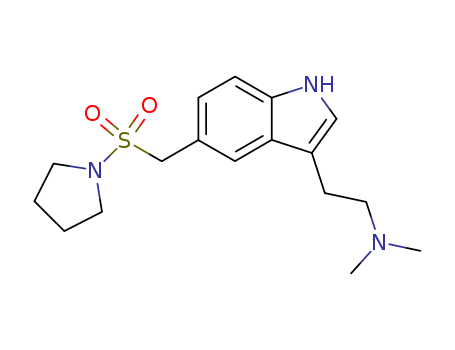
Chemical Property of Almotriptan
Chemical Property:
- Appearance/Colour:light yellow or white power
- Vapor Pressure:1.13E-11mmHg at 25°C
- Melting Point:170-172°C
- Boiling Point:538.7 °C at 760 mmHg
- PKA:16.92±0.30(Predicted)
- Flash Point:279.6 °C
- PSA:64.79000
- Density:1.27 g/cm3
- LogP:3.21630
- XLogP3:1.6
- Hydrogen Bond Donor Count:1
- Hydrogen Bond Acceptor Count:4
- Rotatable Bond Count:6
- Exact Mass:335.16674822
- Heavy Atom Count:23
- Complexity:483
- Purity/Quality:
-
99% *data from raw suppliers
Almotriptan *data from reagent suppliers
Safty Information:
- Pictogram(s):
- Hazard Codes:
- MSDS Files:
-
SDS file from LookChem
Useful:
- Canonical SMILES:CN(C)CCC1=CNC2=C1C=C(C=C2)CS(=O)(=O)N3CCCC3
- Recent ClinicalTrials:Evaluation of the Efficacy of Almotriptan and Ubrogepant for the Acute Treatment of Migraine
- Recent EU Clinical Trials:A Randomized, Parallel-Group, Single-Attack, Open-Label Study to Evaluate the Efficacy of Almotriptan and Ubrogepant for the Acute Treatment of Migraine (ATOM).
-
Description
Almotriptan was first marketed in Spain as a new medicine against acute attacks of
migraine. It is the fifth agent belonging to the “triptan” class to be launched after
sumatriptan, naratriptan, zolmitriptan and rizatriptan. This close structural analog of
sumatriptan can be prepared in six steps from 4-nitrobenzylsulfonyl chloride with a Fischer
indole synthesis as the key step. Almotriptan acts as a dual 5-HT1D/1B agonist with a 35 to
51-fold selectivity versus 5-HT1A and 5-HT7 receptors respectively as well as having
insignificant affinity for the most relevant nonserotonergic receptors (K1>1μM). Its
agonistic effect on 5-HT,n receptors of trigeminal sensory neurons turns off neurogenic
inflammation by inhibiting the release of neuropeptides such as calcitonin gene-related
peptide, neurokinin A and substance P. Concomitantly, its action on the 5-HT1B receptors
in meningeal arteries relieves the vasodilatation of these vessels associated with migraine
attacks. Almotriptan causes selective concentration-dependent vasoconstriction of human
meningeal and temporal arteries (with EC50 of 0.03 and 0.7 μM) compared to basilar (EC50
= 3.5 μM) and pulmonary arteries (EC50>10μM) or rabbit mesenteric and renal arteries
(EC50>100 μM). Although it is predominantly cleared by the kidneys as unchanged drug
(45%) or transformed into inactive metabolites by monoamine oxidase A (MAO-A) and
CYP3A4 enzymes in the liver, almotriptan has the highest oral bioavailability (70%) of the
triptans and has a half-life of 3.5 h. The therapeutic dose of 12.5 mg is well tolerated,
shows a rapid onset of action (30 min) and low recurrence rate compared to sumatriptan.
-
Uses
Serotonin 5HT1B /1D-receptor agonist
-
Therapeutic Function
Migraine therapy
-
Drug interactions
Potentially hazardous interactions with other drugs
Antidepressants: increased risk of CNS toxicity with
citalopram - avoid; possibly increased serotonergic
effects with duloxetine or venlafaxine; increased
serotonergic effects with St John’s wort - avoid.
Antifungals: concentration increased by ketoconazole
(increased risk of toxicity).
Dapoxetine: possible increased risk of serotonergic
effects - avoid for 2 weeks after stopping 5HT1
agonists.
Ergot alkaloids: increased risk of vasospasm - avoid.