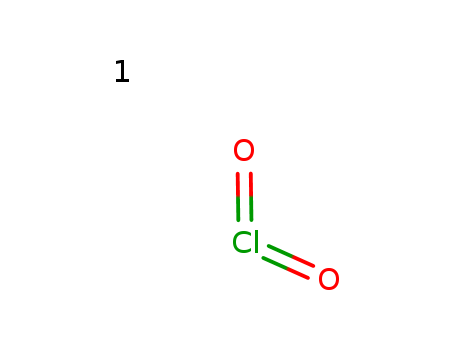
- Chemical Name:Chlorine Dioxide
- CAS No.:10049-04-4
- Deprecated CAS:1002158-67-9,1014743-76-0,1018807-91-4,1333-81-9,1428979-52-5,155808-17-6,56310-06-6,69049-77-0,70134-37-1,72061-90-6,74296-11-0,744149-53-9,77546-17-9,77546-18-0,807261-60-5,93085-22-4
- Molecular Formula:ClO2
- Molecular Weight:67.4518
- Hs Code.:
- European Community (EC) Number:233-162-8
- ICSC Number:0127
- UN Number:9191
- DSSTox Substance ID:DTXSID5023958
- Nikkaji Number:J342.197J
- Wikipedia:Chlorine dioxide,Chlorine_dioxide,Chlorine_peroxide
- NCI Thesaurus Code:C172619
- RXCUI:20799
- Mol file:10049-04-4.mol
Synonyms:Alcide;Aseptrol;chlorine dioxide;chlorine dioxide, 17O-labeled;chlorine dioxide, 35Cl,1-(18)O-labeled;chlorine dioxide, 35Cl-labeled;chlorine dioxide, 37Cl,1-(18)O-labeled;chlorine dioxide, 37Cl-labeled;chlorine dioxide, monohydrate;chlorine oxide;ClO2;DioxiClear;MicroClear;RenNew-D;Tristel


 O,
O, T+,
T+, N
N




