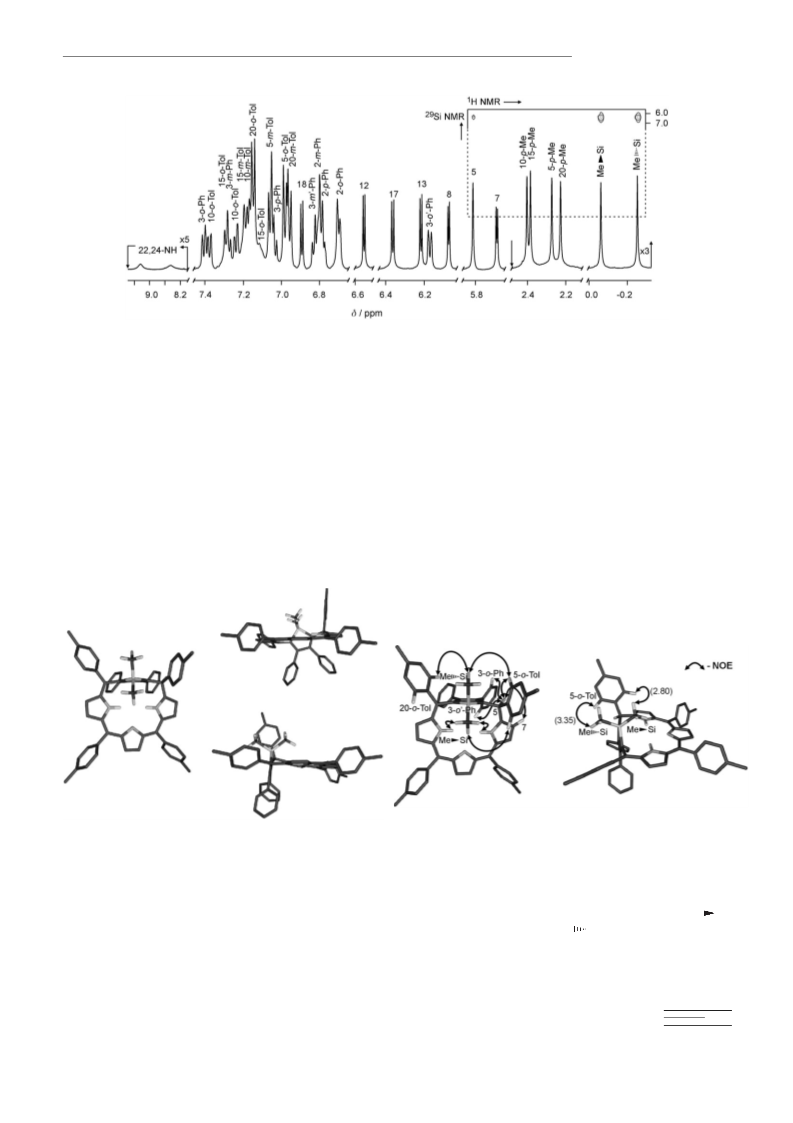
´
L. Latos-Graz˙ynski et al.
(m, 1H; 3-m-Ph), 6.42 (d, 1H; 12), 6.36 (d, 1H; 17), 6.04 (m, 1H; 8), 5.87
(d, 1H; 18), 5.56 (m, 1H; 7), 5.48 (s, 1H; 10), 5.47 (d, 1H; 3-o-Ph), 5.14
(s, 1H; 20), 4.67 (s, 1H; 5), 2.42 (s, 3H; 15-p-Me), 2.30 (s, 3H; 10-p-Me),
2.26 (s, 3H; 5-p-Me), 2.18 (s, 3H; 20-p-Me), 0.01 (s, 3H; Me Si),
ꢀ1.16 ppm (s, 3H; Me Si); 13C NMR (CDCl3, 230 K): d=154.7 (2),
151.5 (3), 146.5 (4), 141.5 (1), 140.1 (15), 139.4 (C1–20-Tol), 138.6 (C4–15-
Tol), 138.3 (C1–2-Ph), 137.9 (C1–10-Tol), 137.6 (C1–5-Tol), 137.4 (C1–3-
Ph), 136.4 (C4–5-Tol), 136.3 (C4–10-Tol, C4–20-Tol), 135.0 (C1–15-Tol),
134.5 (13), 133.9 (16), 130.9 (6), 130.8 (2-m-Ph), 130.6 (9), 129.8 (3-o-Ph),
129.7 (10-m-Tol), 129.3 (10-o-Tol, 19), 129.0 (5-m-Tol), 128.9 (20-m-Tol),
128.8 (20-o-Tol), 128.7 (5-o-Tol), 128.6 (2-o-Ph, 3-o-Ph), 128.5 (3-m-
Ph),128.2 (14, 15-m-Tol), 128.0 (11), 127.9 (2-o-Ph, 15-o-Tol), 127.8 (2-m-
Ph), 127.4 (3-m-Ph), 126.9 (2-p-Ph), 126.4 (2-p-Ph), 125.7 (17), 123.2 (12),
113.4 (18), 106.7 (8), 104.2 (7), 47.0 (20), 45.4 (10), 45.3 (5), 21.3 (15-p-
Me), 21.0 (10-p-Me; 20.9 (5-p-Me), 20.8 (20-p-Me), ꢀ2.9 (Me Si),
ꢀ3.9 ppm (Me Si); 29Si NMR (CDCl3, 298 K): d=5.79 ppm; HRMS
(ESI): m/z calcd for C62H56N3Si+: 870.4243, found: 870.4183 [M+H]+;
13C NMR (CD2Cl2, 250 K): d=165.8 (11), 150.6 (14), 148.6 (3), 146.3 (9),
142.7 (6), 142.3 (4), 141.3 (2), 138.5 (21), 138.3 (C4–15-Tol), 137.5 (C4–21-
Tol), 137.0 (C4–5-Tol), 136.9 (C4–10-Tol), 136.3 (C1–10-Tol), 135.9 (C1–3-
Ph), 135.6 (19), 135.4 (C1–2-Ph), 135.3 (C1–15-Tol), 135.0 (C1–5-Tol),
134.6 (13), 132.7 (16), 132.3 (15), 132.3 (10-o-Tol), 132.0 (10-o-Tol, 15-o-
Tol), 131.3 (15-o-Tol), 130.7 (C1–21-Tol), 130.3 (8), 128.5 (21-m-Tol),
129.2 (5-o-Tol), 129.1 (3-o-Ph, 5-m-Tol), 129.0 (2-o-Ph), 128.84, 128.81
(10-m-Tol), 128.4 (15-m-Tol), 128.3 (2-m-Ph, 15-m-Tol), 128.1 (3-m-Ph),
127.6 (21-o-Tol), 127.4 (12), 127.1 (2-p-Ph, 3-p-Ph), 126.9 (7), 120.3 (17),
116.5 (5), 112.1 (18), 108.7 (10), 54.5 (1), 21.4 (15-p-Me), 21.3 (10-p-Me),
21.2 (5-p-Me), 21.1 ppm (21-p-Me); HRMS (EI): m/z calcd for C60H47N3:
809.3770, found: 809.3771 [M]+; UV/Vis (CH2Cl2, 298 K): lmax
338 (5.90), 386 (5.99), 624 (5.58) 673 nm (5.74 mꢀ1 cmꢀ1).
A
Silver(III) carbacorrole (8-Ag): Compound iso-8 (16 mg, 0.02 mmol) was
R
dissolved in freshly distilled dichloromethane (20 mL), than excess
AgBF4 was added. The solution turned immediately from sea-green to
red. The resulting mixture was stirred for 5 min, and then TEA (excess)
was added. The colour changed to green. The mixture was directly
moved to the top of column chromatography on a silica gel (mesh 70–
230). The first green band moving with dichloromethane was collected
and evaporated to dryness. The residue contains practically pure 8-Ag
complex. Yield: 16.4 mg (90%), 1H NMR (CDCl3, 230 K): d=9.19, 7.89,
5.91 (br, 3 s, 5H; 2-Ph, 210 K), 8.95 (d, 1H; 7), 8.72 (d, 1H; 8), 8.63 (d,
1H; 13), 8.54 (d, 1H; 18), 8.51 (d, 1H; 17), 8.43 (d, 1H; 15-o-Tol), 8.40
(d, 1H; 12), 8.04 (d, 2H; 5-o-Tol, 10-o-Tol), 8.00 (d, 1H; 10-o-Tol), 7.87
(d, 1H; 5-o-Tol), 7.72 (d, 1H; 3-o-Ph), 7.61 (d, 1H; 15-m-Tol), 7.52 (d,
2H; 10-m-Tol, 15-o-Tol), 7.50 (d, 1H; 10-m-Tol), 7.38 (m, 2H; 5-m-Tol,
15-m-Tol), 7.16 (t, 1H; 3-m-Ph), 6.96 (m, 1H; 3-p-Ph), 6.87 (d, 1H; 5-m-
Tol), 6.55 (t, 1H; 3-m-Ph), 5.76 (d, 1H; 3-o-Ph), 5.75 (d, 2H; 21-m-Tol),
4.46 (d, 2H; 21-o-Tol), 2.64 (s, 3H; 10-p-Me), 2.61 (s, 3H; 15-p-Me), 2.39
(s, 3H; 5-p-Me), 1.46 ppm (s, 3H; 21-p-Me); 13C NMR (CDCl3, 230 K):
d=152.7 (1), 152.1, 135.5 (16, 19), 145.0, 140.3 (6, 9), 144.3 (3), 142.2,
137.8 (11. 14), 141.5 (4), 139.1 (C1–10-Tol), 138.0 (C1–15-Tol), 137.3 (C4–
21-Tol), 137.1 (C1–3-Ph), 137.0 (C4–15-Tol), 136.9 (C4–10-Tol), 136.8 (C4–
5-Tol), 136.7 (C1–5-Tol), 136.5, 135.8 (5-o-Tol), 136.3 (2), 134.8 (10-o-Tol,
15-o-Tol), 134.7 (15-o-Tol), 134.6 (10-o-Tol), 132.8 (2-Ph), 132.2 (3-o-Ph),
130.0 (3-o-Ph, 13), 129.5 (8, C1–21-Tol), 128.8 (17), 128.7 (7), 128.4, 127.5
(5-m-Tol), 128.3 (15-m-Tol), 128.2 (21-m-Tol), 128.0 (5, 10-m-Tol), 127.9
(10-m-Tol), 127.1 (3-m-Ph, 12), 127.0 (3-m-Ph), 125.6 (3-p-Ph), 123.5 (21-
o-Tol), 118.5 (15), 117.2 (18), 116.3 (10), 88.5 (21), 21.8 (10-p-Me), 21.7
(15-p-Me), 21.4 (5-p-Me), 20.6 ppm (21-p-Me); HRMS (ESI): m/z calcd
for C60H44N3Ag+: 913.2586, found: 913.2393 [M]+; UV/Vis (CH2Cl2,
UV/Vis (CH2Cl2, 298 K): lmax
A
(3.93 mꢀ1 cmꢀ1).
21-Silaphlorin (7): See the procedure for 6. 1H NMR (CD2Cl2, 230 K):
d=9.20, 8.41 (br, 2 s, 2H; 22,24-NH), 7.42 (d, 1H; 3-o-Ph), 7.39, 7.25 (2d,
2H; 10-o-Tol), 7.31, 7.12 (2d, 2H; 15-o-Tol), 7.29 (m, 1H; 3-m-Ph), 7.20
(d, 2H; 15-m-Tol), 7.18 (d, 2H; 10-m-Tol), 7.16 (2d, 2H; 20-o-Tol), 7.08
(d, 2H; 5-m-Tol), 7.06 (m, 1H; 3-p-Ph), 6.99 (d, 2H; 5-o-Tol), 6.97 (d,
2H; 20-m-Tol), 6.91 (d, 1H; 18), 6.84 (m, 1H; 3-m’-Ph), 6.81 (d, 2H; 2-
m-Ph), 6.79 (m, 1H; 2-p-Ph), 6.71 (d, 2H; 2-o-Ph), 6.57 (d, 1H; 12), 6.37
(d, 1H; 17), 6.23 (d, 1H; 13), 6.18 (d, 1H; 3-o’-Ph), 6.09 (d, 1H; 8), 5.81
(s, 1H; 5), 5.69 (d, 1H; 7), 2.38 (s, 3H; 10-p-Me), 2.37 (s, 3H; 15-p-Me),
2.26 (s, 3H; 5-p-Me), 2.21 (s, 3H; 20-p-Me), ꢀ0.05 (s, 3H; Me Si),
ꢀ0.24 ppm (s, 3H; Me Si); 13C NMR (CD2Cl2, 230 K): d=167.6 (14),
159.8 (2), 154.3 (3), 151.5 (11), 145.5, 142.2 (16, 19), 142.4 (1), 140.5 (C1–
5-Tol), 139.4 (6), 138.2 (C1–2-Ph), 137.9 (C1–15-Tol), 137.9 (C1–3-Ph),
137.7 (C4–10-Tol), 136.55 (C4–5-Tol), 136.5 (C4–15-Tol, C4–20-Tol), 135.9
(12), 135.3 (4), 135.2 (C1–20-Tol), 133.2 (C1–10-Tol), 132.6 (9), 132.1 (10,
15-o-Tol), 131.5 (10-o-Tol), 129.1 (5-m-Tol), 128.8 (20-m-Tol), 128.7 (3-o-
Ph, 20-o-Tol), 128.6 (13, 15-m-Tol), 128.3 (2-o-Ph), 128.2 (3-o’-Ph), 128.1
(17), 128.0 (5-o-Tol), 127.9 (3-m-Ph), 127.8 (10-m-Tol), 127.6 (3-m’-Ph),
126.8 (2-p-Ph), 126.6 (3-p-Ph), 126.5 (18), 126.4 (2-m-Ph), 123.1 (8), 121.9
(20), 110.7 (7), 107.8 (15), 46.6 (5), 21.3 (10-p-Me), 21.2 (15-p-Me), 21.1
(20-p-Me; 21.0 (5-p-Me), ꢀ2.6 (Me Si), ꢀ3.2 ppm (Me Si); 29Si NMR
(CD2Cl2, 298 K): d=6.52; HRMS (ESI): m/z calcd for C62H54N3Si+:
868.4087, found: 868.4104 [M+H]+; UV/Vis (CH2Cl2, 298 K):
lmax
A
298 K): lmax
(5.13 mꢀ1 cmꢀ1).
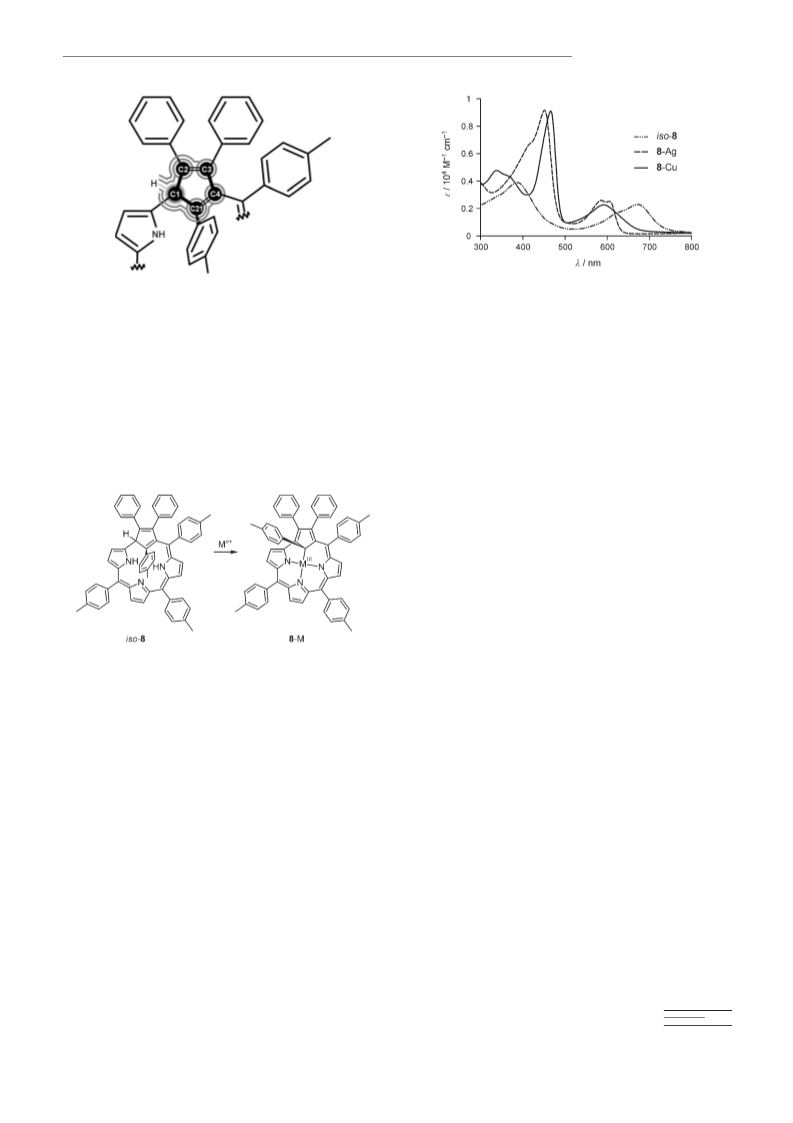
Copper(III) carbacorrole (8-Cu): Compound iso-8 (16 mg, 0.02 mmol)
was dissolved in freshly distilled tetrahydrofuran (20 mL), then excess
Cu(CH3COO2)2·H2O was added. The mixture was heated to reflux for
A
iso-Carbacorrole (iso-8): A 500 mL round-bottomed flask equipped with
a stirring bar and nitrogen inlet, was charged with freshly distilled di-
chloromethane (450 mL). 2,5-Bis(p-tolylhydroxymethyl)-3,4-diphenyl-1,1-
dimethylsilole (5, 0.5 g, 1 mmol), pyrrole (0.21 mL, 3 mmol), p-tolylalde-
hyde (0.24 mL, 2 mmol) and water (0.036 mL, 2 mmol) were added and
the solution was purged with nitrogen for 20 min at room temperature.
The reaction mixture was protected from light and BF3·Et2O (0.158 mL,
1.25 mmol) was added. After 1 h of stirring at room temperature, DDQ
(0.908 g, 4 mmol) was added and stirring was continued for a further
5 min. The solvent was removed with a vacuum rotary evaporator. The
dark residue was dissolved in dichloromethane and subjected to prelimi-
nary chromatography separation on a short basic alumina (grade II)
column. The fast-moving fraction was collected and separated again by
chromatography on a basic alumina (grade II) column using as the eluent
a mixture of dichloromethane/hexane (3:7 v/v). iso-8 eluted first as a see-
green fraction. The crude product was further purified by column chro-
matography basic alumina (grade II, dichloromethane/hexane 1.5:8.5 v/
v). Yield of iso-8: 8 mg (1%), 1H NMR (CD2Cl2, 250 K): d=9.98 (s, 1H;
24-NH), 8.64 (s, 1H; 22-NH), 7.47, 7.33 (2d, 2H; 10-o-Tol), 7.36 (d, 1H;
15-o-Tol), 7.31 (d, 2H; 15-o-Tol, 10-m-Tol), 7.28 (d, 1H; 7), 7.26 (d, 1H;
10-m-Tol), 7.22, 7.19 (2d, 2H; 15-m-Tol), 7.17 (d, 2H; 21-o-Tol), 7.16 (d,
2H; 5-o-Tol), 7.14–7.11 (m, 5H; 2-o,m,p-Ph), 7.10 (t, 1H; 3-p-Ph), 7.05
(t, 2H; 3-m-Ph), 6.96 (d, 2H; 5-m-Tol), 6.93 (d, 1H; 8), 6.88 (d, 1H; 13),
6.86 (d, 2H; 21-m-Tol), 6.75 (d, 2H; 3-o-Ph), 6.66 (m, 1H; 18), 6.59 (d,
1H; 12), 6.39 (d, 1H; 17), 6.23 (s, 1H; 1), 2.44 (s, 3H; 10-p-Me), 2.39 (s,
3H; 15-p-Me), 2.23 (s, 3H; 5-p-Me), 2.12 ppm (s, 3H; 21-p-Me);
AHCTREUNG
AHCTREUNG
1 h. The solvent was removed under reduced pressure. The residue was
dissolved in dichloromethane and separated by chromatography on a
silica gel (mesh 70–230) column. The first red-green band was collected
and evaporated to dryness. The residue contains practically pure 8-Cu
complex. Yield: 16.6 mg (95%), 1H NMR (CDCl3, 230 K): d=8.85, 6.30
(2d, 2H; 2-o-Ph, 190 K), 8.66 (d, 1H; 7), 8.60 (d, 1H; 18), 8.49 (d, 1H;
13), 8.48 (d, 1H; 8), 8.47 (d, 1H; 17), 8.23 (d, 1H; 12), 8.22 (d, 1H; 15-o-
Tol), 7.92 (d, 1H; 10-o-Tol), 7.88 (d, 2H; 5-o-Tol, 10-o-Tol), 7.71 (d, 1H;
3-o-Ph), 7.69 (d, 1H; 5-o-Tol), 7.64 (d, 1H; 15-o-Tol), 7.58 (d, 1H; 15-m-
Tol), 7.50 (d, 1H; 10-m-Tol), 7.46 (d, 1H; 10-m-Tol), 7.42 (d, 1H; 15-m-
Tol), 7.34 (t, 1H; 2-m-Ph), 7.32 (d, 1H; 5-m-Tol), 7.24 (t, 1H; 3-m-Ph),
7.18 (t, 1H; 2-m-Ph), 7.05 (m, 1H; 3-p-Ph), 6.98 (m, 1H; 2-p-Ph, 190 K)
6.82 (d, 1H; 5-m-Tol), 6.61 (t, 1H; 3-m-Ph), 6.00 (d, 1H; 3-o-Ph), 5.63 (d,
2H; 21-m-Tol), 4.21 (d, 2H; 21-o-Tol), 2.59 (s, 3H; 10-p-Me), 2.58 (s,
3H; 15-p-Me), 2.37 (s, 3H; 5-p-Me), 1.36 ppm (s, 3H; 21-p-Me);
13C NMR (CDCl3, 230 K): d=149.6 (1), 148.7, 142.2 (6, 9), 146.1, 138.2
(16, 19), 144.6, 141.0 (11, 14), 143.7 (3), 141.0 (C1–2-Ph), 139.6 (4), 138.5
(C1–10-Tol), 138.4 (C4–21-Tol), 137.5 (C4–10-Tol), 137.4 (C4–5-Tol), 137.3
(C4–15-Tol), 137.2 (C1–3-Ph), 137.0 (C1–15-Tol), 136.5 (2), 136.3 (C1–5-
Tol), 135.1 (5-o-Tol), 134.2, 134.0 (15-o-Tol), 134.1 (10-o-Tol), 133.9, 133.8
(5-o-Tol, 10-o-Tol), 132.8, 129.5 (3-o-Ph), 132.3 (2-o-Ph), 130.7 (5), 130.5
(C1–21-Tol), 130.4 (13), 129.8 (8), 129.0, 125.9 (2-m-Ph), 128.6 (17),
4872
ꢀ 2008 Wiley-VCH Verlag GmbH & Co. KGaA, Weinheim
Chem. Eur. J. 2008, 14, 4861 – 4874

 Skonieczny, Janusz
Skonieczny, Janusz
 Latos-Grazynski, Lechoslaw
Latos-Grazynski, Lechoslaw
 Szterenberg, Ludmila
Szterenberg, Ludmila