A. Sen et al.
by taking the area for methylcyclohexane and dividing by the total area
(methylcyclohexane and toluene).
and heated to 808C for 9 h. The toluene layer was then analyzed by GC.
The reaction was resumed after charging with 300 psi of H2. After heat-
ing at 808C for 20 min the toluene layer was re-analyzed by GC. The
heating/GC analysis/hydrogen re-charge was repeated after 30, 30, 60,
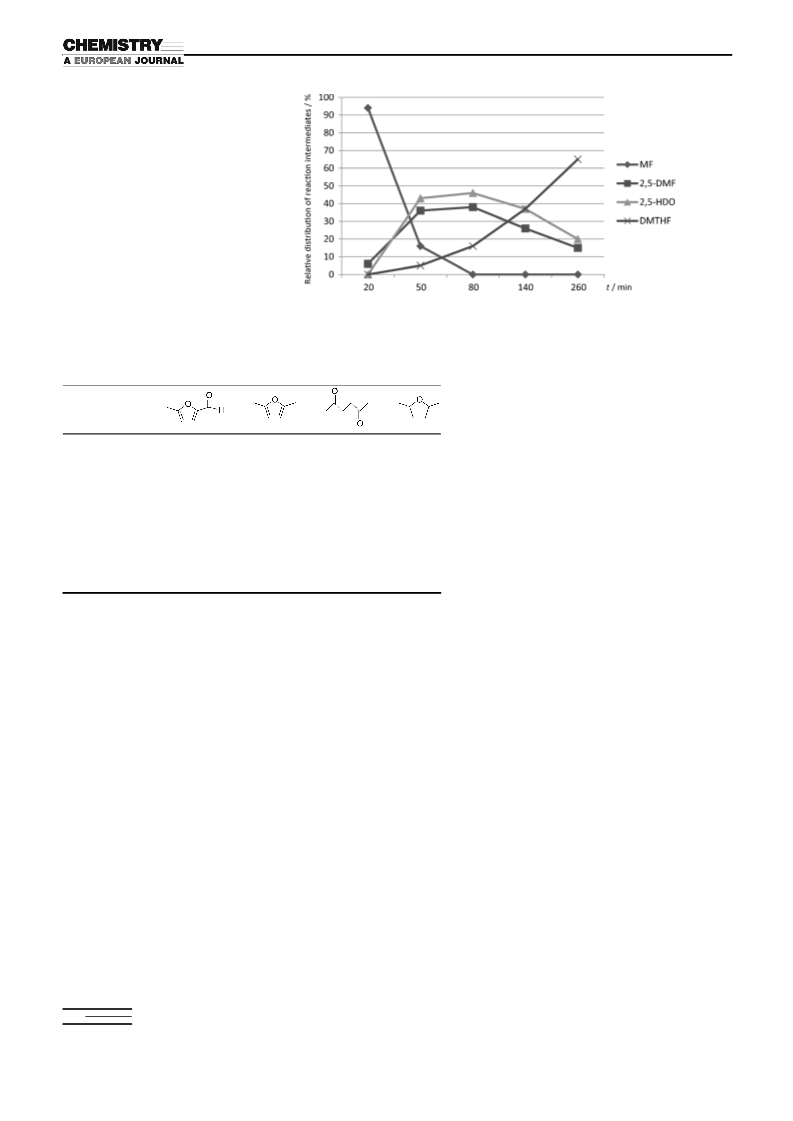
and 120 min. The relative percentages (Table 1) of the intermediates
were obtained by taking the area of the intermediate and dividing by the
total area of all the intermediates.
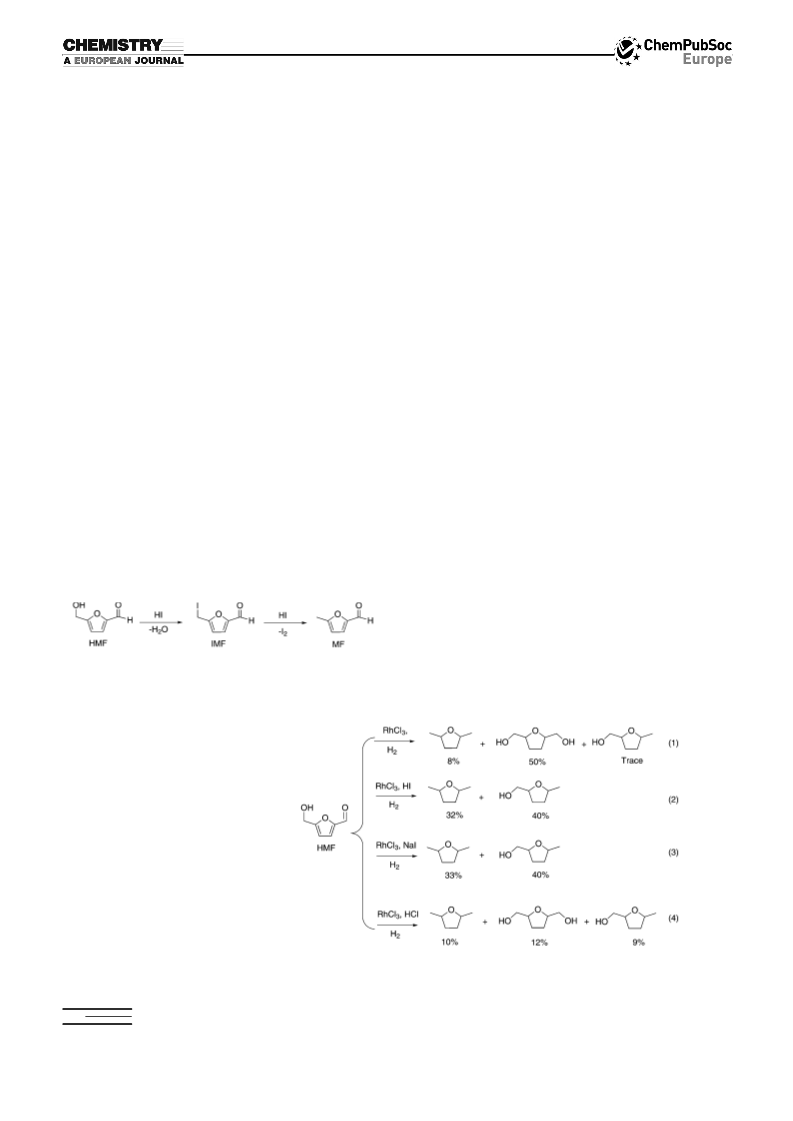
Procedures for reactions shown in Scheme 4: Equation (1): RhCl3·3H2O
(0.05 mmol, 13.2 mg), water (2 mL), and toluene (2 mL) were added to a
glass reactor liner in open air. Then the vial was put into a high pressure
stainless steel reactor, flushed three times with H2, and charged with
300 psi of H2. The bomb was placed in an oil bath and heated to 1408C
for 2 h. After heating both layers of the recovered solution were clear
and colorless with suspended black particles at the solvent interface.
Fructose (1 mmol, 180 mg) and HI (1.5 mmol, 57 wt% in water, 0.2 mL)
were then added to the reaction mixture. The bomb was recharged with
hydrogen (300 psi) and heated at 1408C for 3.5 h. The recovered toluene
layer was orange and clear, and was characterized by GC. DMTHF for-
mation was not observed.
Independent synthesis of DMTHF starting with the major intermediates:
General procedure (Scheme 6): The intermediate (1 mmol), RhI3
(0.05 mmol, 24.2 mg) HI (1.5 mmol, 57 wt% in water, 0.2 mL), water
(1.8 mL), and toluene or benzene (2 mL) were added to a glass reactor
liner in open air. Then the vial was put into a high pressure stainless steel
reactor, flushed three times with H2, and charged with 300 psi of H2. The
bomb was placed in an oil bath and heated to 1258C for 4–12 h. The re-
action with MFM was done on a smaller scale to minimize side products
from the highly reactive MFM starting material. The toluene layer was
Equation (2): RhI3 (0.05 mmol, 24.2 mg) water (2 mL), and toluene
(2 mL) were added to a glass reactor liner in open air. Then the vial was
put into a high pressure stainless steel reactor, flushed three times with
H2, and charged with 300 psi of H2. The bomb was placed in an oil bath
and heated to 1258C for 2.5 h. After heating, both layers of the recovered
solution were clear and colorless with suspended black particles coating
the stirrer bar in the water layer. Fructose (1 mmol, 180 mg) and HI
(1.5 mmol, 57 wt% in water, 0.2 mL) were then added to the reaction
mixture. The bomb was recharged with hydrogen (300 psi) and heated at
1258C for 16 h. The recovered toluene layer was yellow-orange and
slightly cloudy, whereas the water layer was yellow and clear with black
solid suspended at the solvent interface. The toluene layer was character-
1
characterized and quantified by GC and H NMR.
Reaction with HMF: HMF (1 mmol, 126 mg) was heated for 12 h in ben-
zene/water. DMTHF yield was 54%.
Reaction with MF: MF (1 mmol, 99.5 mL) was heated for 12 h in toluene/
water. DMTHF yield was 100%.
Reaction with MFM: MFM (0.2 mmol, 21 mL) and HI (0.30 mmol,
57 wt% in water, 40 mL), was heated for 4 h in benzene/water. DMTHF
yield was 41%, 2’2-methylenebis(5-methyl-furan) yield was 18%, HDO
yield was 3%.
Reaction with 2,5-DMF: DMF (1 mmol, 106.5 mL) was heated for 12 h in
toluene/water. DMTHF yield was 95%.
1
ized and quantified by GC and H NMR (40% yield of DMTHF).
Equation (3): RhI3 (0.05 mmol, 24.2 mg), HI (1.5 mmol, 57 wt% in water,
0.2 mL), toluene (2 mL) and water (1.8 mL) were added to a glass reac-
tor liner in open air. Then the vial was put into a high pressure stainless
steel reactor, flushed three times with helium, and charged with 300 psi
of helium. The bomb was placed in an oil bath and heated to 1258C for
3.5 h. After heating the water solution was cloudy with a brown color
whereas the toluene solution was clear and colorless. Fructose (1 mmol,
180 mg) was then added to the reaction mixture. The bomb was re-
charged with hydrogen (300 psi) and heated at 1258C for 14 h. The recov-
ered toluene layer was clear and colorless, whereas the water layer was
yellow and clear with black solid suspended at the solvent interface. The
toluene layer was characterized and quantified by GC and 1H NMR
(82% yield of DMTHF).
Reaction with 2,5-HDO: HDO (1 mmol, 117 mL) was heated for 5 h in
toluene/water. DMTHF yield was 100%.
Reaction with 2,5-hexanediol: 2,5-hexanediol (1 mmol, 123 mL) was
heated for 4 h in toluene/water. DMTHF yield was 100%.
Deuterium labeling experiments: Conversion of Fructose to DMTHF
using D2: Fructose (1 mmol, 180 mg), RhCl3·3H2O (0.10 mmol, 26 mg),
HI (1.5 mmol, 57 wt% in water, 0.2 mL), water (1.8 mL), and
[D6]benzene (2 mL) were added to a glass reactor liner in open air. Then
the vial was put into a high pressure stainless steel reactor, flushed three
times with D2, and charged with 60 psi of D2. The bomb was placed in an
oil bath and heated to 1408C for 26 h. After the reaction was complete
the benzene layer was dark opaque whereas the water layer was red. The
benzene layer was washed with saturated sodium thiosulfate solution
(aq), and filtered through glass wool. The benzene layer was analyzed by
GC, GC-MS, and NMR spectroscopy (1H and 13C).
Equation (4): RhI3 (0.05 mmol, 24.2 mg), HI (1.5 mmol, 57 wt% in water,
0.2 mL), toluene (2 mL) and water (1.8 mL) were added to a glass reac-
tor liner in open air. Then the vial was put into a high pressure stainless
steel reactor, flushed three times with hydrogen, and charged with 300
psi of hydrogen. The bomb was placed in an oil bath and heated to
1258C for 2.5 h. After heating the toluene solution was clear and color-
less, whereas the water layer was clear and yellow. Suspended black solid
was present at the solvent interface and on the bottom of the water
layer. Fructose (1 mmol, 180 mg) was then added to the reaction mixture.
The bomb was recharged with hydrogen (300 psi) and heated at 1258C
for 19 h. The recovered toluene layer was slightly cloudy with a green
tint, while the water layer was yellow and clear with black solid suspend-
ed at the solvent interface. The toluene layer was characterized and
Conversion of Fructose to DMTHF using DI: Fructose (1 mmol, 180 mg),
RhCl3·3H2O (0.05 mmol, 13.2 mg), HI (1.5 mmol, 57 wt% in water,
0.2 mL), D2O (2 mL), and [D6]benzene (2 mL) were added to a glass re-
actor liner in open air. Then the vial was put into a high pressure stain-
less steel reactor, flushed three times with H2, and charged with 300 psi
of H2. The bomb was placed in an oil bath and heated to 1408C for 6 h.
After the reaction was complete the benzene was filtered through glass
wool to remove suspended particles, washed with saturated sodium thio-
sulfate solution (aq), and filtered through glass wool. The benzene layer
was analyzed by GC, GC-MS, and NMR spectroscopy (1H and 13C). See
Table 2 for % deuteron incorporation into DMTHF.
1
quantified by GC and H NMR (22% yield of DMTHF).
Reaction of RhI3, HI, and H2 without fructose: RhI3 (0.30 mmol,
145 mg), HI (9 mmol, 57 wt% in water, 1.2 mL), and water (6 mL) were
added to a glass reactor liner in open air. Then the vial was put into a
high pressure stainless steel reactor, flushed three times with hydrogen,
and charged with 300 psi of hydrogen. The bomb was placed in an oil
bath and heated to 1258C for 2 days. The water was carefully removed
by pipette and the suspended black solid was dried in a 1008C oven for
8 h. The black solid was analyzed by ICP-MS: 78.0% Rh, 23.0% I.
Equilibration of 2,5-DMF and 2,5-HDO with DI: DMF (1 mmol,
106 mL), RhCl3·3H2O (0.10 mmol, 26 mg), HI (1.5 mmol, 57 wt% in
water, 0.2 mL), D2O (2 mL), and [D6]benzene (2 mL) were added to a
glass reactor liner in open air. Then the vial was put into a high pressure
stainless steel reactor, flushed three times with H2, and charged with
300 psi of H2. The bomb was placed in an oil bath and heated to 1408C
for 12 h. The recovered benzene solution was clear with a light beige tint
while the water layer was still a dark red color. The benzene layer was
analyzed by GC, GC-MS, and NMR spectroscopy (1H and 13C). See
Tables 3 and 4 for % deuteron incorporation of 2,5-DMF and 2,5-HDO.
Fructose conversion to DMTHF: Identification of reaction intermediates:
Fructose (1 mmol, 180 mg), RhCl3·3H2O (0.10 mmol, 26 mg) HI
(10 mmol, 57 wt% in water, 1.4 mL), water (3 mL), and toluene (4 mL)
were added to a glass reactor liner in open air. Then the vial was put into
a high pressure stainless steel reactor, flushed three times with helium,
and charged with 300 psi of helium. The bomb was placed in an oil bath
Interconversion of cis/trans isomers of DMTHF: DMTHF (0.83 mmol,
100 mL), RhCl3·3H2O (0.10 mmol, 26 mg), HI (1.5 mmol, 57 wt% in
water, 0.2 mL), D2O (2 mL), and [D6]benzene (2 mL) were added to a
glass reactor liner in open air. Then the vial was put into a high pressure
&
8
&
ꢀ 2012 Wiley-VCH Verlag GmbH & Co. KGaA, Weinheim
Chem. Eur. J. 0000, 00, 0 – 0
ÝÝ
These are not the final page numbers!

 Grochowski, Matthew R.
Grochowski, Matthew R.
 Yang, Weiran
Yang, Weiran
 Sen, Ayusman
Sen, Ayusman