EPSRC Mass Spectrometry Service University of Wales Swansea,
UK using the ionization method specified. Removal of solvent
refers to evaporation at reduced pressure using a rotary evaporator
followed by the removal of trace volatiles using a vacuum pump.
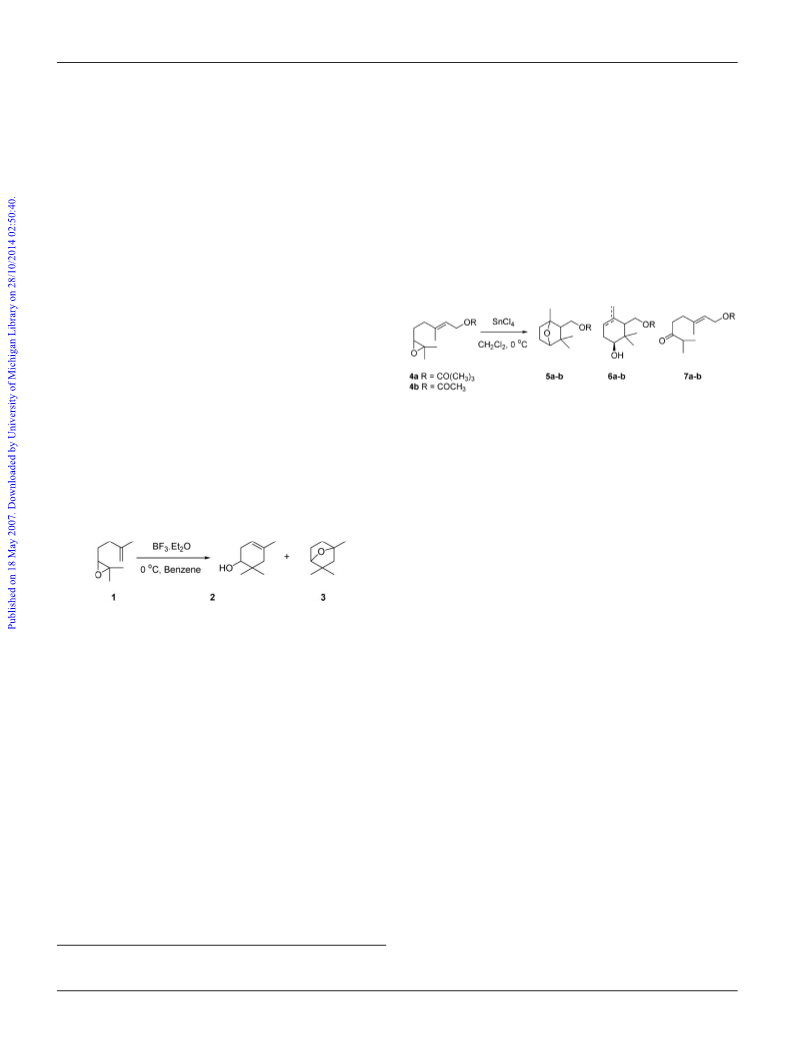
6,7-Epoxygeranyl acetate 4b
1
(71% yield); H NMR (CDCl3; 400 MHz) d = 1.19 (3H, s), 1.24
(3H, s), 1.56–1.63 (2H, m), 1.66 (3H, s), 1.98 (3H, s), 2.03–2.21
(2H, m), 2.63 (1H, t, J = 7 Hz), 4.52 (2H, d, J = 7 Hz), 5.32 (1H,
t, J = 7 Hz); 13C NMR (CDCl3; 100 MHz) d = 171.5, 141.6, 119.3,
64.3, 61.6, 58.8, 36.6, 27.5, 25.2, 21.4, 19.1, 16.9; mmax (film)/cm−1
(neat) 2962, 2926, 1736, 1448, 1378, 1228, 1121, 1022, 954 and
679; MS (EI) m/z 212 (M)+; HRMS (ES, NH3) calculated for
C12H24NO3 (M + NH4)+ 230.1751, found (M + NH4)+ 230.1748.
Geranyl pivalate
To a solution of geraniol (4.00 g, 25.9 mmol) in pyridine
(10 ml) was added trimethylacetyl chloride (3.52 g, 29.2 mmol)
and a catalytic quantity of 4-dimethylaminopyridine (158.4 mg,
1.30 mmol) and the reaction mixture was left to stir at room
temperature for 6 hours. Dichloromethane (40 ml) was added and
the organic phase washed with water (2 × 50 ml), hydrochloric
acid (2 × 20 ml, 1 M solution) and sodium hydrogen carbonate
solution (4 × 20 ml, 10% solution). The organic phase was then
dried over magnesium sulfate and the solvent removed under
reduced pressure to give a yellow oil which was purified by column
chromatography (hexane → 10% ethyl acetate : hexane) to give the
product geranyl pivalate as a colourless oil (5.86 g, 95%); 1H NMR
(CDCl3; 400 MHz) d = 1.12 (9H, s), 1.53 (3H, s), 1.61 (3H, s), 1.63
(3H, s), 1.94–2.08 (4H, m), 4.50 (2H, d, J = 7 Hz), 5.00 (1H, t, J =
7 Hz), 5.25 (1H, t, J = 7 Hz); 13C NMR (CDCl3; 100 MHz) d =
179.0, 142.0, 132.1, 124.2, 119.1, 61.7, 39.9, 39.1, 27.6, 26.7, 26.1,
18.1, 16.8; mmax (film)/cm−1 (neat) 2970, 2931, 1727, 1480, 1397,
1281, 1147, 939 and 771; MS (EI) m/z 238, (M)+; HRMS (EI)
calculated for C15H26O2 (M)+ 238.1927, found (M)+ 238.1926.
Typical experimental procedure for the indium
tribromide-mediated cyclization of 6,7-epoxygeranyl pivalate
To a solution of 6,7-epoxygeranyl pivalate (115 mg, 0.45 mmol) in
anhydrous dichloromethane (10 ml) under a nitrogen atmosphere
was added indium tribromide (161 mg, 0.45 mmol) and the
reaction stirred at room temperature for 4 hours. The solvent
was then removed under reduced pressure and the crude reaction
mixture passed through a short plug of silica which was washed
with 10% ethyl acetate : hexane. Removal of the solvent under
reduced pressure gave the product 2,2-dimethylpropionic acid 1,3,3-
trimethyl-7-oxa-bicyclo[2.2.1] hept-2-ylmethyl ester 5a (102 mg,
1
89%); H NMR (CDCl3; 400 MHz) d = 0.98 (3H, s), 1.03 (3H,
s), 1.13 (9H, s), 1.29 (3H, s), 1.36–1.69 (5H, m), 3.71 (1H, d, J =
5 Hz), 3.91 (1H, dd, J = 11 and 7 Hz), 4.06 (1H, dd, J = 11 and
7 Hz); 13C NMR (CDCl3; 100 MHz) d = 177.4, 85.0, 84.7, 62.5,
53.3, 43.9, 37.6, 37.4, 26.2, 24.9, 24.8, 22.2, 17.3; mmax (film)/cm−1
(neat) 2964, 2876, 2256, 1720, 1458, 1384, 1287, 1167, 1075 and
919; MS (EI) m/z 254 (M)+; HRMS (ES, NH3) calculated for
C15H27O3 (M + H)+ 255.1955, found (M + H)+ 255.1958.
Geranyl acetate
1
(99% yield); H NMR (CDCl3; 400 MHz) d = 1.61 (3H, s), 1.69
(3H, s), 1.71 (3H, s), 2.07 (3H, s), 2.08–2.15 (4H, m), 4.60 (2H,
d, J = 7 Hz), 5.09 (1H, t, J = 7 Hz), 5.35 (1H, t, J = 7 Hz); 13
C
NMR (CDCl3; 100 MHz) d = 171.5, 142.6, 132.2, 124.5, 118.6,
61.8, 39.9, 26.7, 26.1, 21.4, 18.1, 16.8; mmax (film)/cm−1 (neat) 2968,
2919, 2857, 1739, 1443, 1365, 1227, 1021, 953 and 607; MS (EI)
m/z 196 (M)+; HRMS (ES, NH3) calculated for C12H24NO2 (M +
NH4)+ 214.1802, found (M + NH4)+ 214.1802.
2,3-Epoxygeranyl acetate 8
To a solution of geraniol (6.22 g, 40.3 mmol) and vanadyl
acetylacetonate (149.3 mg, 0.56 mmol) in chloroform (50 ml)
was added an anhydrous solution of tert-butyl hydroperoxide (5–
6 M solution in decane, 8 ml, 44.4 mmol) dissolved in chloroform
(2 ml) drop-wise over 20 minutes and the reaction heated to reflux
for 6 hours. The reaction mixture was then washed with sodium
metabisulfite solution (2 × 40 ml, 10% solution), the organic phase
was dried over magnesium sulfate and the solvent was removed
under reduced pressure. The crude product obtained was then
stirred at room temperature with acetic anhydride (20 ml) and
pyridine (25 ml) for 18 hours. Dichloromethane was then added
(40 ml) and the reaction mixture washed with water (2 × 50 ml),
hydrochloric acid (2 × 20 ml, 1 M solution) and sodium hydrogen
carbonate solution (4 × 25 ml, 10% solution). The organic phase
was then dried over magnesium sulfate and the solvent removed
under reduced pressure to give a yellow oil which was purified by
column chromatography (hexane → 10% ethyl acetate : hexane)
to give the product 2,3-epoxygeranyl acetate 8 as a colourless oil
(5.75 g, 67%); 1H NMR (CDCl3; 400 MHz) d = 1.30 (3H, s), 1.42–
1.51 (2H, m), 1.60 (3H, s), 1.68 (3H, s), 2.04–2.08 (2H, m), 2.09
(3H, s), 2.98 (1H, dd, J = 4 and 7 Hz), 4.02 (1H, dd, J = 7 and
12 Hz), 4.30 (1H, dd, J = 4 and 12 Hz), 5.07 (1H, t, J = 7 Hz);
13C NMR (CDCl3; 100 MHz) d = 171.3, 132.6, 123.6, 63.8, 61.0,
60.0, 38.7, 26.1, 24.0, 21.2, 18.1, 17.2; mmax (film)/cm−1 (neat) 2978,
2928, 2251, 1714, 1651, 1445, 1378, 1219, 1146, 1037, 914, 870
6,7-Epoxygeranyl pivalate 4a
To a solution of geranyl pivalate (1.63 g, 6.85 mmol) in
dichloromethane (75 ml) at room temperature was added 3-
chloroperoxybenzoic acid (77%, 1.25 g, 7.24 mmol) and sodium
hydrogen carbonate (608 mg, 7.24 mmol) and the reaction mixture
was left to stir at room temperature for 48 hours. The reaction
mixture was then washed with sodium metabisulfite solution (2 ×
50 ml, 10% solution) and sodium hydrogen carbonate solution
(4 × 50 ml, 10% solution). The organic phase was then dried
over magnesium sulfate and the solvent removed under reduced
pressure to give a yellow oil which was purified by column
chromatography (hexane → 10% ethyl acetate : hexane) to give
the product 6,7-epoxygeranyl pivalate 4a as a colourless oil (1.14 g,
66%); 1H NMR (CDCl3; 400 MHz) d = 1.12 (9H, s), 1.19 (3H, s),
1.23 (3H, s), 1.55–1.63 (2H, m), 1.65 (3H, s), 2.02–2.20 (2H, m),
2.63 (1H, t, J = 7 Hz), 4.50 (2H, d, J = 7 Hz), 5.30 (1H, t, J =
7 Hz); 13C NMR (CDCl3; 100 MHz) d = 138.7, 130.7, 122.9, 122.3,
60.2, 58.4, 38.5, 26.2, 25.4, 24.7, 16.7, 15.2; mmax (film)/cm−1 (neat)
2962, 2933, 1726, 1481, 1459, 1281, 1148, 939 and 771; MS (EI)
m/z 254 (M)+; HRMS (ES, NH3) calculated for C15H27O3 (M +
H)+ 255.1955, found (M + H)+ 255.1957.
This journal is
The Royal Society of Chemistry 2007
Org. Biomol. Chem., 2007, 5, 1979–1982 | 1981
©

 Smith, Brendan M.
Smith, Brendan M.
 Skellam, Elizabeth J.
Skellam, Elizabeth J.
 Oxley, Samantha J.
Oxley, Samantha J.
 Graham, Andrew E.
Graham, Andrew E.