10.1002/anie.201807642
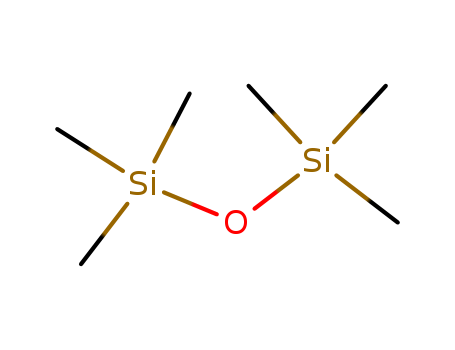
The research investigates the enhanced solubility of halide-containing organometallics in diiodomethane (CH2I2) compared to other haloalkane solvents. The study hypothesizes that the solvent's complex halogen bonding is responsible for the improved solubility. Experiments involved preparing and characterizing a series of palladium and platinum isocyanide complexes using techniques like CHN elemental analysis, high-resolution mass spectrometry, FT-IR, and NMR spectroscopy. Solubility was measured using quantitative 1H NMR spectroscopy with hexamethyldisiloxane as an internal standard. The researchers also calculated the electrostatic potential of the s-holes (VS(r)max) in the solvents to assess their halogen bonding ability. The results showed that CH2I2 had the highest solubility for the organometallic model compounds, suggesting that its strong s-hole donating ability leads to uniquely strong solvent-(metal complex) halogen bonding, which was further supported by crystal structure analyses and quantum chemical calculations.


 F
F




