
Journal of Medicinal Chemistry p. 9488 - 9520 (2019)
Update date:2022-08-15
Topics:
 Fyfe, Tim J.
Fyfe, Tim J.
 Kellam, Barrie
Kellam, Barrie
 Sykes, David A.
Sykes, David A.
 Capuano, Ben
Capuano, Ben
 Scammells, Peter J.
Scammells, Peter J.
 Lane, J. Robert
Lane, J. Robert
 Charlton, Steven J.
Charlton, Steven J.
 Mistry, Shailesh N.
Mistry, Shailesh N.
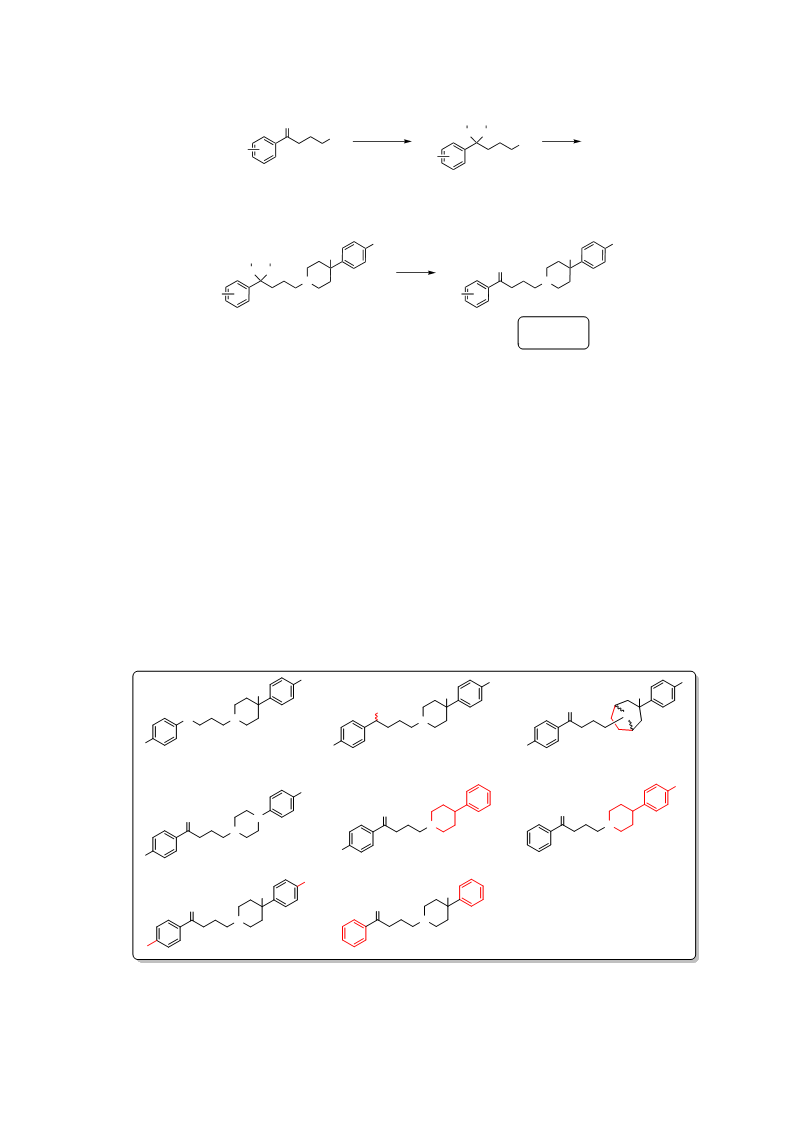
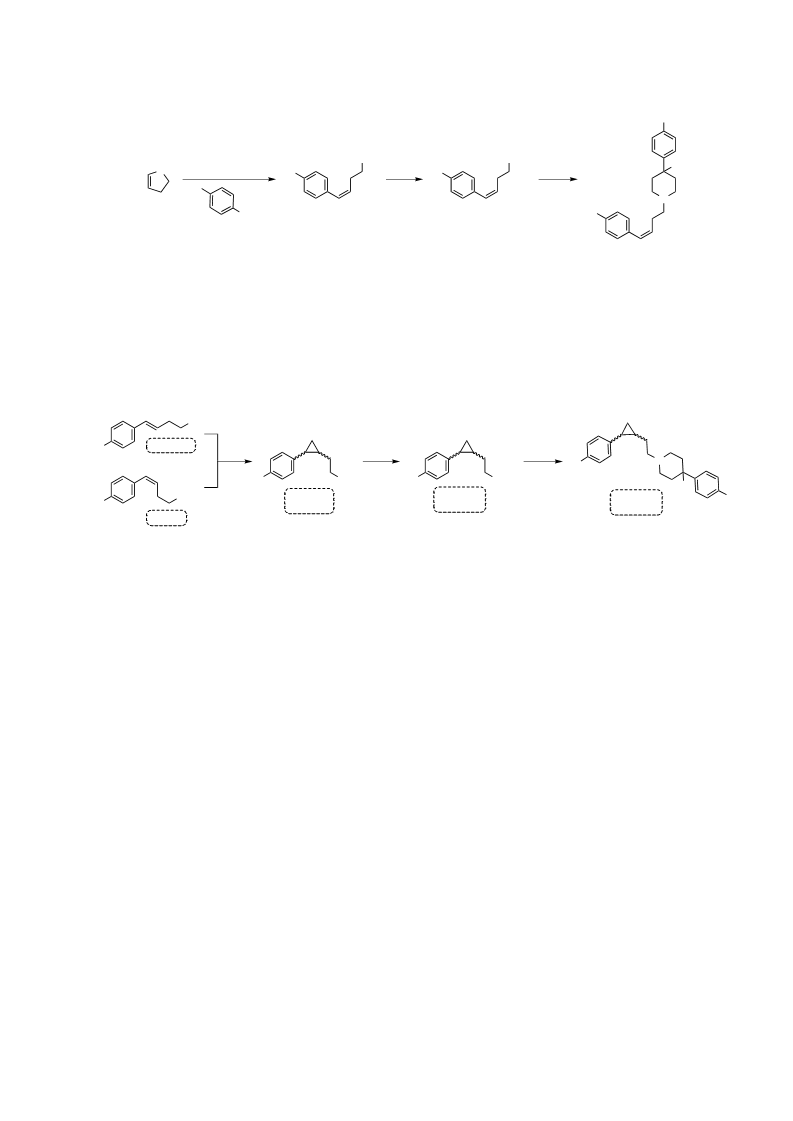
Haloperidol is a typical antipsychotic drug (APD) associated with an increased risk of extrapyramidal side effects (EPSs) and hyperprolactinemia relative to atypical APDs such as clozapine. Both drugs are dopamine D2 receptor (D2R) antagonists, with contrasting kinetic profiles. Haloperidol displays fast association/slow dissociation at the D2R, whereas clozapine exhibits relatively slow association/fast dissociation. Recently, we have provided evidence that slow dissociation from the D2R predicts hyperprolactinemia, whereas fast association predicts EPS. Unfortunately, clozapine can cause severe side effects independent of its D2R action. Our results suggest an optimal kinetic profile for D2R antagonist APDs that avoids EPS. To begin exploring this hypothesis, we conducted a structure-kinetic relationship study of haloperidol and revealed that subtle structural modifications dramatically change binding kinetic rate constants, affording compounds with a clozapine-like kinetic profile. Thus, optimization of these kinetic parameters may allow development of novel APDs based on the haloperidol scaffold with improved side-effect profiles.
View More





















Jiangxi Dongbang Pharmaceutical Co., Ltd.
Contact:+86-795-4433603, 4433388
Address:Fengxin Industrial Park, Fengxin County, Jiangxi Province, P.R.C
website:http://www.synchemie.com/
Contact:+86-574-87642758
Address:Room 901, Yinyi Bund Building, 132 Renmin Road
Jinan Hongfangde Pharmatech Co.,Ltd
Contact:86-531-88870908
Address:F Bldg. West Unit North Area of Univ. Tech. Garden Xinyu Rd. Jinan New & High Tech Industry Development Zone Shandong, China
AllyChem Co., Ltd., Dalian, China(BBChem)
Contact:+86-411-62313318/62313328
Address:No.5 of Jinbin Road, Jinzhou New District, Dalian City, Liaoning Province, P.R.China
Shenyang Mole pharmaceutical Technology Development Co.,Ltd
Contact:+86-24-31204918/13889278616
Address:No.44, wanliutang road, shenhe District of Shenyang
Doi:10.1021/ja054085l
(2005)Doi:10.1021/ol052128x
(2005)Doi:10.1021/ja003124k
(2001)Doi:10.1007/BF00568269
()Doi:10.1016/S0040-4039(00)88321-5
(1983)Doi:10.1002/recl.19831020505
(1983)