
|
Basic Information |

|
Post buying leads |

|
Suppliers |

|
Cas Database |
- Name 4-Butyl-4H-1,2,4-triazole
- EINECS240-351-9
- CAS No. 16227-10-4
- Density1.04g/cm3
- PSA30.71000
- LogP1.07820
- SolubilityN/A
- Melting PointN/A
- FormulaC6H11 N3
- Boiling Point220.9°Cat760mmHg
- Molecular Weight125.173
- Flash Point87.4°C
- Transport InformationN/A
- AppearanceN/A
- SafetyA poison by ingestion and skin contact. When heated to decomposition it emits toxic fumes of NOx.
- Risk CodesN/A
- Molecular Structure
- Hazard SymbolsN/A
- SynonymsN/A
- Article Data9
4-Butyl-4H-1,2,4-triazole Synthetic route
- 288-88-0
1,2,4-Triazole

- 109-65-9
1-bromo-butane

A
- 6086-22-2
1-butyl-1H-[1,2,4]triazole

B
- 16227-10-4
4-n-butyl-1,2,4-triazole
Conditions

| Conditions | Yield |
|---|---|
| With 1,8-diazabicyclo[5.4.0]undec-7-ene In tetrahydrofuran at 0 - 20℃; Alkylation; | A 85% B n/a |
| With aluminum oxide; potassium fluoride; tetrabutylammomium bromide for 24h; Ambient temperature; Yield given. Yields of byproduct given; | |
| With sodium hydroxide In N,N-dimethyl-formamide at 20℃; Alkylation; |
- 288-88-0
1,2,4-Triazole

- 109-65-9
1-bromo-butane

A
- 6086-22-2
1-butyl-1H-[1,2,4]triazole

B
- 16227-10-4
4-n-butyl-1,2,4-triazole
Conditions

| Conditions | Yield |
|---|---|
| With lithium hydroxide; tetrabutylammomium bromide for 24h; Product distribution; Ambient temperature; also benzotriazole; other alkyl halides; var. base as reagents; also reactions in the absence of base; var. temperatures and reaction time; also with conventional heating or microwave heating instead of phase transfer catalysis; |
- 109-73-9
N-butylamine

- 122-51-0
orthoformic acid triethyl ester

- 624-84-0
formic acid hydrazide

- 16227-10-4
4-n-butyl-1,2,4-triazole
Conditions

| Conditions | Yield |
|---|---|
| Stage #1: orthoformic acid triethyl ester; formic acid hydrazide In methanol Heating; Stage #2: N-butylamine In methanol Heating; Further stages.; |
Conditions

| Conditions | Yield |
|---|---|
| With magnesium carbonate at 200℃; for 20h; Inert atmosphere; | 5.25 g |
- 74-83-9
methyl bromide

- 16227-10-4
4-n-butyl-1,2,4-triazole

- 1268852-02-3
4-butyl-1-methyl-4H-1,2,4-triazol-1-ium bromide
Conditions

| Conditions | Yield |
|---|---|
| In acetonitrile for 36h; Reflux; Inert atmosphere; Schlenk technique; | 96% |
- 507-19-7
t-butyl bromide

- 16227-10-4
4-n-butyl-1,2,4-triazole
Conditions

| Conditions | Yield |
|---|---|
| In acetonitrile for 36h; Reflux; Inert atmosphere; Schlenk technique; | 92% |
- 16227-10-4
4-n-butyl-1,2,4-triazole

- 1343472-85-4
BH2(C2H2N3(C4H9))2(1+)*I(1-)=[BH2(C2H2N3(C4H9))2]I
Conditions

| Conditions | Yield |
|---|---|
| With trimethylamine-borane; iodine In toluene for 24h; Reflux; | 85.8% |
- 75-22-9
trimethylamine-borane

- 7553-56-2
iodine

- 16227-10-4
4-n-butyl-1,2,4-triazole

- 1343472-85-4
BH2(C2H2N3(C4H9))2(1+)*I(1-)=[BH2(C2H2N3(C4H9))2]I
Conditions

| Conditions | Yield |
|---|---|
| In toluene I2 added to toluene soln. of Me3NBH3 , stirred at room temp. for 1 h, ligand added (1:2:4 molar ratio), mixt. refluxed for 24 h; filtered off, washed (hexane, Et2O, toluene), dried (80°C, vac.),recrystd. (hot EtOH), elem. anal.; | 85.8% |
4-Butyl-4H-1,2,4-triazole Chemical Properties
BUTRIZOL 's Molecular formula: C6H11N3
BUTRIZOL 's Molar mass: 125.17
Structure: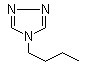
EINECS: 240-351-9
Synonyms: 4-BUTYL-4H-1,2,4-TRIAZOLE;4-butyl-1,2,4-triazole;Triazbutil;4-N-BUTYL-4H-1,2,4-TRIAZOLE;Dithane R-24;Butrizol;RH-124
BUTRIZOL 's Molar mass: 125.17
Structure:

EINECS: 240-351-9
Synonyms: 4-BUTYL-4H-1,2,4-TRIAZOLE;4-butyl-1,2,4-triazole;Triazbutil;4-N-BUTYL-4H-1,2,4-TRIAZOLE;Dithane R-24;Butrizol;RH-124
4-Butyl-4H-1,2,4-triazole Uses
BUTRIZOL is agricultural Chemical.
4-Butyl-4H-1,2,4-triazole Toxicity Data With Reference
| 1. | orl-rat LD50:50 mg/kg | 85ARAE Agricultural Chemicals, 4 (1976/77),94. | ||
| 2. | skn-rbt LD50:315 mg/kg | FMCHA2 Farm Chemicals Handbook .(Meister Publishing,Willoughy, OH.: )1983,C131. |
4-Butyl-4H-1,2,4-triazole Safety Profile
A poison by ingestion and skin contact. When heated to decomposition it emits toxic fumes of NOx.
RTECS: XZ4150000
RTECS: XZ4150000
Related Products
- 16227-12-64H-1,2,4-Triazole,4-phenyl-
- 16228-00-52,4-Imidazolidinedione,3-(hydroxymethyl)-5,5-dimethyl-
- 162284-50-66-O-Benzyl-2,3-di-O-acetyl-methyl-α-D-glucopyranoside
- 162285-30-54-Oxazolidinecarboxylicacid, hydrochloride, (4S)- (9CI)
- 16228-67-41-Propanone,3-(3-azabicyclo[3.2.2]non-3-yl)-1-(2-thienyl)-, hydrochloride (1:1)
- 162303-51-71-Butanol,titanium(4+) salt, monohydrate, homopolymer (9CI)
- 162303-66-4H-D-Val-Leu-Arg p-nitroanilide
- 1623-05-8Propane,1,1,1,2,2,3,3-heptafluoro-3-[(1,2,2-trifluoroethenyl)oxy]-
- 162307-09-72-Azabicyclo[2.2.1]hept-5-en-3-one,2-acetyl-
- 1623-08-1Phosphoric acid,bis(phenylmethyl) ester
Hot Products
- 104987-11-3Tacrolimus
- 141-53-7Sodium formate
- 8001-54-5Quaternary ammonium compounds, alkylbenzyldimethyl, chlorides
- 9003-39-8Povidone
- 10161-34-9Trenbolone acetate
- 402957-28-2Telaprevir
- 68-19-9Cyanocobalamin
- 7631-86-9Silicon dioxide
- 302-79-4Tretinoin
- 77-92-9Citric acid

