Journal of the American Chemical Society
Article
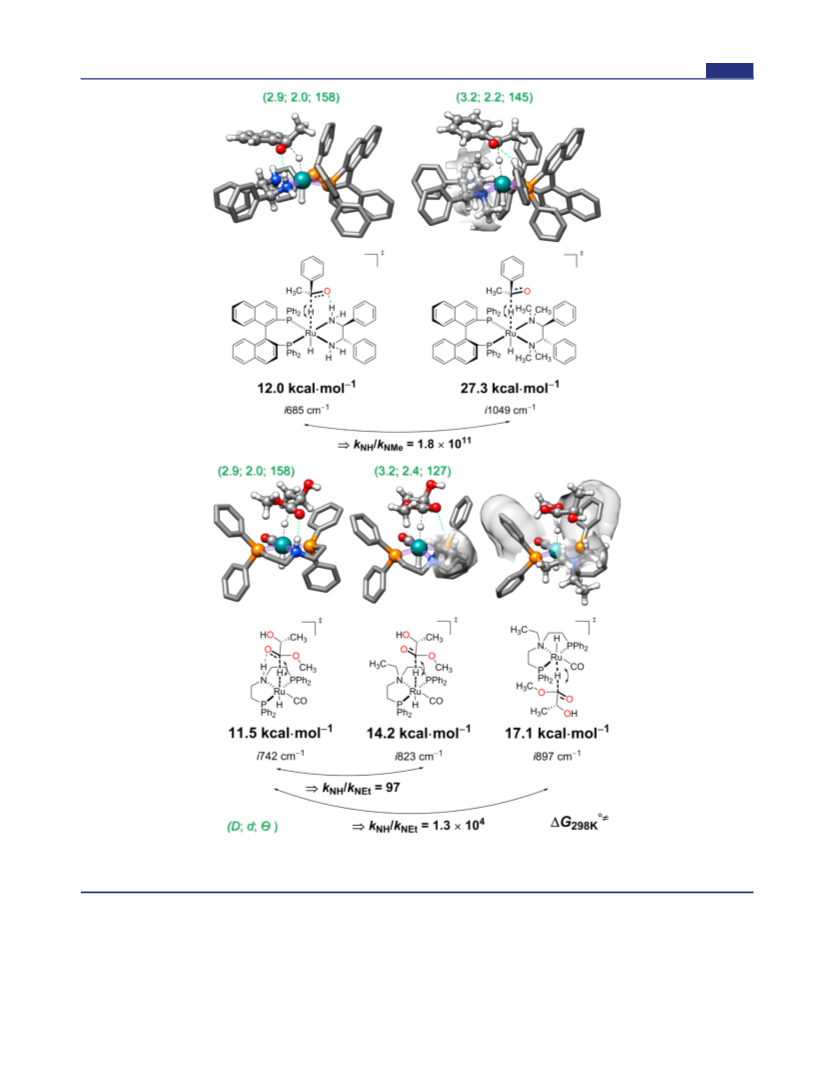
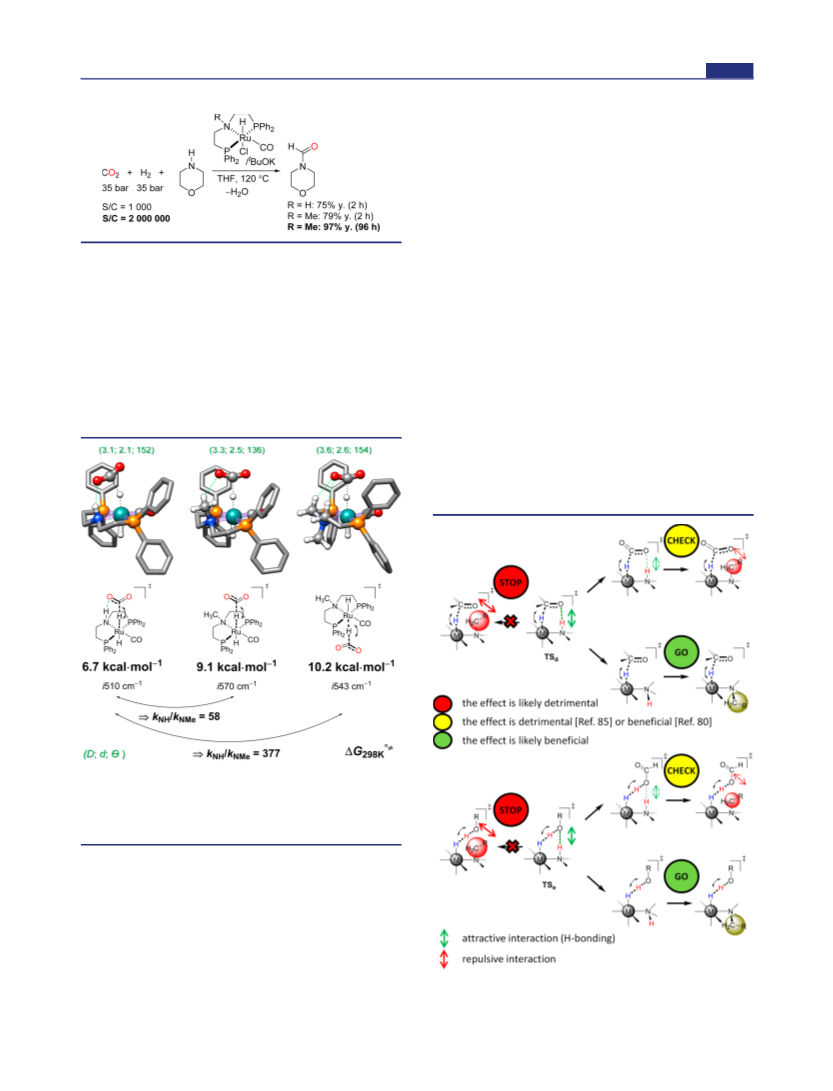
but rather from the energetic span defined from the turnover-
determining transition state (TDTS), the turnover-determining
intermediate (TDI), and sometimes the reaction driving force, ref
38. In catalytic cycles of hydrogenation with Noyori’s and Ru-
MACHO catalysts, hydride transfer is found as the TDTS, refs 35a, 37.
We assume that alkylation of the NH functionality will not
dramatically change the relative energy of the catalyst’s TDI; therefore,
the relative rate of hydride transfer could be approximated to be the
relative rate of the catalytic reaction.
(46) Note that for this complex, in contrast to the fully methylated
Noyori catalyst, the two hydrides are inequivalent and the substrate
may approach both hydrides within the outer-sphere. The barrier for
the outer-sphere hydride transfer anti to N(Alk) is computationally
prohibitive. The origin of this is an additional substrate/ligand Ph-
groups repulsion within the corresponding CSC.
(47) A possible reason for this is much less repulsion originating
within the CSC of ethylated Ru-MACHO catalyst and anti-methyl
(R)-lactate with respect to the fully methylated Noyori catalyst and
acetophenone: indeed upon ethylation, the relative position of the
anti-methyl (R)-lactate is almost unaffected (and the corresponding
TS is stabilized by a C−H···O HBI between one C−H group of the
ethyl substituent and the oxygen atom of the substrate); however,
acetophenone is more significantly displaced within the fully
methylated Noyori catalyst with respect to the initial geometry
observed in the TS for the nonmethylated catalyst.
(48) (a) Lundgren, R. J.; Stradiotto, M. Chem. - Eur. J. 2008, 14,
10388. (b) Lundgren, R. J.; Rankin, M. A.; McDonald, R.; Schatte, G.;
Stradiotto, M. Angew. Chem. 2007, 119, 4816.
(49) Leong, C. G.; Akotsi, O. M.; Ferguson, M. J.; Bergens, S. H.
Chem. Commun. 2003, 750.
(50) Abdur-Rashid, K.; Faatz, M.; Lough, A. J.; Morris, R. H. J. Am.
Chem. Soc. 2001, 123, 7473.
(51) (a) Dub, P. A.; Scott, B. L.; Gordon, J. C. Organometallics 2015,
34, 4464. (b) Xie, J.-H.; Liu, X.-Y.; Xie, J.-B.; Wang, L.-X.; Zhou, Q.-L.
Angew. Chem., Int. Ed. 2011, 50, 7329.
(52) Abdur-Rashid, K.; Clapham, S. E.; Hadzovic, A.; Harvey, J. N.;
Lough, A. J.; Morris, R. H. J. Am. Chem. Soc. 2002, 124, 15104.
(53) Blaser, H.-U., Federsel, H.-J., Eds. Asymmetric Catalysis On
Industrial Scale: Challenges, Approaches, and Solutions, 2nd ed.; Wiley-
VCH Verlag GmbH & Co. KGaA: 2010.
(63) (a) de Koning, P. D.; Jackson, M.; Lennon, I. C. Org. Process Res.
Dev. 2006, 10, 1054. (b) Ohkuma, T.; Koizumi, M.; Doucet, H.; Pham,
T.; Kozawa, M.; Murata, K.; Katayama, E.; Yokozawa, T.; Ikariya, T.;
Noyori, R. J. Am. Chem. Soc. 1998, 120, 13529.
(64) (a) Gladiali, S.; Alberico, E. Chem. Soc. Rev. 2006, 35, 226.
(b) Bar, R.; Sasson, Y. Tetrahedron Lett. 1981, 22, 1709. (c) Heydrich,
G.; Gralla, G.; Rauls, M.; Schmidt-Leithoff, J.; Ebel, K.; Krause, W.;
Oehlenschlaeger, S.; Jaekel, C.; Friedrich, M.; Bergner, E. J.; Kashani-
Shirazi, N.; Paciello, R. Patent WO2009068444A2; BASF SE,
Germany, 2009.
(65) John, J. M.; Takebayashi, S.; Dabral, N.; Miskolzie, M.; Bergens,
S. H. J. Am. Chem. Soc. 2013, 135, 8578.
(66) Kuze, N.; Ishikawa, A.; Kono, M.; Kobayashi, T.; Fuchisawa, N.;
Tsuji, T.; Takeuchi, H. J. Phys. Chem. A 2015, 119, 1774.
(67) (a) Ramanjaneyulu, B. T.; Mahesh, S.; Vijaya Anand, R. Org.
Lett. 2015, 17, 6. (b) Duncton, M. A. J.; Singh, R. Org. Lett. 2013, 15,
4284. (c) Tran, G.; Meier, R.; Harris, L.; Browne, D. L.; Ley, S. V. J.
Org. Chem. 2012, 77, 11071. (d) Zhang, Y.; Zhang, X.; Xie, Y.; Huang,
L. Patent CN102603646A; Zhejiang University: P. R. China, 2012.
(e) Fritz, S. P.; West, T. H.; McGarrigle, E. M.; Aggarwal, V. K. Org.
Lett. 2012, 14, 6370. (f) Borkin, D. A.; Landge, S. M.; Toeroek, B.
Chirality 2011, 23, 612. (g) Prakash, G. K. S.; Paknia, F.; Mathew, T.;
Mloston, G.; Joschek, J. P.; Olah, G. A. Org. Lett. 2011, 13, 4128.
(h) Nie, J.; Li, X.-J.; Zheng, D.-H.; Zhang, F.-G.; Cui, S.; Ma, J.-A. J.
Fluorine Chem. 2011, 132, 468. (i) Wen, L.; Shen, Q.; Lu, L. Org. Lett.
2010, 12, 4655. (j) Mimura, H.; Kawada, K.; Yamashita, T.; Sakamoto,
T.; Kikugawa, Y. J. Fluorine Chem. 2010, 131, 477. (k) Cho, E. J.;
Senecal, T. D.; Kinzel, T.; Zhang, Y.; Watson, D. A.; Buchwald, S. L.
Science 2010, 328, 1679. (l) Gulevich, A. V.; Shpilevaya, I. V.;
Nenajdenko, V. G. Eur. J. Org. Chem. 2009, 2009, 3801. (m) Gagosz,
F.; Zard, Z. S. Org. Synth. 2007, 84, 32.
(68) (a) Wang, J.; San
Sorochinsky, A. E.; Fustero, S.; Soloshonok, V. A.; Liu, H. Chem. Rev.
2014, 114, 2432. (b) Torok, B.; Sood, A.; Bag, S.; Kulkarni, A.; Borkin,
́
chez-Rosello, M.; Acena, J. L.; del Pozo, C.;
́
̃
̈
̈
D.; Lawler, E.; Dasgupta, S.; Landge, S.; Abid, M.; Zhou, W.; Foster,
M.; LeVine, H.; Torok, M. ChemMedChem 2012, 7, 910. (c) Filler, R.;
̈
̈
Saha, R. Future Med. Chem. 2009, 1, 1370. (d) Ojima, I. Fluorine in
Medicinal Chemistry and Chemical Biology; John Wiley & Sons, Ltd:
2009. (e) Quirmbach, M.; Steiner, H. Chim. Oggi 2009, 27, 23.
(f) Hagmann, W. K. J. Med. Chem. 2008, 51, 4359. (g) Purser, S.;
Moore, P. R.; Swallow, S.; Gouverneur, V. Chem. Soc. Rev. 2008, 37,
(54) Dub, P. A.; Gordon, J. C. Patent WO2015191505A1; Los
Alamos National Security, LLC, USA, 2015.
(55) (a) Blomenkemper, M.; Schroeder, H.; Pape, T.; Hahn, F. E.
Inorg. Chim. Acta 2011, 366, 76. (b) Palmer, D. C.; Taylor, E. C. J. Org.
Chem. 1986, 51, 846.
320. (h) Muller, K.; Faeh, C.; Diederich, F. Science 2007, 317, 1881.
̈
(i) Yale, H. L. J. Med. Pharm. Chem. 1959, 1, 121.
(69) (a) Fluorine and the Environment: Agrochemicals, Archaeology,
Green Chemistry & Water; Tressaud, A., Ed.; Elsevier: 2006. (b) Coe,
B. J.; Glenwright, S. J. Coord. Chem. Rev. 2000, 203, 5.
(70) (a) Tomashenko, O. A.; Grushin, V. V. Chem. Rev. 2011, 111,
4475. (b) Yamada, S.; Morita, C. J. Am. Chem. Soc. 2002, 124, 8184.
(71) Chen, X.; Jia, W.; Guo, R.; Graham, T. W.; Gullons, M. A.;
Abdur-Rashid, K. Dalton Trans. 2009, 1407.
(56) Dub, P. A.; Scott, B. L.; Gordon, J. C. Dalton Trans. 2016, 45,
1560.
(57) (a) Eschweiler, W. Ber. Dtsch. Chem. Ges. 1905, 38, 880.
(b) Clarke, H. T.; Gillespie, H. B.; Weisshaus, S. Z. J. Am. Chem. Soc.
1933, 55, 4571.
(58) We reported the identity of complex Ir1 in a previous
contribution (see ref 51a), but the details of its synthesis and
characterization, including its X-ray structure, are reported for the first
time in this work.
́
(72) Hayes, J. M.; Deydier, E.; Ujaque, G.; Lledos, A.; Malacea-
Kabbara, R.; Manoury, E.; Vincendeau, S.; Poli, R. ACS Catal. 2015, 5,
4368.
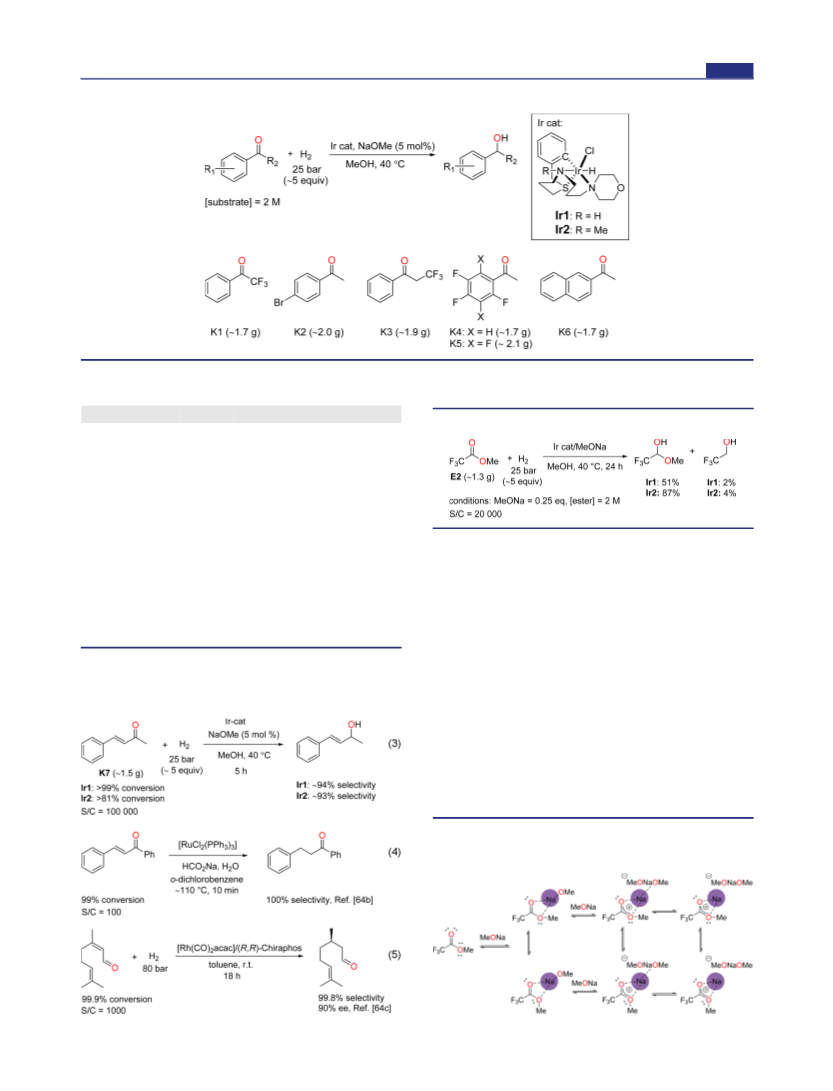
(73) Although anti-E2 is computed to be 7.45 kcal·mol−1 more stable
then syn-E2 (ΔG°298K), both conformers were considered in the
computational analysis.
(59) (a) Xie, M.; Zhu, G.; Hu, Y.; Gu, H. J. Phys. Chem. C 2011, 115,
20596. (b) Capparelli, A. L.; Maranon, J.; Sorarrain, O. M.; Filgueria,
̃
(74) Fukui, K. Acc. Chem. Res. 1981, 14, 363.
R. R. J. Mol. Struct. 1974, 23, 145.
(75) Further analysis of the catalytic cycle for this catalyst and its
methylated analog reveal that hydride transfer (and not H−H bond
cleavage) is the TDTS; see the next section.
(60) The reaction also easily proceeds in other alcoholic solvents
such as iPrOH, EtOH for both catalysts. However, the reaction is not a
i
net hydrogenation (e.g., in PrOH), since transfer hydrogenation also
(76) If X = Na, an additional Na···O interaction is present.
(77) Hartmann, R.; Chen, P. Angew. Chem., Int. Ed. 2001, 40, 3581.
(78) The activation barrier for the MEHT within the IrNNa complex
is computed to be 2.4 kcal·mol−1 uphill versus the IrNH complex. In
fact, the corresponding TS is stabilized by a N−Na···O interaction
(there is no N−H···O interaction within the IrNH complex), but this
effect does not sufficiently compensate for the unfavorable
thermodynamics of the reaction IrNH + MeONa = IrNNa +
MeOH (7.0 kcal·mol−1 on free energy scale). However, this
partially takes place as verified by separate experiments.
(61) Barrio, P.; Castarlenas, R.; Esteruelas, M. A.; Lledos
́
, A.;
Maseras, F.; Onate, E.; Tomas, J. Organometallics 2001, 20, 442.
̀
̃
(62) It is also interesting to note that both Ir1 and Ir2 seem to be
much more efficient catalysts for the hydrogenation of K7 than
Noyori’s catalyst, although in nonasymmetric fashion (see ref 63). For
example, 1.46 g of K7 is quantitatively hydrogenated with only 5.4 ×
10−2 mg of Ir1.
O
J. Am. Chem. Soc. XXXX, XXX, XXX−XXX

 Dub, Pavel A.
Dub, Pavel A.
 Scott, Brian L.
Scott, Brian L.
 Gordon, John C.
Gordon, John C.