S. Goswami, A. K. Adak / Tetrahedron Letters 43 (2002) 8371–8373
8373
References
well with water and then with ethanol, and dried in
vacuum. The bright yellow solid (1.2 g, 70%, mp>350°C)
after pivaloylation with pivalic anhydride followed by
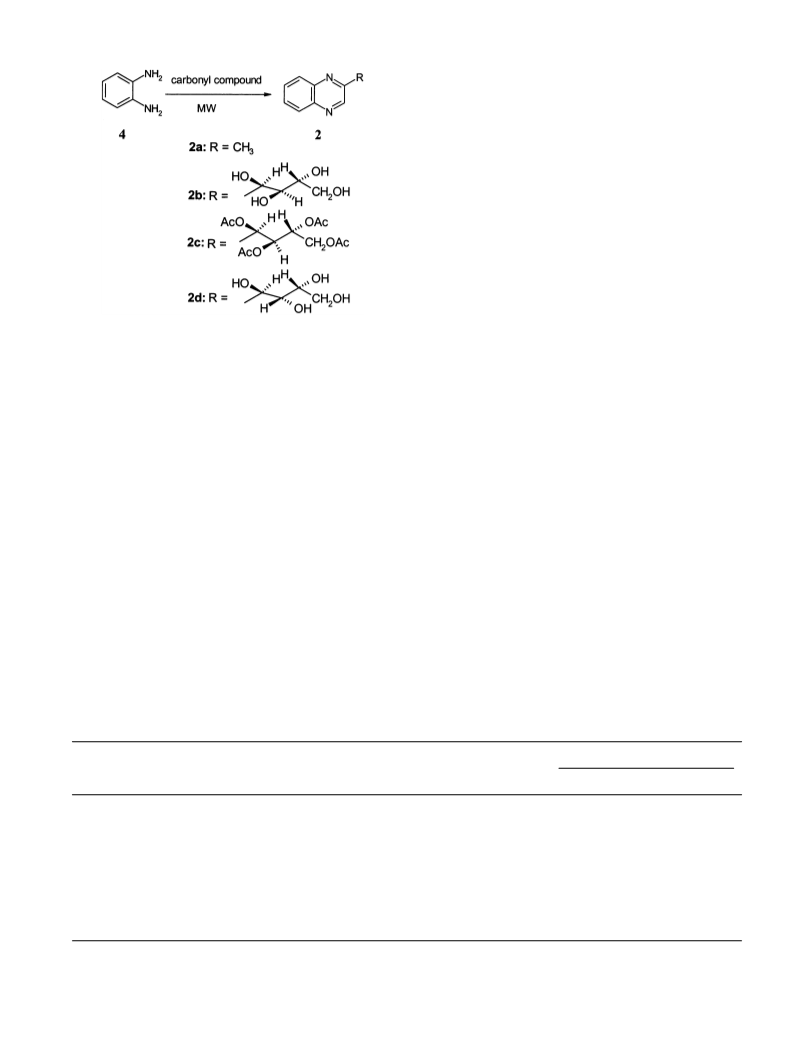
purification gave a cream coloured solid 1b (1.4 g, 78%,
mp 230–232°C). Compounds 1d, 1f and 2c were obtained
by condensation followed by direct acetylation of 1c, 1e
and 2b, respectively.
1. (a) Hopkins, F. G. Nature 1889, 40, 335; (b) Hopkins, F.
G. Nature 1891, 45, 197; (c) Hopkins, F. G. Nature 1892,
45, 581.
2. (a) Pilato, R. S.; Erickson, K. A.; Greaney, M. A.; Stiefel,
E. I.; Goswami, S. P.; Kilpatric, L.; Spiro, T. G.; Taylor,
E. C.; Rhiengold, A. L. J. Am. Chem. Soc. 1991, 113,
9372–9374; (b) Taylor, E. C.; Goswami, S. P. Tetrahedron
Lett. 1992, 32, 7357–7360; (c) Goswami, S. P.; Adak, A.
K. Tetrahedron Lett. 2002, 43, 503–505.
3. (a) Pateman, J. A.; Coves, D. J.; Rever, B. M.; Roberts,
D. B. Nature 1964, 201, 58; (b) Hille, R. Chem. Rev. 1996,
96, 2757–2816; (c) Collison, D.; Garner, C. D.; Joule, J.
A. Chem. Soc. Rev. 1996, 25; (d) Goswami, S. P. Hetero-
cycles 1993, 35, 1552–1572; (e) Johnson, J. L.; Wuebbens,
M. M.; Rajagopalan, K. V. J. Biol. Chem. 1989, 264,
13440–13447; (f) For a recent paper in this series, see:
Bradshaw, B.; Dinsmore, A.; Ajana, W.; Collison, D.;
Garner, C. D.; Joule, J. A. J. Chem. Soc., Perkin Trans.
1 2001, 3239–3244.
4. (a) Waring, P.; Armarego, W. L. F. Aust. J. Chem. 1985,
38, 629–631; (b) Taylor, E. C.; Perlman, K. L.; Sword, I.
P.; Sequin-Frey, M.; Jacobi, P. A. J. Am. Chem. Soc.
1973, 95, 6407–6412; (c) Russell, J. R.; Garner, C. D.;
Joule, J. A. Synlett 1992, 711–712.
5. (a) Caddick, S. Tetrahedron 1995, 51, 10403–10432; (b)
Hennkens, P. H. H.; Ottenhejm, H. C.; Rees, D. C.
Tetrahedron 1997, 53, 5643–5678; (c) Loupy, A.; Pettit,
A.; Hamelin, J.; Texier-Boulet, F.; Jacquault, P.; Mathe,
D. Synthesis 1998, 1213–1233; (d) Verma, R. S. Green
Chem. 1999, 43–55; (e) for a recent review on microwave
assisted organic synthesis, see: Lidstrom, P.; Tierney, J.;
Wathey, B.; Westman, J. Tetrahedron 2001, 57, 9225–
9283.
6. Typical experimental procedure: A mixture of 2,5,6-tri-
aminopyrimidin-4(3H)-one hydrochloride 3 (2.0 g) and
methylglyoxal (1.5 g, 40% in water) was placed in a
microwave oven (BPL 800G, indicates the commercial
name of the microwave oven) and subjected to irradiation
at 150 W for an optimised time (62 s). Water was then
added and the resulting slurry was centrifuged. The solid
separated was filtered through a sintered funnel, washed
7. (a) Karrer, P.; Schwyzer, R.; Erden, B.; Siegwart, A.
Helv. Chim. Acta 1947, 30, 1031; (b) Sato, H.; Nakajima,
M.; Tanaka, H. J. Chem. Soc. Jpn. 1951, 72, 868–870.
Chem. Abstr. 1953, 5946–5947; (c) Jones, R. G.; Korn-
feld, E. C.; McLaughlin, K. C. J. Am. Chem. Soc. 1950,
72, 3539–3542; (d) Gerchakov, S.; Whitman, P. J.;
Schultz, H. P. J. Med. Chem. 1966, 9, 266.
8. (a) Taylor, E. C.; Dumas, D. J. J. Org. Chem. 1981, 46,
1394; (b) Taylor, E. C.; Ray, P. S. J. Org. Chem. 1987,
52, 3994.
1
9. The H NMR spectra were found to be identical to those
reported earlier. Compound 1b (78%). Mp 320–322°C. 1H
NMR (CDCl3, 500 MHz): l 12.33 (br s, 1H, NH), 8.72
(s, 1H, C7-H), 8.35 (br s, 1H, NH), 2.75 (s, 3H, C6-CH3),
1.36 (s, 9H). Compound 1f (40%). Mp 108–110°C. 1H
NMR (CDCl3, 500 MHz): l 12.60 (br s, 1H, NH amide),
10.23 (br s, 1H, lactam NH), 8.94 (s, 1H, C7-H), 6.05 (d,
1H, C1%-H, J=8.9 Hz), 5.72 (dd, 1H, C%2-H, J=2.6, 2.6
Hz), 5.59–5.56 (m, 1H, C%3-H), 4.24 (qd, 2H, C%4-H2,
J=5.5, 5.5, 6.9 Hz, 6.9), 2.45 (s, 3H, -NHCOCH3), 2.15,
2.13, 2.05, 1.96 (4×s, 12H, -OCOCH3×4). [h]2D5 −17.31 (c
1, chloroform). Mass (FAB, MH+): 494 (100%). Com-
1
pound 2a (95%). H NMR (CDCl3, 500 MHz): l 8.58 (s,
1H, quinoxalin-2-yl), 7.92 (d, 1H, J=8.0 Hz), 7.87 (d,
1H, J=8.0 Hz), 7.60–7.54 (m, 2H), 2.66 (s, 3H). Com-
1
pound 2c (65%). Mp 110–112°C. H NMR (CDCl3, 500
MHz): l 8.84 (s, 1H, quinoxalin-2-yl), 8.10–8.08 (m, 2H),
7.79–7.77 (m, 2H), 6.33 (d, 1H, J=3.1 Hz), 5.79 (dd, 1H,
C2%-H, J=3.1, 3.1 Hz), 5.40–5.37 (m, 1H, C3%-H), 4.25 (qd,
2H, C4%-H2, J=2.7, 2.7, 5.0, 5.0 Hz), 2.22, 2.09, 2.04, 1.91
(4×s, 12H, 4×OCOCH3). Mass (FD, M+): 418 (100%).
10. (a) Russell, J. R.; Garner, C. D.; Joule, J. A. J. Chem.
Soc., Perkin Trans. 1 1992, 1245–1249; (b) Horton, D.;
Miller, M. J. J. Org. Chem. 1965, 30, 2457.
11. Goswami, S. P.; Adak, A. K. Synth. Commun. 2002, 33
(3).

 Goswami, Shyamaprosad
Goswami, Shyamaprosad
 Adak, Avijit Kumar
Adak, Avijit Kumar