Y. Abiedalla et al.
The potential for structural modification within the
cathinone chemical framework is quite vast and a large
number of precursor substances are commercially available.
These synthetic designer substances present unique challenges
in forensic analysis compared to those natural product derived
drugs (THC, cocaine, etc.) synthesized by a plant in an
enzymatically controlled (isomeric specific) process.
Technologies, Santa Clara, CA, USA). The mass spectral
scan rate was 2.86 scans/s. The GC system was operated
in splitless mode with a helium (grade 5) flow rate of
0.7 mL/min. The mass spectrometer was operated in the
electron ionization (EI) mode with an ionization voltage
of 70 eV and a source temperature of 230°C. The GC
injector was maintained at 250°C and the transfer line
at 280°C. The GC/MS analysis for the amino ketones
was carried out on a column (30 m × 0.25 mm i.d.) coated
with 0.10 μm film of Crossbond® 5% diphenyl, 95%
dimethyl polysiloxane (Rtx-5) purchased from Restek
Corporation (Bellefonte, PA, USA). The temperature
program consisted of an initial hold at 70°C for 1.0 min,
ramped up to 250°C at a rate of 30°C/min followed by
a hold at 250°C for 15 min.
GC/MS System 2 consisted of a model 7890A gas
chromatograph and a 7683B autoinjector coupled with a
model 240 ion trap mass spectrometer (all from Agilent
Technologies). The mass spectral scan rate was 2.86 scans/s.
The GC system was operated in splitless mode with a helium
(grade 5) flow rate of 0.7 mL/min and the column head
pressure was 10 psi. The mass spectrometer was operated in
the electron impact (EI) mode using an ionization voltage of
70 eV and a source temperature of 230°C. The MS/MS
excitation amplitudes ranged from 0.2 to 1.6 V. The GC
injector was maintained at 250°C and the transfer line at
280°C. The GC studies were performed on a column (30 m ×
0.25 mm i.d.) coated with 0.5 μm 100% trifluoropropyl methyl
polysiloxane (Rtx-200) purchased from Restek Corporation.
Chromatographic analysis was done using the temperature
program described for System 1 above. Samples for analysis
were dissolved and diluted in high-performance liquid
chromatography (HPLC)-grade acetonitrile (Fisher Scientific,
Fairlawn, NJ, USA) and introduced via the autoinjector using
an injection volume of 1 μL.
Chemical derivatization procedures often yield compounds
with altered or unique fragmentation pathways in mass
spectrometry as well as improved chromatographic properties.
Acylation of primary and secondary amines yields amides with
decreased nitrogen basicity and this reduction in electron
availability on nitrogen can yield different mass spectral
fragmentation pathways.[17] These different fragment ions
often provide additional structural information about the
parent primary or secondary amine. The perfluoroacyl
derivatives[17] can provide the best results among the
common acylation products yielding unique fragment ions
for clear structural information. Tertiary amines do not form
stable acylation products in most cases and therefore the
identification of regioisomeric molecules via mass spectral
fragmentation patterns is a greater analytical challenge. A
gas chromatography/mass spectrometry (GC/MS) method
has been described for separation and differentiation of the
3,4- and the 2,3-isomers of MDPV and the infrared (IR)
spectra of the 2,3-isomers were also compared with the
corresponding 3,4-isomers.[18] Mixtures of closely related
cathinones have been evaluated by standard ion mobility
spectrometry (IMS) and this technique compared to
electrospray ionization high-performance IMS methods.[19]
Multiple reaction monitoring techniques in reversed-phase
gradient elution liquid chromatography/tandem mass
spectrometry (LC/MS/MS) have been used to screen a
number of cathinone derivatives including mephedrone,
butylone, MDPV, flephedrone, methylone and methedrone
in forensic case samples.[20] The fragmentation properties
of the cathinone derivatives have been compared by GC/EI-
MS and LC/ESI-QTOFMS methods.[21] The precursor ions of
these substances are hard to obtain by EI-MS, whereas the
protonated molecular ions can be observed clearly by ESI-
QTOFMS. Furthermore, two major characteristic cleavages
are produced in the EI mode, leading to the formation of
iminium and acyl ions. Secondary and tertiary fragmentations
of these ions were reported to assist in molecular structure
identification.[21] In the case of ESI-QTOFMS, characteristic
fragments are produced via loss of water and other
fragmentation pathways not observed in EI-MS. Product ion
mass spectra can often provide structural information on
regioisomeric compounds and this is especially useful for
compounds yielding equivalent regioisomeric fragment ions.
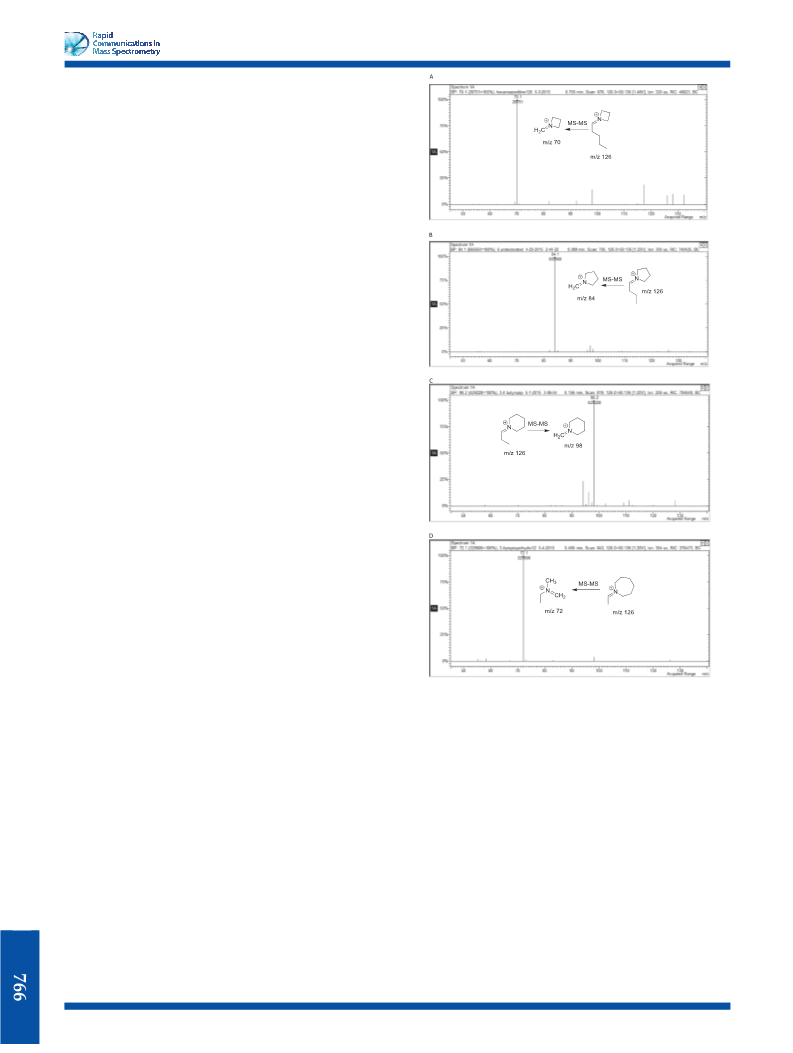
This report describes the use of product ion spectra in the
analysis of regioisomeric iminium cations generated in EI-MS
for a series of cathinone-type tertiary amines.
Synthetic methods
Precursor materials including piperonal, azetidine, pyr-
rolidine, piperidine, hexamethyleneimine (hexahydro-1H-
azepine) and various n-alkylmagnesium halides were
purchased from Aldrich Chemical Co. (Milwaukee, WI,
USA) or Alfa Aesar Chemical Co. (Ward Hill, MA, USA).
Samples of 2,2,3,3,4,4,5,5-pyrrolidine-D8, 2,2,5,5-pyrrolidine-
D4, piperadine-D10, and deuterated bromoalkanes were
purchased from CDN Isotopes (Pointe Claire, Quebec,
Canada). The synthetic methods needed to prepare the
various isomeric and homologous aminoketones in this study
are well established in the chemical literature and in our
laboratory. The procedures used in this project were those
reported by Kavanagh et al.[18] These desired compounds
were prepared from the substituted benzaldehydes via a
four-step synthetic procedure. The condensation of alkyl-
magnesium halides (Grignard reagents) with piperonal (3,4-
methylenedioxybenzaldehyde) yields the corresponding
methylenedioxybenzyl alcohols. Oxidation of these benzyl
alcohols with potassium dichromate yields the methylene-
dioxyalkylphenones. Alpha-bromination of the ketones at
the activated methylene carbon gives the alpha-bromoketones
and subsequent displacement of the bromide ion by the
nitrogen of the individual secondary amines yields the desired
EXPERIMENTAL
Instrumentation
GC/MS System
1 consisted of a model 7890A gas
chromatograph and a 7683B autoinjector coupled with a
5975C VL mass-selective detector (all from Agilent
wileyonlinelibrary.com/journal/rcm
Copyright © 2016 John Wiley & Sons, Ltd.
Rapid Commun. Mass Spectrom. 2016, 30, 763–772

 Abiedalla, Younis
Abiedalla, Younis
 Abdel-Hay, Karim
Abdel-Hay, Karim
 Deruiter, Jack
Deruiter, Jack
 Randall Clark
Randall Clark