolving the intermediate product in water and conducting backflow which is then neutralized with triethylamine and evaporated out solvent in the vacuum. Finally by column chromatography, the product is prepared.
You can still convert the following datas into molecular structure:
(1)SMILES: O=C1/N=C(\C=C/N1C[C@H](OCP(=O)(O)O)CO)N
(2)Std. InChI: InChI=1S/C8H14N3O6P/c9-7-1-2-11(8(13)10-7)3-6(4-12)17-5-18(14,15)16/h1-2,6,12H,3-5H2,(H2,9,10,13)(H2,14,15,16)/t6-/m0/s1
(3)Std. InChIKey: VWFCHDSQECPREK-LURJTMIESA-N