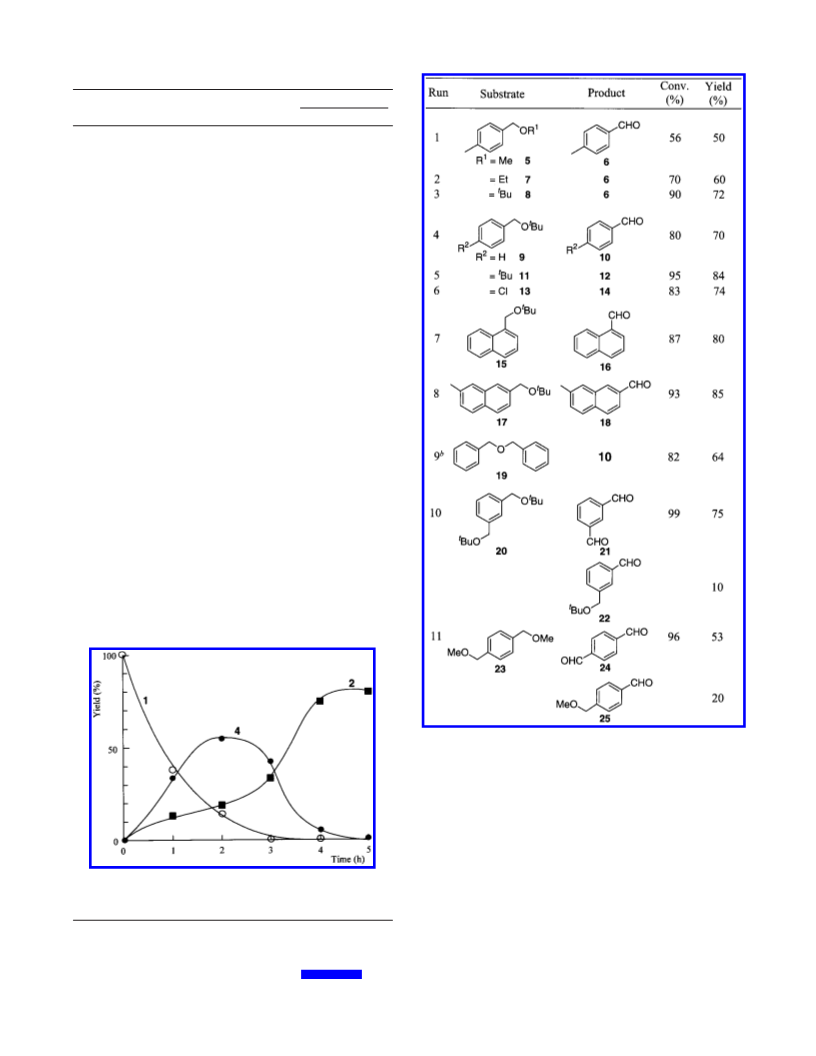
Oxygenation Using Nitric Oxide
J . Org. Chem., Vol. 64, No. 13, 1999 4679
NMR (CDCl3, 67.5 MHz) δ 192.2, 136.8, 134.6, 133.7, 132.7,
131.2, 128.6, 128.3, 127.7, 121.8, 21.5.
adamantyl cation with NO was examined. Under a NO
atmosphere, 1-bromoadamantane (31) was allowed to
react with tributyltinhydride and AIBN in MeCN/AcOH,
producing N-1-adamantylacetoamide (32) (56%) and ada-
mantane (33) (44%) (eq 6). This indicates that the
3-ter t-Bu toxym eth ylben zaldeh yde (22): 1H NMR (CDCl3,
270 MHz) δ 10.6 (s, 1 H), 8.46-7.45 (m, 4 H), 4.51 (s, 2 H),
1.31 (s, 9 H); 13C NMR (CDCl3, 67.5 MHz) δ 192.2, 136.3, 134.9,
133.2, 128.8, 128.4, 128.3, 73.6, 63.2, 27.5.
4-Meth oxym eth ylben za ld eh yd e (25): 1H NMR (CDCl3,
270 MHz) δ 8.79 (s, 1 H), 6.66 (d, J ) 7.8 Hz, 2 H), 6.29 (d, J
) 7.8 Hz, 2 H), 3.33 (s, 2 H), 2.23 (s, 3 H); 13C NMR (CDCl3,
67.5 MHz) δ 191.9, 145.3, 135.6, 129.8, 127.6, 73.8, 58.4.
1,1′-Oxod iisoch r om a n e (27): 1H NMR (CDCl3, 270 MHz)
δ 7.18 (m, 8 H), 6.11 (s, 2 H), 4.34 (dt, J ) 6.2 and 11.3 Hz, 2
H), 4.11-4.04 (m, 2 H), 3.11 (dt, J ) 6.2 and 11.3 Hz, 2 H),
2.68-2.61 (m, 2 H); 13C NMR (CDCl3, 67.5 MHz) δ 134.1, 133.8,
128.4, 128.0, 127.5, 126.3, 92.7, 58.1, 27.9. Anal. Calcd. for
C18H18O3: C, 76.48; H, 6.42. Found: C, 76.43, H, 6.38. The
compound 27 (C18H18O3; Mr ) 282.34) crystallized in the
orthorhombic space group C2/c with cell dimensions of a )
23.355(6) Å, b ) 4.346(3) Å, and c ) 15.926 (5) Å; â ) 117.98-
(2)°, V ) 1427(1) Å3 and an occupation of Z ) 4 in cell unit.
Data were collected at 24.0 ( 1 °C on a AFC7R Rigaku
diffractometer (Mo KR radiation), to a maximum 2q ) 55.0°,
giving 1649 unique reflections; the strucutre was solved by
direct methods (SIR88) and refined within full matrix least
squares, yielding R ) 0.059, Rw ) 0.113 (GOF ) 1.76) for 1495
unique reflections with I > 1.10σ(I).
resulting adamantyl radical C generated from 31 by the
action of Bu3SnH/AIBN was converted into an adamantyl
cation D under the influence of NO, to give amide 32
through the reaction with MeCN, which is known as the
Ritter reaction.7
P r oced u r e for th e Rea ction of Isoch r om a n (26) to
1-Eth oxyisoch r om a n e (29). To a solution of 26 (1 mmol) in
acetonitrile (5 cm3) in a three-necked flask was added NHPI
(0.1 mmol). The flask was cooled to -78 °C to freeze the solvent
and degassed in vacuo and filled with Ar gas. Then the frozen
solvent was melted at room temperature and refrozen to
reiterate the evacuation-Ar purge procedure. The series of
operations was repeated three times, and then NO was added
to the reaction vessel. After the reaction mixture was allowed
to react under an atmospheric pressure of NO at 60 °C for 5
h, EtOH (5 cm3) was added, then the reaction mixture was
stirred at room temperature for 0.5 h. The products were
purified and identified by the same method as previously
described.
In conclusion, we have developed a new type of
oxygenation through a carbocation as a transient inter-
mediate, using an NO/NHPI system. This method pro-
vides a facile synthetic route to aldehydes or acetals,
which so far have been difficult to obtain. The most
striking characteristic of the present reaction is that it
did not produce a carboxylic acid since the reaction is
based on the capture of water with carbocation.
Exp er im en ta l Section
Gen er a l P r oced u r es. The starting materials such as 1,
5, 7, 19, 23, 26, and 31, and catalysts used were commercially
available and used without further purification. The com-
pounds 8, 9, 11, 13, 15, 17, and 20 were synthesized by the
reaction of the coresponding alcohols with sodium tert-butoxide
in anhydride THF at room temperature and were isolated by
column chromatography on silica gel (hexane/ethyl acetate )
10:1). GC analysis was performed with a flame ionization
1
1-Eth oxyisoch r om a n e (29): H NMR (CDCl3, 270 MHz)
δ 7.20-7.07 (m, 4 H), 5.54 (s, 1H), 4.19-3.63 (m, 4 H), 3.05-
2.92 (m, 1 H), 2.61-2.56 (m, 1 H), 1.28 (t, J ) 7.0 Hz, 3 H);
13C NMR (CDCl3, 67.5 MHz) δ 134.3, 133.9, 128.3, 127.9, 127.3,
126.2, 96.4, 63.3, 57.7, 27.9, 15.2.
1-E t h oxyp h t h a la n e (30): 1H NMR (CDCl3, 270 MHz) δ
7.29-7.10 (m, 4 H), 6.13 (s, 1H), 5.11-4.87 (m, 4 H), 3.70-
3.49 (m, 2 H),1.14 (t, J ) 7.0 Hz, 3 H); 13C NMR (CDCl3, 67.5
MHz) δ 139.7, 137.5, 128.8, 127.4, 122.7, 120.7, 106.5, 71.9,
62.7, 15.2.
1
detector using a 0.2 mm × 25 m capillary column (OV-1). H
and 13C NMR were measured at 270 and 67.5 MHz, respec-
tively, in CDCl3 with Me4Si as the internal standard. The
yields of all products except for 27 were estimated from the
peak areas based on the internal standard technique. The yield
of 27 is isolated yield.
P r oced u r e for th e Rea ction of 1-Br om oa d a m a n ta n e
(31) w ith NO in th e P r esen ce of Bu 3Sn H a n d AIBN. To a
solution of 31 (1 mmol) in a mixed solvent of acetonitrile and
acetic acid (5:1 cm3) in a three-necked flask was added Bu3-
SnH (1.2 mmol) and AIBN (0.1 mmol). The flask was cooled
to -78 °C to freeze the solvent, degassed in vacuo, and filled
with Ar gas. Then, the frozen solvent was melted at room
temperature and refrozen to reiterate the evacuation-Ar
purge procedure. The series of operations was repeated three
times, and then NO was added to the reaction vessel. The
reaction mixture was allowed to react under an atmospheric
pressure of NO at 75 °C for 20 h. The products were purified
by the same method as previously described. Products 32 and
33 were identified by comparison of the isolated products with
authentic samples.
A Typ ica l P r oced u r e for th e Rea ction of 1. To a solution
of 1 (1 mmol) in acetonitrile (5 cm3) in a three-necked flask
was added NHPI (0.1 mmol). The flask was cooled to -78 °C
to freeze the solvent and degassed in vacuo and filled with Ar
gas. Then the frozen solvent was melted at room temperature
and refrozen to reiterate the evacuation-Ar purge procedure.
The series of operations was repeated three times, and then
NO was added to the reaction vessel. The reaction mixture
was allowed to react under an atmospheric pressure of NO at
60 °C for 5 h. After solvent was removed in vacuo, the products
were isolated by column chromatography on silica gel (chlo-
roform) and characterized by 1H and 13C NMR, respectively.
Products 2, 6, 10, 12, 14, 16, 21, and 24 were identified by
comparing of the isolated products with authentic samples.
1-Hyd r oxyp h th a la n e (4): 1H NMR (CDCl3, 270 MHz) δ
10.2 (s, 1 H), 7.86-7.23 (m, 8 H), 6.41 (s, 1 H), 5.28 (m, 2 H),
5.13-5.09 (m, 3 H); 13C NMR (CDCl3, 67.5 MHz) δ 192.5, 140.4,
139.9, 133.7, 131.8, 129.2, 128.8, 127.9, 127.7, 123.1, 123.0,
121.0, 120.9, 106.6, 72.2, 63.4.
Ack n ow led gm en t. This work was partially sup-
ported by Research for the Future program J SPS.
Su p p or tin g In for m a tion Ava ila ble: Copies of 1H and 13C
NMR spectra for the compounds 2, 4, 16, 18, 21, 22, 24, 25,
27, 29, 30, and 32 and X-ray of C18H18O3. This information is
7-Meth yl-2-n a p h th yla ld eh yd e (18): 1H NMR (CDCl3, 270
MHz) δ 9.99 (s, 1 H), 8.07-7.35 (m, 6 H), 2.42 (s, 3 H); 13C
J O982406O

 Eikawa, Masahiro
Eikawa, Masahiro
 Sakaguchi, Satoshi
Sakaguchi, Satoshi
 Ishii, Yasutaka
Ishii, Yasutaka