Angewandte
Research Articles
Chemie
amount of starphene dimer or higher oligomers that could be
the central ring. However, despite the positive value of the
central ring, the coplanar arrangement of the three pentacene
units likely allows their partial electronic coupling.
[25]
formed during the thermal decarbonylation. Storing 1 in
a glove box for one month at room temperature did not lead
13
to any change in the C CP-MAS NMR spectrum (See
Supporting Information, Figure S20) confirming its good
stability in the solid-state form.
STM Experiments
To elucidate the electronic properties of 1, we measured
its absorption spectrum in a frozen matrice and compared it
In order to verify the chemical and electronic structure of
the [16]starphene 1 via low-temperature STM experiments,
the molecules needed to be deposited onto a clean surface
under ultra-high-vacuum (UHV). Attempts to sublime pre-
cursors 8a and 8b under UHV proved unsuccessful. Instead,
saturated solutions of the two precursors in acetonitrile were
deposited onto an Au(111) surface in high vacuum with an
atomic layer injection source (“ALI”, see Supporting Infor-
mation for details). The samples were then post-annealed at
1508C for 5 minutes to desorb a significant proportion of the
contaminants present in the solution. Large-scale STM
images recorded at 4.3 K before and after the annealing
process are presented in the Supporting Information (Fig-
ure S33). Very low coverage of starphene-shaped molecules
was observed after this process, identified by their distinctive
shape in the STM images. To our surprise, the formation of
starphene-shaped structures was observed only when the
precursor with a table-like structure (8b) was deposited. The
isomer 8a likely desorbs before being decarbonylated. An
STM image showing two of these starphene-shaped molecules
is presented in Figure 4a. Functionalisation of the STM probe
with a CO molecule and scanning in the repulsive CO-
adsorbate interaction regime provides HR-STM images that
[20,26]
with that of pentacene
similar conditions from a carbonylated pentacene precursor
see Figure S29 for its structure). Solutions of 8a, 8b,
respectively, were prepared in toluene with concentrations
(Figure 3c) prepared under
(
À4
À1
1
0
molL and the spectra recorded at room temperature
(
Figure S27a, S28a). The samples were then cooled to 5 K,
irradiated with UV light (310 nm) for 15 min to generate
starphene 1 and measured again (see Supporting Information
for further experimental details). A strong and broad
absorption band with maximum at around 479 nm and then
three weaker absorption bands with maxima at 558, 595 and
6
(
1
41 nm appear in the spectrum of 1. The obvious red shift
55 nm) of the longest wavelength absorption band of
compared to that of pentacene (at 586 nm) indicates
electronic conjugation among the pentacene arms in 1. Based
[26]
on the comparison with the literature data, it seems that the
electronic properties of 1, diluted in frozen matrices, are
similar to those of hexacene (rather than of longer acenes).
Finally, MS MALDI analysis (Figure S23) of 1 revealed
a mass of 828.5 Da, which agrees with the predicted value of
8
28.3 Da.
[31]
resolve the internal structure of molecules at surfaces. Such
images clearly show the distinguishable differences in the
middle rings of all three arms of the starphene objects found
on the surface when compared to the adjacent aromatic rings
(Figure 4b). While these features coincide with the carbonyl-
functionalised rings of 8b, we believe a decarbonylation has
already taken place and those rings are instead hydrogenated
(Figure 4c). The following findings support our hypothesis:
(I) occasionally, the different ring does not coincide with the
originally functionalised position, but with a neighbouring
ring (Supporting Information, Figure S34); (II) the post-
deposition annealing treatment has been performed at
a temperature at which the decarbonylation is readily known
to set in (see Figure 2); (III) finally, STM images of precursors
closely related to 8b, with bridging ketones, which have been
used to form long acenes at surfaces in other studies, usually
yield a greatly enhanced signal at the location of the ketone
Theoretical Calculations
The molecular orbitals of [16]starphene 1, including its
Kohn–Sham HOMO and LUMO, respectively, were calcu-
lated. The molecular geometry was optimised at the B3LYP/
6
-31G(d,p) level of theory, and the molecular orbitals were
calculated at the B3LYP/6–311 ++ g(2d,p) level of theory.
The HOMO and LUMO are delocalised over the entire
conjugated backbone of 1, and the HOMO–LUMO gap was
determined as 1.64 eV, which is smaller than that of com-
[27]
parable calculations on pentacene (2.20 eV) or hexacene
[
28]
(
1.76 eV). Together with the calculated HOMO–LUMO
gap (1.95 eV) of angularly fused pentacenes (corresponding
to two thirds of starphene 1), the values manifest evolution of
the HOMO–LUMO gap when going from pentacene to
starphene (Figure S32), thus supporting the hypothesis that
the three pentacene arms are partially conjugated. To
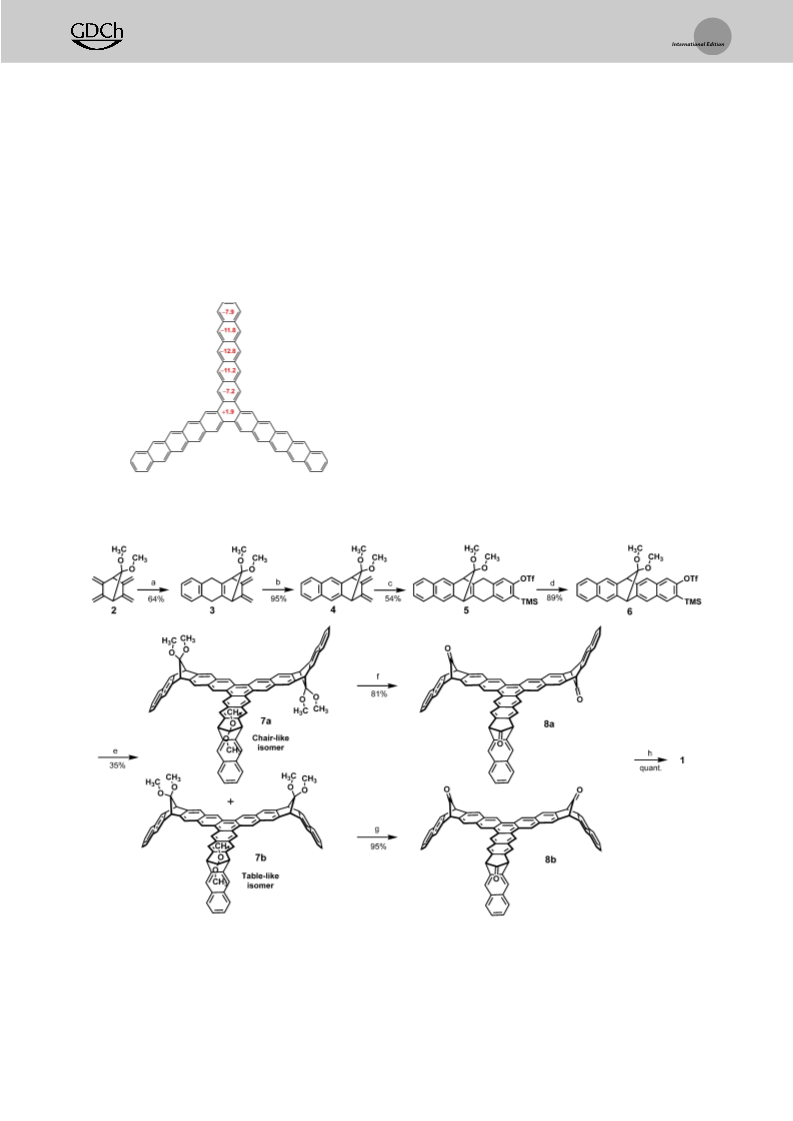
evaluate the local aromaticity of 1, nucleus-independent
chemical shift (NICS) values were calculated (using B3LYP/
[32,33]
bridge.
In contrast to this, we typically observe a reduced
signal in the centre of the arms of these objects when imaging
at a voltage that is within the energy gap of the molecule
(Figure S35), in line with other studies of hydrogenated
[29]
6
–311 ++ g(2d,p) level of theory). The NICS values 1.0
[34]
above the molecular plane NICS(1) of 1 (Figure 1) on the
benzene moieties along a pentacene arm range from À7.2 to
À12.8, indicating their high degree of aromaticity. The
NICS(1) values increase towards the middle rings of the arms
suggesting an increase of aromaticity. On the other hand, the
NICS(1) value of the central ring of 1 is + 1.9, displaying
acenes on surfaces. Such hydrogenation breaks the con-
jugation along the starphene arms, resulting in what can be
seen as “independent” conjugated moieties. Indeed, it has
been previously shown that each of the conjugated sections
hosts molecular orbitals that are extremely similar in energy
and wavefunction symmetry to those of the correspondingly
[30]
[34,35]
almost no aromaticity.
This positive value suggests that
sized acene.
In this case, the molecule comprises naph-
single bonds interconnect the three pentacene arms and form
thalene moieties at the end of each arm and a [7]starphene in
Angew. Chem. Int. Ed. 2021, 60, 7752 – 7758
ꢀ 2021 Wiley-VCH GmbH
7755

 Holec, Jan
Holec, Jan
 Cogliati, Beatrice
Cogliati, Beatrice
 Lawrence, James
Lawrence, James
 Berdonces-Layunta, Alejandro
Berdonces-Layunta, Alejandro
 Herrero, Pablo
Herrero, Pablo
 Nagata, Yuuya
Nagata, Yuuya
 Banasiewicz, Marzena
Banasiewicz, Marzena
 Kozankiewicz, Boleslaw
Kozankiewicz, Boleslaw
 Corso, Martina
Corso, Martina
 de Oteyza, Dimas G.
de Oteyza, Dimas G.
 Jancarik, Andrej
Jancarik, Andrej
 Gourdon, Andre
Gourdon, Andre