Journal of the American Chemical Society
Page 4 of 5
(
(
4) Huang, F. C.; Chan, W. K.; Moriarty, K. J.; Zhang, D. C.;
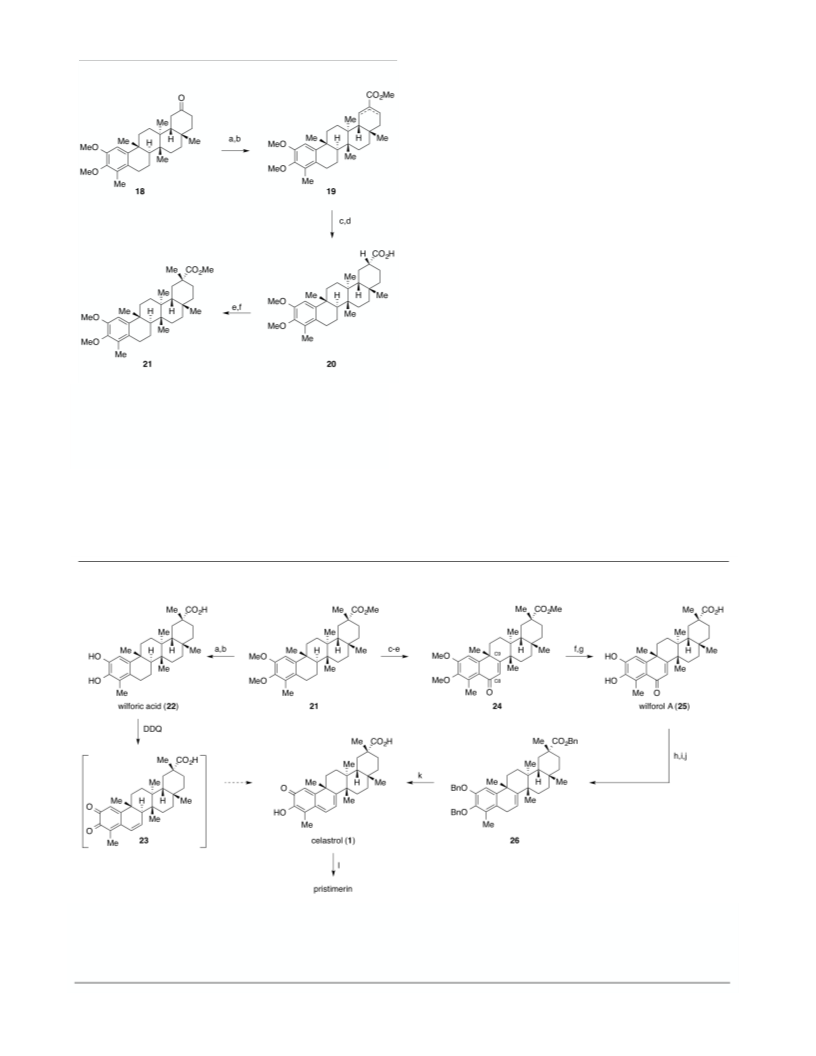
Subsequent LiAlH reduction with careful monitoring of
the reaction’s progress by TLC with gradual warming from
4
1
2
3
4
5
6
7
8
9
1
1
1
1
1
1
1
1
1
1
2
2
2
2
2
2
2
2
2
2
3
3
3
3
3
3
3
3
3
3
4
4
4
4
4
4
4
4
4
4
5
5
5
5
5
5
5
5
5
5
6
Chang, M. N.; He, W.; Yu, K. T.; Zilberstein, A. Bioorg. Med.
Chem. Lett. 1998, 8, 1883.
5) Trott, A.; West, J. D.; Klaić, L.; Westerheide, S. D.; Silverman,
R. B.; Morimoto, R. I.; Morano, K. A. Mol. Biol. Cell 2008, 19,
1104.
−
78 to −30 °C allowed access to the desired alcohol while
limiting reduction of the hindered E-ring ester. The result-
ing active benzyl-allylic alcohol was deoxygenated via
ionic reduction with TFA and Et SiH to provide the per-
(6) Mu, T.-W.; Ong, D. S. T.; Wang, Y.-J.; Balch, W. E.; Yates, J.
3
2
9
R.; Segatori, L.; Kelly, J. W. Cell 2008, 134, 769.
7) Ngassapa, O.; Soejarto, D. D.; Pezzuto, J. M.; Farnsworth, N. R.
J. Nat. Prod. 1994, 57, 1.
8) Liu, J.; Lee, J.; Salazar Hernandez, M. A.; Mazitschek, R.
Ozcan, U. Cell 2015, 161, 999.
benzyl ester 26 in moderate yield over two steps. In a
single operation, perbenzyl derivative 26 was subjected to
hydrogenation using Pd/C and upon indication (by TLC) of
completion the flask was opened to atmosphere and stirred
vigorously to afford celastrol (1) as a red-orange solid in
(
(
0
1
2
3
4
5
6
7
8
9
0
1
2
3
4
5
6
7
8
9
0
1
2
3
4
5
6
7
8
9
0
1
2
3
4
5
6
7
8
9
0
1
2
3
4
5
6
7
8
9
0
(9) De Santana, C. F. Rev Inst Antibiot. Recife 1971, 11, 37.
(10) Melo, A. M.; Jardim, M. L.; De Santana, C. F.; Lacet, Y.; Lobo
Filho, J.; e Ivan Leoncio, O. G. Rev. Inst. Antibiot. (Recife).
1974, 14, 9.
75% yield.
Celastrol in turn was esterified using
TMSCHN in methanol/benzene to produce the closely
2
3
0
related natural product pristimerin (27).
(
11) U.S. National Institutes of Health. United States Clinical Trials
Online Registry; National Institutes of Health:
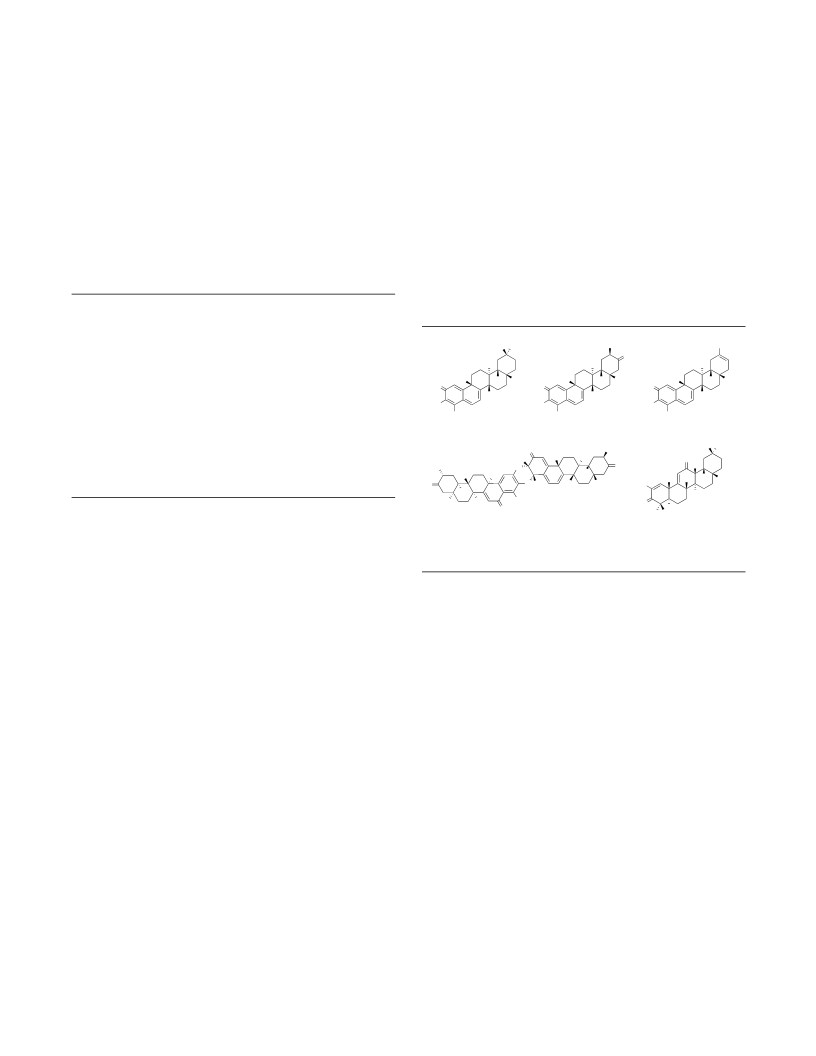
In conclusion a platform utilizing a polyene cascade
was developed to provide accessed the pentacyclic frame-
work of the celastroid class of triterpenoids lending to the
total synthesis of celastrol and related natural products wil-
foric acid, wilforol A, and pristimerin. The allylic alcohol
cyclization precursor 16 is accessed in >5 gram quantities
in twelve steps (longest linear) commencing from 2,3-
dimethylbutadiene with an overall yield of 21%. The de-
veloped cascade employs ferric chloride as an activator in a
dilute solution of CH Cl to generate the pentacycle in 38%
(accessed October 26, 2014).
(12) a) Klaic, L.; Morimoto, R. I.; Silverman, R. B. ACS Chem. Biol.
012, 7, 928. (b) Klaic, L.; Trippier, P. C.; Mishra, R. K.;
Morimoto, R. I.; Silverman, R. B. J. Am. Chem. Soc. 2011, 133,
9634.
2
1
(
13) Campanelli, A. R.; D’Alagni, M.; Marini-Bettolo, G. B. FEBS
Lett. 1980, 122, 256.
(14) a) Johnson, W. S. Angew .Chem. Int. Ed. 1976, 15, 9. b)
Johnson, W. S.; Semmelhack, M. F.; Sultanbawa, M. U. S.;
Dolak, L. A. J. Am. Chem. Soc., 1968, 90, 2994.
(
(
2
2
yield on gram scale and showcases the utility of this rea-
gent for polyene cyclizations. Through this intermediate
the first syntheses of celastrol and pristimerin were com-
pleted in 31 and 32 (longest linear steps respectively) as
well as wilforic acid and wilforol A.
15) Yoder, R. A.; Johnston, J. N. Chem. Rev. 2005, 105, 4730.
16) Kurti, L. .; Chein, R.-J.; Corey, E. J. J. Am. Chem. Soc. 2008,
1
30, 9031.
(17) a) Corey, E. J.; Ursprung, J. J. J. Am. Chem. Soc. 1955, 77,
3667. b) Corey, E. J.; Ursprung, J. J. J. Am. Chem. Soc. 1955,
7
7, 3668
(
(
18) Corey, E. J.; Ursprung, J. J. J. Am. Chem. Soc. 1956, 78, 5041.
19) Ireland, R. E.; Walba, D. M. Tetrahedron Lett. 1976, 17, 1071.
ASSOCIATED CONTENT
Supporting Information.
Experimental methods and spectral data is available free of
charge via the Internet at http://pubs.acs.org.
(20) Ireland, R. E.; Dawson, M. I.; Welch, S. C.; Hagenbac A.;
Bordner, J.; Trus, B. J. Am. Chem. Soc. 1973, 95, 7829.
(21) Ireland, R. E.; Evans, D. A.; Glover, D.; Rubottom, G. M.;
Young, H. J. Org. Chem. 1969, 34, 3717.
(
22) Ireland, R. E.; Bey, P.; Cheng, K. F.; Czarny, R. J.; Moser, J. F.;
Trust, R. I. J. Org. Chem. 1975, 40, 1000.
23) Ireland, R. E.; Mckenzie, T. C.; Trust, R. I. J. Org. Chem. 1975,
40, 1007.
AUTHOR INFORMATION
Corresponding Author
(
*
(24) Sweeting, O. J.; Johnson, J. R. J. Am. Chem. Soc. 1946, 68,
057.
25) Cook, S. P.; Danishefsky, S. J. Org. Lett. 2006, 8, 5693.
1
Funding Sources
(
Financial support was provided by the Welch Foundation
(26) Dess, D. B.; Martin, J. C. J. Org. Chem. 1983, 48, 4155.
(27) Stork, G. T.; Szmuskovicz, J. J. Am. Chem. Soc. 1954, 76, 2029.
(
F-1694) and the University of California, San Diego.
(
(
(
28) Sen, S. E.; Roach, S. L.; Smith, S. M.; Zhang, Y. Z. Tetrahedron
Lett. 1998, 39, 3969.
29) Kursanov, D. N.; Parnes, Z. N.; Loim, N. M. Synthesis (Stuttg).
Notes
The authors declare no competing financial interest.
1
974, 9, 633.
30) Nakanishi, K.; Kakisawa, H.; Hirata, Y. J. Am. Chem. Soc. 1955,
7, 3169.
ACKNOWLEDGMENT
7
The authors thank, at UT Austin, Dr. Ben Shoulders, Steve
Sorey, Angela Spangenberg, and Howard Johnson for their
assistance with NMR experiments as well as helpful dis-
cussions regarding the subject of this papers. The authors
also thank Dr. Vince Lynch for assistance with X-ray crys-
tallography.
REFERENCES
(
(
(
1) Gunatilaka, A. A. L. Triterpenoid Quinonemethide and Related
Compounds, Springer-Verlag/Wien, Austria; 1996.
2) Kiaei, M.; Kipiani, K.; Petri, S.; Chen, J.; Calingasan, N. Y.;
Beal, M. F. Neurodegener. Dis. 2005, 2, 246.
3) Zhang, Y.-Q.; Sarge, K. D. J. Mol. Med. (Berl). 2007, 85, 1421.
ACS Paragon Plus Environment

 Camelio, Andrew M.
Camelio, Andrew M.
 Johnson, Trevor C.
Johnson, Trevor C.
 Siegel, Dionicio
Siegel, Dionicio