JOURNAL OF CHEMICAL RESEARCH 2015 551
O
O
COCl
c
OH
O
O
OH
OH
O
O
O
a
b
O
O
O
O
O
2
3
4
O
OH
O
OH
HO
O
O
O
e
d
O
O
OH
O
1
5
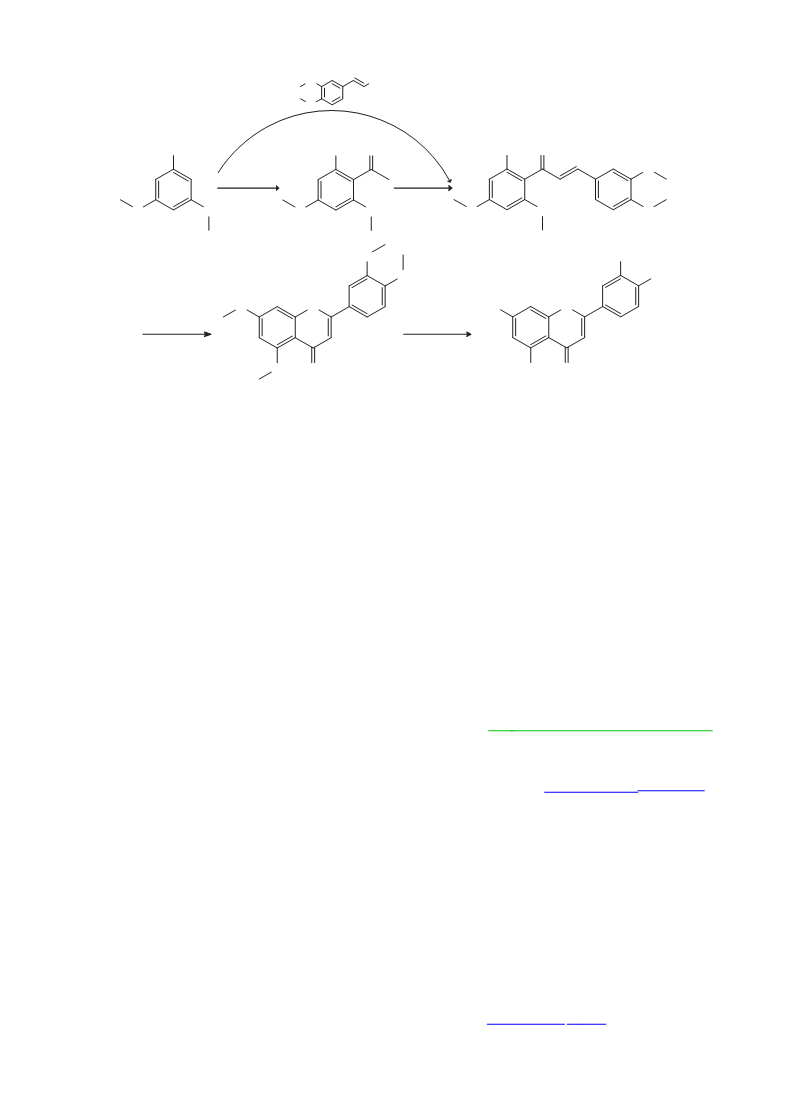
Scheme 1 Reagents and conditions:(a) ZnCl , CH3COOH, 145 °C, 2 h, 79%; (b) 3,4-dimethoxybenzaldehyde, KOH, room temperature, 78 h, 81%;
(c)3,4-dimethoxycinnamoyl chloride2, BF3·Et2O, reflux, 1.5 h, 55%; (d) I2, DMSO, 120 °C, 5 h, 82%; (e) Py·HCl, 190 °C, 6 h, 89%.
1
temperature apparatus, H NMR spectral data were recorded on a
Bruker Avance 400 NMR spectrometer or a Bruker DRX 500 NMR
spectrometer, chemical shifts were reported in ppm against internal
tetramethylsilane. Mass spectra were determined on VG Auto Spec-
3000 spectrometer and reported as m/z. All reagents were purchased
from Tansoole-reagent, China, and used without further purification.
temperature and H2O (100 mL) was added. The mixture was stirred
for another 1 h and cooled to approximately 0 °C for several hours. The
precipitate was filtered off, washed with water and recrystallised from
ethyl acetate to give compound 1 as yellow crystals (1.3 g, 89%); m.p.
329–330 °C (lit.39 328–330 °C); IR νmax (KBr/cm−1): 3490 (OH), 1664
1
(C=O);H NMR (500 MHz, DMSO-d6) (δ, ppm): 12.95 (s, 1H, OH),
10.86 (s, 1H, OH), 9.95 (s, 1H, OH), 9.44 (s, 1H, OH), 7.43–7.41 (m, 1H),
7.39 (d, J = 2.1 Hz, 1H), 6.89 (d, J = 8.3 Hz, 1H), 6.68 (s, 1H), 6.44 (d,
J = 1.9 Hz,1H), 6.19 (d, J = 1.9 Hz, 1H); MS (m/z): 309 [M+Na]+.
2′-Hydroxy-3,4,4′,6′-tetramethoxychalcone (4)
(Scheme 1, Step b): Potassium hydroxide (11.2 g, 0.2 mol) was added
to methanol (90 mL). After cooling to room temperature, compound
3 (2.0 g, 0.01 mol) and 3,4-dimethoxybenzaldehyde (1.8 g, 0.011 mol)
were added to the solution. It was stirred for 78 h at room temperature.
Then the mixture was acidified to pH 5 with 10% aqueous HCl. The
precipitate was filtered off, washed with water and recrystallised from
ethanol to give yellow crystals of compound 4 (2.8 g, yield 81%).
(Scheme 1, Step c): A mixture of 3,5-dimethoxyphenol 2 (3.1 g,
0.02 mol) and 3,4-dimethoxycinnamoyl chloride (5.0 g, 0.022 mol)
was dissolved in BF3–Et2O complex (20 mL) and heated to reflux
for 1.5 h, and then quenched with water (100 mL). Filtration and
recrystallisation from ethanol gave yellow crystals of compound 4
(3.8 g, 55%); m.p. 154–156 °C (lit.37 154–155 °C); IR νmax (KBr/cm−1):
3516 (OH), 1684 (C=O), 1625 (C=C); 1H NMR (500 MHz, DMSO-d6)
(δ, ppm): 14.17(s, 1H, OH), 7.68 (d, J = 15.5 Hz, 1H), 7.32 (d, J = 15.6
Hz, 1H), 7.23 (dd, J = 8.3,1.9 Hz, 1H), 7.16 (d, J = 1.9 Hz, 1H), 6.95 (d,
J = 8.3 Hz, 1H), 6.12 (d, J = 2.3 Hz, 1H), 5.96 (d, J = 2.3 Hz, 1H), 3.94
(s, 3H, OCH3), 3.92 (s, 3H, OCH3), 3.91 (s, 3H, OCH3), 3.85 (s, 3H,
OCH3); MS (m/z): 345 [M+H]+.
This work was supported by the National Natural Science
Foundation of China (NSFC) (no. 21062009) and Analysis
and Testing Foundation of Kunming University of Science
and Technology (no. 20150728). This support is gratefully
acknowledged.
Received 12 August 2015; accepted 20 August 2015
Published online: 1 September 2015
References
1
2
M. Horinaka, T. Yoshida, T. Shiraishi, S. Nakata, M. Wakada, R. Nakanishi,
H. Nishino, H. Matsui and T. Sakai, Oncogene, 2005, 24, 7180.
H.W.C. Leung, C.L. Kuo, W.H. Yang, C.H. Lin and H.Z. Lee, Eur. J.
Pharmacol., 2006, 508, 77.
3
4
3′,4′,5,7-Tetramethoxyflavone (5): Compound 4 (3.4 g, 0.01mol)
and iodine (0.3 g) in DMSO (20 mL) were stirred at 120 °C for 5 h
and then 2.0% NaHSO3 (50 mL) was added to remove the iodine.
The precipitate was filtered off, washed with water and recrystallised
from ethanol to give white crystals of compound 5 (2.8 g, 82%); m.p.
192–193 °C (lit.38 193 °C); IR νmax (KBr/cm−1): 1647 (C=O), 1632
S. Roy, S. Mallick, T. Chakraborty, N. Ghosh, A.K. Singh, S. Manna and S.
Majumdar, Food Chem., 2015, 173, 1172.
5
6
G. Seelinger, I. Merfort and C.M. Schempp, Planta Med., 2008, 74, 1667.
P.C. Lv, H.Q. Li, J.Y. Xue, L. Shi and H.L. Zhu, Eur. J. Med. Chem., 2009,
44, 908.
7
8
9
L. Xu, W.H. Su, J. Jin, J.W. Chen, X.J. Li, X.Y. Zhang, M.Y. Sun, S.Y. Sun,
P.H. Fan, D. An, H.F. Zhang, X.G. Zhang, W. Kong, T.H. Ma and C.L.
Jiang, Viruses, 2014, 6, 2778.
D. Sawmiller, S. Li, M. Shahaduzzaman, A.J. Smith, D. Obregon, B.
Giunta, C.V. Borlongan, P.R. Sanberg and J. Tan, Int. J. Mol. Sci., 2014,
15, 895.
1
(C=C);H NMR (500 MHz, DMSO-d6) (δ, ppm): 7.52 (dd, J = 8.5, 2.1
Hz, 1H), 7.33 (d, J = 2.1 Hz, 1H), 6.96 (d, J = 8.5 Hz, 1H), 6.63 (s, 1H),
6.58 (d, J = 2.4 Hz, 1H), 6.39 (d, J = 2.4 Hz, 1H), 3.98 (s, 3H, OCH3),
3.97 (s, 3H, OCH3), 3.94 (s, 3H, OCH3), 3.92 (s, 3H, OCH3); MS (m/z):
343 [M+H]+.
Q. Zhou, B. Yan, X. Hu, X.B. Li, J. Zhang and J. Fang, Mol. Cancer Ther.,
2009, 8, 1684.
Luteolin (1): A mixture of compound 5 (1.7 g, 5 mmol) and excess
pyridine hydrochloride (5.8 g, 0.05 mol) was heated at 190 °C for
6 h under a N2 atmosphere. The mixture was then cooled to room
10 E. Bagli, M. Stefaniotou, L. Morbidelli, M. Ziche, K. Psillas, C. Murphy

 Wang, Qian
Wang, Qian
 Zhang, Ji
Zhang, Ji
 Liu, Man
Liu, Man
 Yang, Jian
Yang, Jian
 Zhang, Xiang-Ming
Zhang, Xiang-Ming
 Zhou, Lei
Zhou, Lei
 Cao, Lang
Cao, Lang
 Liao, Xia-Li
Liao, Xia-Li