J. Keskiväli et al. / Applied Catalysis A: General 534 (2017) 40–45
41
with a flame ionization detector (GC-FID). The GC-FID runs were
performed using Agilent Technologies 6890N Network GC System
fitted with Agilent HP-INNOWAX column (length 30 m, internal
diameter 0.25 mm and stationary phase thickness 0.25 m). The
calibration curves were plotted using standard samples with dif-
ferent concentrations of HMF (99%, Sigma Aldrich), furfural (≥98%,
Sigma Aldrich), pure 1 (column chromatography, EtOAc: hexane
1:1.5, Rf = 0.33), 2 (distillation in vacuo, bp. 96–97 ◦C at 1.5 mbar)
and pure 2-methyldecane (4) to calculate the selectivities, con-
versions and yields (See SI for more details). The yield of 3 was
determined from the calibration curve prepared from pure 4.
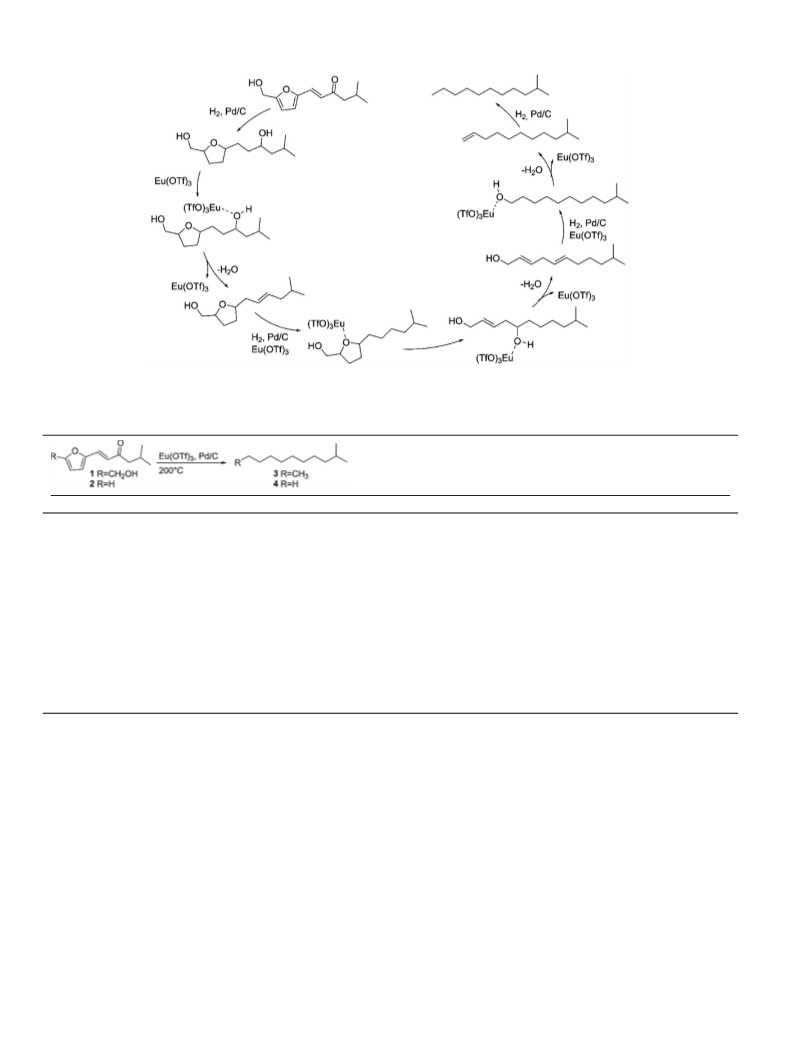
Scheme 1. The high yield two-step procedure from biomass-based furans to
branched C11 and C12 alkanes.
3. Results and discussion
producing alkanes in high overall yields of 86% and 94% from HMF
and industrially available furfural respectively (Scheme 1).
The aldol condensation of biomass-derived furfurals and
ketones is a classical, efficient and straightforward method of
combining aldehydes and ketones. In respect to biofuel research,
particular, the focus has been on the aldol condensation of acetone
with furfurals using different heterogeneous Brønsted catalysts
such as Mg-Al-oxide, MgO-ZrO2 and Nit-NaY (nitrogen-substituted
zeolite) [13,25,26]. As in fuel applications branched alkanes are pre-
ferred over linear alkanes due to their lower melting points and
higher octane numbers. In this respect, MIBK is an attractive choice
of substrate for the production of branched C12 and C11 furans via
the aldol condensation of HMF and furfural. In addition, MIBK is
available from acetone (product of acetone-butanol-ethanol fer-
mentation), it is regarded as a biomass-based chemical [27,28].
We initiated the study by investigating a solvent-free aldol con-
densation of HMF and furfural with MIBK using NaOH as a Brønsted
catalyst. Under the optimized reaction conditions (50 ◦C, 6 h, molar
ratio of 1:4 (furfurals:MIBK) and 13 mol-% of NaOH, See Scheme 1,
page S7 and Table S2 in SI), the products 1 and 2 were obtained in
excellent yields of 95% and 96% respectively. Due to the absence
of water, the amount of NaOH catalyst was significantly reduced
HMF (97 mol-% of NaOH) and furfural (37 mol-% of NaOH) [23,24].
Based on product analysis (GC-FID, 1H NMR) we did not observe any
from the acidified water phase (1H & 13C NMR) [29,30].
In the reports of Sutton et al. and Song et al. the HDO reac-
tions were conducted in two steps, in which the reaction routes
ring-opening of the 2,5-disubstituted furans, such as 1, to acyclic
peratures the La(OTf)3 cleaves the acetates and the desired alkane
is obtained through reduction of the formed C C double bonds
[15,32]. In contrast, in aprotic solvents such as n-octane, the simi-
higher temperatures, alkanes are formed through the Hf(OTf)4 cat-
H2-Pd/C (Scheme 2) [19,21].
2. Methods and materials
2.1. General procedure for the synthesis of 1 and 2
Desired amount of HMF/furfural (furfural (≥98%), HMF (99%)
Sigma Aldrich), MIBK (≥98.5% Sigma Aldrich) and base catalyst
(NaOH (≥98%), KOH (90%) and K2CO3 (99%) Sigma Aldrich, Na2CO3
(≥99.5%) Fischer and Ca(OH)2 (96%) Merck) were placed in this
order into an 8 ml glass vial with a magnetic stirring bar and the
vial was closed with a cap. The mixture was heated at appropriate
temperature in an oil bath with a thermostat and the stirring was
set to 900 rpm (See SI for more details). After the required time,
the reaction solution was cooled down to room temperature and
EtOAc (5 ml) was added, followed by washing with saturated aque-
ous NaCl-solution (2 ml). The washing solution was extracted with
ethyl acetate (EtOAc, 3 × 5 ml). The organic layers were combined
and the solvents were evaporated in vacuo to give the desired prod-
ucts 1 and 2 as dark-brownish oils. To determine the yields, the
products were dissolved in EtOAc (5 ml). From this solution, an
aliquot of 0.5 ml was taken, 0.1 ml of internal standard (acetophe-
none) was added and the resulting solution was diluted with EtOAc
(10 ml). The yields of 1 and 2 were determined from this solution
with GC-FID.
2.2. General Procedure for HDO of 1 and 2 to 3 and 4
Desired amount of substrate (1 or 2) was weighed and placed
into the glass insert with magnetic stirring bar. After this, appro-
priate amount of solvent, cocatalyst (HOTf (≥99%), Eu(OTf)3 (98%),
YbCl3 (99.9%), Nd(OTf)3 (98%), FeCl3 (97%) Sigma Aldrich; Hf(OTf)4
(98%), Fe(OTf)3 (90% tech.), EuCl3 (99.9%), Sc(OTf)3 (98%), Y(OTf)3,
LaCl3 (99.9%) and La(OTf)3 (99%) Alfa Aesar; InCl3 (98%) Fluka; and
AlCl3 (98.7%) Baker) and 0.4 mol-% of Pd/C (5 w-%, Sigma Aldrich)
were added. The glass insert was placed into the autoclave, which
was then closed and placed into a heating mantle (Roth 30 S). The
autoclave was pressurized with H2 to 40 bars (at room tempera-
ture) and heated at 200 ◦C for desired time (See SI for more details).
After heating, the reaction solution was cooled to room tempera-
ture, filtered and diluted with EtOAc or hexane (depending on the
solvent used) to 50 ml. From this solution, an aliquot of 1.25 ml was
taken, 0.1 ml of internal standard (acetophenone) was added and
the resulting solution was diluted with EtOAc or hexane (10 ml).
The yields of 3 and 4 were determined from this solution with
GC-FID.
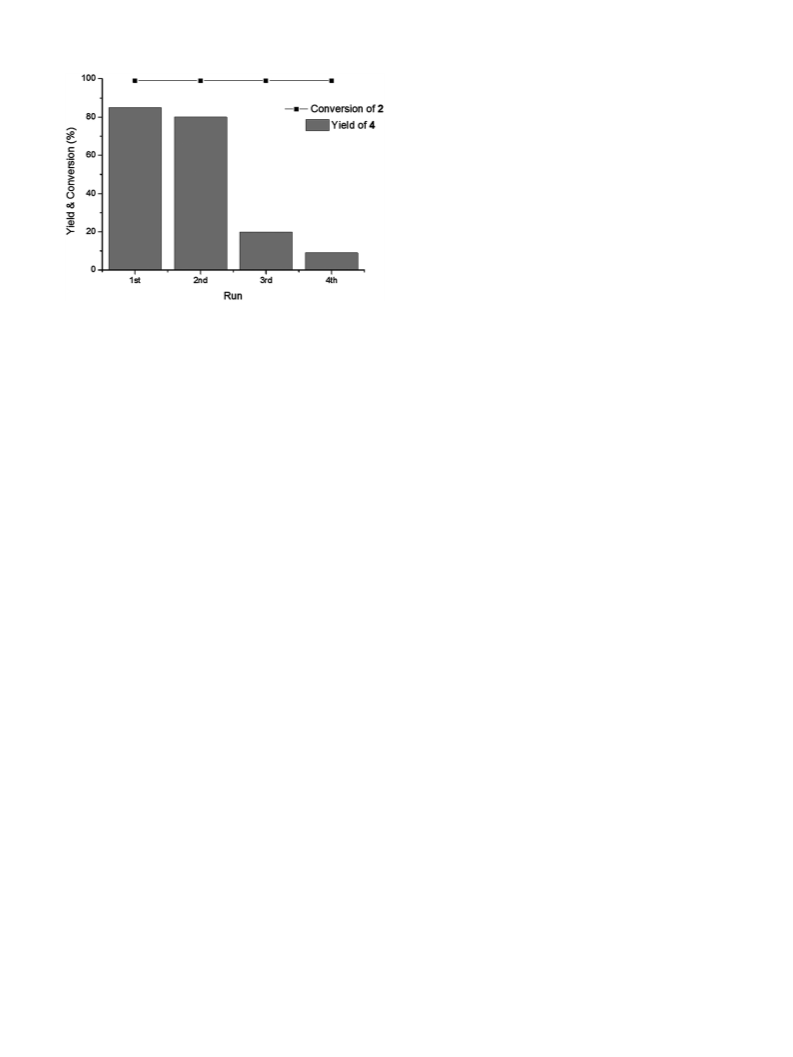
We investigated the one-step HDO of 1 and 2 using Pd/C and
metal triflates as catalysts. First, we studied Pathway A conditions
(Scheme 2) for the two-step HDO of both HMF-based 1 and furfural-
based 2 using AcOH as a solvent and La(OTf)3 as a cocatalyst [15].
Under these conditions alkanes 3 and 4 were obtained in fair yields
of 49% and 51% respectively. Interestingly, 1H NMR studies after
2.3. Analysis
The yields of aldol condensation products 1 and 2 as well as alka-
nes 3 and 4 were determined by a gas chromatograph equipped

 Keskiv?li, Juha
Keskiv?li, Juha
 Wrigstedt, Pauli
Wrigstedt, Pauli
 Lagerblom, Kalle
Lagerblom, Kalle
 Repo, Timo
Repo, Timo