
Bioorganic and Medicinal Chemistry Letters (2021)
Update date:2022-08-16
Topics:
 Yamamoto, Hirofumi
Yamamoto, Hirofumi
 Sakai, Naoki
Sakai, Naoki
 Ohte, Satoshi
Ohte, Satoshi
 Sato, Tomohiro
Sato, Tomohiro
 Sekimata, Katsuhiko
Sekimata, Katsuhiko
 Matsumoto, Takehisa
Matsumoto, Takehisa
 Nakamura, Kana
Nakamura, Kana
 Watanabe, Hisami
Watanabe, Hisami
 Mishima-Tsumagari, Chiemi
Mishima-Tsumagari, Chiemi
 Tanaka, Akiko
Tanaka, Akiko
 Hashizume, Yoshinobu
Hashizume, Yoshinobu
 Honma, Teruki
Honma, Teruki
 Katagiri, Takenobu
Katagiri, Takenobu
 Miyazono, Kohei
Miyazono, Kohei
 Tomoda, Hiroshi
Tomoda, Hiroshi
 Shirouzu, Mikako
Shirouzu, Mikako
 Koyama, Hiroo
Koyama, Hiroo
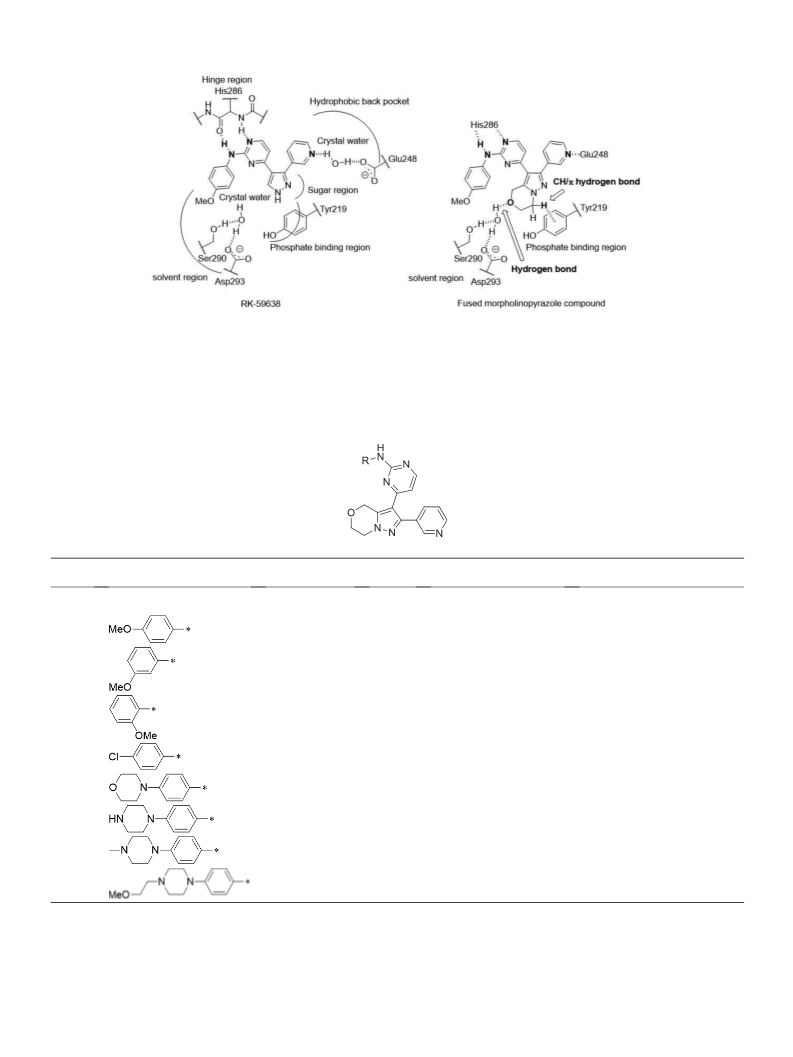
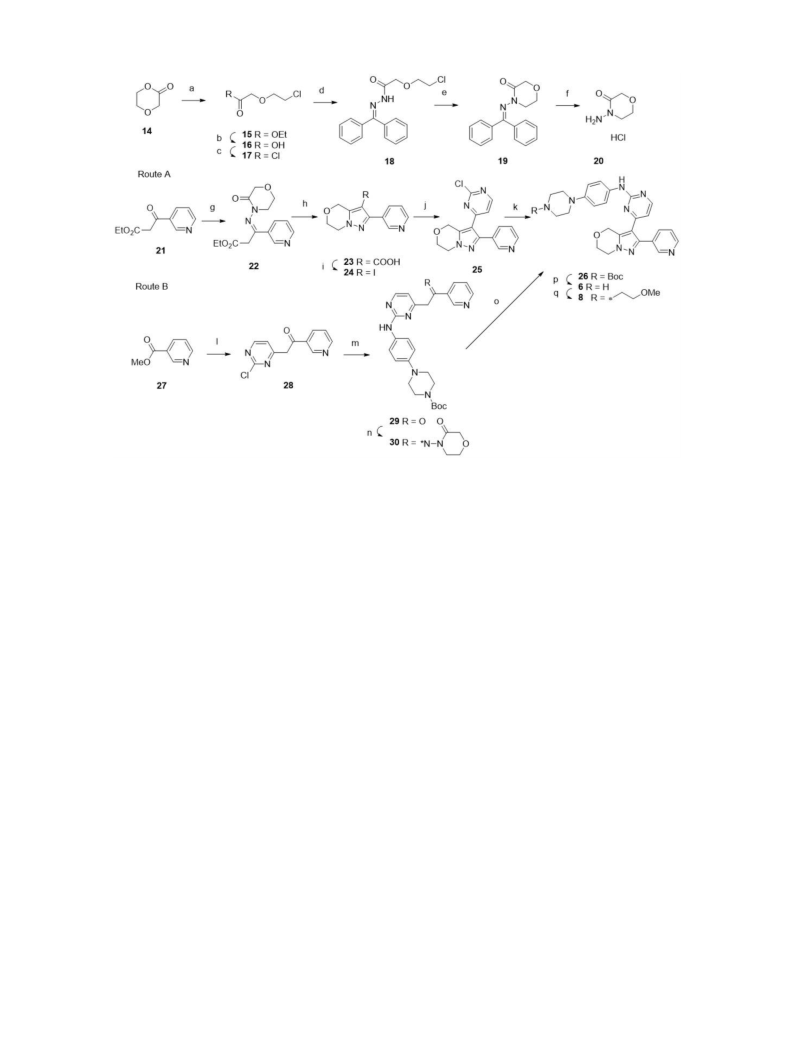
Mutant activin receptor-like kinase-2 (ALK2) is associated with the pathogenesis of fibrodysplasia ossificans progressiva, making it an attractive target for therapeutic intervention. We synthesized a new series of bicyclic pyrazoles and evaluated their mutant ALK2 enzyme inhibitory activities, leading to the identification of 8 as the most potent inhibitor. This compound showed moderate microsomal metabolic stability and human ether-a-go-go related gene (hERG) safety. In C2C12 cells carrying mutant ALK2 (R206H), 8 efficiently inhibited the bone morphogenetic protein (BMP)-induced alkaline phosphatase activity.
View More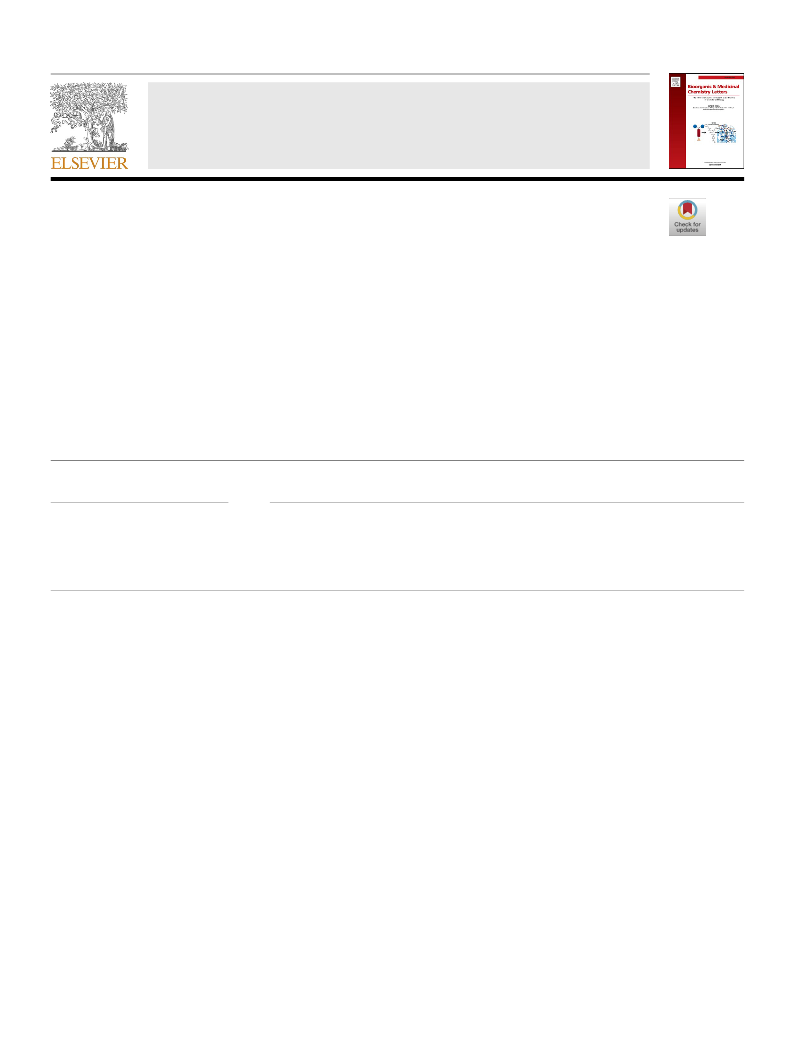

Hangzhou Haiqiang Chemical Co.,Ltd.
Contact:+86-571-86960370
Address:Add: 5/F, Around Town North Road,No. 10, Hangzhou, Zhejiang,China
Guangxi Nanning Guangtai Agriculture Chemical Co.,Ltd
Contact:+86-771-2311266
Address:Room703,Building12, Software Park Phase II,NO.68,Keyuan Road,Nanning City,Guangxi,China
Xiamen XM-Innovation Chemical Co., Ltd
Contact:+86-592-3216205
Address:Unit Q, 11/F, No.1 Office Building, Wuyuan Bay Business Center, Huli District, Xiamen City, Fujian Province, P.R.C
Taizhou KEDE Chemical.Co.,Ltd.
Contact:86-576-84613060
Address:Jiangkou Chemical Zoon
Xinxiang Junlong Biological Technology Co., Ltd.
website:https://junlongbio.lookchem.com/
Contact:86-13525059581
Address:Xinxiang City, Henan Province
Doi:10.1039/f19888402397
(1988)Doi:10.1016/j.steroids.2014.10.002
(2015)Doi:10.1021/ja00283a009
(1986)Doi:10.1016/S0031-9422(02)00288-1
(2002)Doi:10.1016/0022-328X(85)88056-6
(1985)Doi:10.1021/jo9801216
(1998)