Products Categories
| CAS No.: | 1006-94-6 |
|---|---|
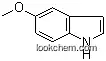
| Name: | 5-Methoxyindole |
| Molecular Structure: | |
|
|
|
| Formula: | C9H9NO |
| Molecular Weight: | 147.177 |
| Synonyms: | Indole, 5-methoxy-;1H-Indole,5-methoxy-;1/C9H9NO/c1-11-8-2-3-9-7(6-8)4-5-10-9/h2-6,10H,1H;Methoxy-5 indole [French];Femedol;5-methoxy indole;5-methyloxyl indole;5-Methoxylindole;5-methoxy-indole;5-Methyoxy Indole;5-Methoxyindole 98%;Methoxy-5 indole [French];1H-Indole, 5-methoxy-;Methoxy-5 indole;5-Methoxy-1H-indole;Indol-5-yl methyl ether;1H-Indole, 5-methoxy- (9CI); |
| EINECS: | 213-745-3 |
| Density: | 1.169 g/cm3 |
| Melting Point: | 52-55 °C(lit.) |
| Boiling Point: | 311.9 °C at 760 mmHg |
| Flash Point: | 109.2 °C |
| Solubility: | insoluble in water |
| Appearance: | white to light brownish crystalline powder |
| Hazard Symbols: |
 Xi Xi
|
| Risk Codes: | 36/37/38 |
| Safety: | 26-36-24/25-37/39 |
| Transport Information: | 50kgs |
| PSA: | 25.02000 |
| LogP: | 2.17650 |
- 144851-82-1METHYL2-AMINO-3-FLUOROBENZOATE
- 483366-12-7(2S,4R)-1-Boc-2-cyano-4-hydroxypyrrolidine
- 173606-50-3BOC-10-AMINODECANOIC ACID
- 361456-36-2METHYL (R)-(+)-ISOCYANATO-3-PHENYLPROPI&
- 5156-58-1N-(1-Benzyl-4-pipperidinyl)-N-phenylpropanamide HCl
- 81281-59-67-Benzylideneaminotheophylline
- 50288-62-5threo-Phenyl-2-piperidyl acetamide
- 82993-81-5D-threo-Ritalinic acid hydrochloride
- 47087-37-6Z-D-Glu-OMe
- 1219080-61-1IMIDAZOLE-2-BORONIC ACID

- 139717-71-8
5-methoxy-N-(4-toluenesulfonyl)-1H-indole


- 1006-94-6
5-methoxylindole

| Conditions | Yield |
|---|---|
| With sodium hydride In N,N-dimethyl acetamide at 60℃; for 5h; Inert atmosphere; | 90% |
| With formic acid; (4,4'-di-tert-butyl-2,2'-dipyridyl)-bis-(2-phenylpyridine(-1H))-iridium(III) hexafluorophosphate; N-ethyl-N,N-diisopropylamine In acetonitrile at 20℃; for 24h; Inert atmosphere; Sealed tube; Irradiation; | 85% |
| With caesium carbonate In tetrahydrofuran; methanol at 64℃; for 2.5h; | |
| With cetyltrimethylammonim bromide; potassium hydroxide In tetrahydrofuran; water for 120h; Reflux; Green chemistry; |

- 10601-19-1
5-methoxyindole-3-carboxaldehyde


- 1006-94-6
5-methoxylindole

| Conditions | Yield |
|---|---|
| With palladium diacetate; potassium carbonate In ethyl acetate at 150℃; under 12929 Torr; for 0.833333h; Microwave irradiation; Molecular sieve; | 90% |
| With perchloric acid adsorbed on silica gel; anthranilic acid amide In acetonitrile at 80℃; for 6h; | 74% |

- 10242-01-0
5-Methoxyindole-3-carboxylic acid


- 1006-94-6
5-methoxylindole

| Conditions | Yield |
|---|---|
| With potassium carbonate In ethanol at 140℃; Schlenk technique; | 99% |

- 32989-62-1
4-methoxy-trans-2-[β-(dimethylamino)vinyl]-nitrobenzene


- 1006-94-6
5-methoxylindole

| Conditions | Yield |
|---|---|
| With hydrogen; palladium on activated charcoal In benzene at 25℃; under 2280 Torr; for 27h; | 68% |
| palladium In benzene |

- 845619-77-4
1-(5-methoxy-1H-indol-1-yl)-2,2-dimethylpropan-1-one


- 1006-94-6
5-methoxylindole

| Conditions | Yield |
|---|---|
| With water; 1,8-diazabicyclo[5.4.0]undec-7-ene In tetrahydrofuran for 18h; Reflux; | 99% |
| With lithium diisopropyl amide In tetrahydrofuran; hexane at 40 - 45℃; for 2h; | 92% |

- 126759-31-7
2-vinyl-4-methoxynitrobenzene


- 1006-94-6
5-methoxylindole

| Conditions | Yield |
|---|---|
| With carbon monoxide; triphenylphosphine; palladium diacetate In acetonitrile under 3040 Torr; for 19h; Heating; | 63% |
| Multi-step reaction with 2 steps 1: 50 percent / O2 (1 atm) / Na2PdCl4 / methanol / 24 h / Ambient temperature 2: 63 percent / Fe, acetic acid, 10percent HCl / ethanol / Temp. 70 - 75 deg C, 1 h. Temp. 85 deg C, 2 h View Scheme | |
| Multi-step reaction with 2 steps 1: 85 percent / oxygen / palladium(II) chloride, copper(I) chloride / 1,2-dimethoxy-ethane / 24 h / 50 - 60 °C 2: 1.) H2, 2.) aq. HCl / 10percent rhodium-carbon / 1.) ethanol, room temperature, 1 atmosphere, 3 h, 2.) room temperature, 3 h View Scheme |

- 20357-24-8
5-methoxy-2-nitro-benzaldehyde


- 86302-43-4
(tert-Butoxycarbonylmethylene)triphenylphosphorane


- 1006-94-6
5-methoxylindole

| Conditions | Yield |
|---|---|
| With triphenylphosphine In diphenylether at 260℃; for 1h; | 51% |


- 1006-94-6
5-methoxylindole

| Conditions | Yield |
|---|---|
| With hydrogen In N,N-dimethyl-formamide at 330 - 340℃; under 15001.5 Torr; for 10h; Pressure; Temperature; |

- 89302-15-8
2-(5-methoxy-2-nitrophenyl)acetonitrile


- 1006-94-6
5-methoxylindole

| Conditions | Yield |
|---|---|
| With hydrogen In methanol at 20℃; under 760.051 Torr; for 12h; | 88% |
| With hydrogen; acetic acid; 10% palladium on active carbon In ethanol under 2280 Torr; for 2h; Ambient temperature; | 83% |
| With hydrogen at 20℃; under 760.051 Torr; for 24h; Schlenk technique; | 68% |

| Conditions | Yield |
|---|---|
| With copper(l) iodide In N,N-dimethyl-formamide for 6h; Reflux; | 98% |
Specification
The 5-Methoxyindole, with the CAS registry number 1006-94-6, is also known as 1H-Indole, 5-methoxy- (9CI). It belongs to the product categories of Blocks; Indoles Oxindoles; Heterocycles; Indoles and Derivatives; Indole Derivative; Pyrroles & Indoles; Indoline & Oxindole; Indole; Indoles; Heterocyclic Compounds; Indole Series; Indole Derivatives; Simple Indoles; Chiral Compound; Pyrroles & Indoles; Building Blocks; Heterocyclic Building Blocks. Its EINECS registry number is 213-745-3. This chemical's molecular formula is C9H9NO and molecular weight is 147.17. Its IUPAC name is called 5-methoxy-1H-indole. What's more, this chemical's classification code is Drug / Therapeutic Agent. 5-Methoxyindole is used as intermediate of pharmaceuticals.
Physical properties of 5-Methoxyindole: (1)ACD/LogP: 2.06; (2)ACD/LogD (pH 5.5): 2.06; (3)ACD/LogD (pH 7.4): 2.06; (4)ACD/BCF (pH 5.5): 21.57; (5)ACD/BCF (pH 7.4): 21.57; (6)ACD/KOC (pH 5.5): 313.65; (7)ACD/KOC (pH 7.4): 313.65; (8)#H bond acceptors: 2; (9)#H bond donors: 1; (10)#Freely Rotating Bonds: 1; (11)Index of Refraction: 1.637; (12)Molar Refractivity: 45.2 cm3; (13)Molar Volume: 125.8 cm3; (14)Surface Tension: 45.7 dyne/cm; (15)Density: 1.169 g/cm3; (16)Flash Point: 109.2 °C; (17)Enthalpy of Vaporization: 53.08 kJ/mol; (18)Boiling Point: 311.9 °C at 760 mmHg; (19)Vapour Pressure: 0.00101 mmHg at 25°C.
Preparation of 5-Methoxyindole: this chemical can be prepared by 5-methoxy-indole-2-carboxylic acid. This reaction will need reagents quinoline and copper oxide-chromium oxide. The reaction temperature is 200 - 210 °C.

Uses of 5-Methoxyindole: it can be used to produce (5-methoxy-indol-3-yl)-oxoacetyl chloride. This reaction will need reagent diethyl ether.

When you are using this chemical, please be cautious about it as the following:
This chemical may cause inflammation to the skin or other mucous membranes. It is irritating to eyes, respiratory system and skin. In case of contact with eyes, you should rinse immediately with plenty of water and seek medical advice. Whenever you will contact it, please wear suitable protective clothing, gloves and eye/face protection.
You can still convert the following datas into molecular structure:
(1)Canonical SMILES: COC1=CC2=C(C=C1)NC=C2
(2)InChI: InChI=1S/C9H9NO/c1-11-8-2-3-9-7(6-8)4-5-10-9/h2-6,10H,1H3
(3)InChIKey: DWAQDRSOVMLGRQ-UHFFFAOYSA-N
The toxicity data is as follows:
| Organism | Test Type | Route | Reported Dose (Normalized Dose) | Effect | Source |
|---|---|---|---|---|---|
| mouse | LD50 | intraperitoneal | 370mg/kg (370mg/kg) | European Journal of Medicinal Chemistry--Chimie Therapeutique. Vol. 9, Pg. 453, 1974. |