Products Categories
| CAS No.: | 141-97-9 |
|---|---|
| Name: | Ethyl acetoacetate |
| Article Data: | 296 |
| Molecular Structure: | |
|
|
|
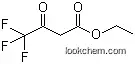
| Formula: | C6H10O3 |
| Molecular Weight: | 130.144 |
| Synonyms: | Ethyl beta-ketobutyrate;FEMA No. 2415;Acetoacetic ester;Ethyl 3-oxobutyrate;Active acetyl acetate;3-Oxobutanoic acid ethyl ester;Ethyl acetylacetate;Ethylester kyseliny acetoctove [Czech];Acetoctan ethylnaty [Czech];1-Ethoxybutane-1,3-dione;Active acetylacetate;Butanoic acid, 3-oxo-, ethyl ester;Ethyl 3-oxobutanoate; |
| EINECS: | 205-516-1 |
| Density: | 1.021 g/cm3 |
| Melting Point: | -43 °C |
| Boiling Point: | 180.6 °C at 760 mmHg |
| Flash Point: | 67 °C |
| Solubility: | water: 116 g/L (20 °C) |
| Appearance: | Colourless liquid |
| Hazard Symbols: |
 Xi Xi
|
| Risk Codes: | 36 |
| Safety: | 26-24/25 |
| Transport Information: | UN 1993 |
| PSA: | 43.37000 |
| LogP: | 0.52860 |
- 144851-82-1METHYL2-AMINO-3-FLUOROBENZOATE
- 483366-12-7(2S,4R)-1-Boc-2-cyano-4-hydroxypyrrolidine
- 173606-50-3BOC-10-AMINODECANOIC ACID
- 361456-36-2METHYL (R)-(+)-ISOCYANATO-3-PHENYLPROPI&
- 5156-58-1N-(1-Benzyl-4-pipperidinyl)-N-phenylpropanamide HCl
- 81281-59-67-Benzylideneaminotheophylline
- 50288-62-5threo-Phenyl-2-piperidyl acetamide
- 82993-81-5D-threo-Ritalinic acid hydrochloride
- 47087-37-6Z-D-Glu-OMe
- 73441-42-6METHYL-5-CHLORO-2,2-DIMETHYLVALERATE

| Conditions | Yield |
|---|---|
| With potassium carbonate In acetone for 1h; Ambient temperature; | 100% |

- 141-97-9
ethyl acetoacetate

| Conditions | Yield |
|---|---|
| With water at 20℃; under 900.09 Torr; for 0.226667h; Flow reactor; | 98% |

| Conditions | Yield |
|---|---|
| With zinc In tetrahydrofuran 1) 30 deg C, 2 h, 2) reflux, 18 h; | 97% |

| Conditions | Yield |
|---|---|
| With cerium(III) chloride; sodium iodide In acetonitrile for 1.5h; Heating; | 96% |
| With polyaniline-sulfate salt; water for 0.75h; Heating; | 90% |
| With boron trifluoride diethyl etherate; tetraethylammonium iodide In chloroform for 6h; Heating; | 69% |
| palladium (II) ion In acetone for 23h; Ambient temperature; | 97 % Chromat. |
- 92208-06-5
3,3-dimethoxybutyric acid ethyl ester

| Conditions | Yield |
|---|---|
| With water at 20℃; under 900.09 Torr; for 0.226667h; Flow reactor; | 96% |

| Conditions | Yield |
|---|---|
| With sulfuric acid at 85 - 125℃; for 6h; | 95% |
| With sulfuric acid | |
| With dmap |

| Conditions | Yield |
|---|---|
| With ytterbium(III) triflate at 110℃; for 3h; Neat (no solvent); | 94% |
| In neat (no solvent) at 110℃; for 3h; Green chemistry; | 92% |
| With zinc(II) sulfate In toluene at 80℃; for 6h; | 87% |

| Conditions | Yield |
|---|---|
| With lithium hexamethyldisilazane In tetrahydrofuran at -40℃; for 0.333333h; Claisen condensation; Inert atmosphere; | A 94% B n/a |

- 141-78-6
ethyl acetate

- 54013-04-6
ethyl 5-chloro-2-pyrazinecarboxylate

A
- 1215002-59-7
ethyl 3-(5-chloropyrazin-2-yl)-3-oxopropanoate

B
- 141-97-9
ethyl acetoacetate

| Conditions | Yield |
|---|---|
| With lithium hexamethyldisilazane In tetrahydrofuran at -50 - -30℃; for 0.333333h; Claisen condensation; Inert atmosphere; | A 94% B n/a |


| Conditions | Yield |
|---|---|
| Esterification; | 92% |
| In 5,5-dimethyl-1,3-cyclohexadiene Reflux; | 69% |
| In 5,5-dimethyl-1,3-cyclohexadiene at 150℃; |
- 13598-36-2Phosphonic acid
- 137071-32-0Pimecrolimus
- 77591-33-4Thymosin beta 4 acetate
- 24634-61-5Potassium sorbate
- 14897-39-3Rifamycin sodium salt
- 98-86-2Acetophenone
- 69-65-8D-Mannitol
- 107-22-2Glyoxal
- 104987-11-3Tacrolimus
- 141-53-7Sodium formate
- 8001-54-5Quaternary ammonium compounds, alkylbenzyldimethyl, chlorides
- 9003-39-8Povidone
- 10161-34-9Trenbolone acetate
- 402957-28-2Telaprevir
- 68-19-9Cyanocobalamin
Consensus Reports
Reported in EPA TSCA Inventory.
Specification
The Ethyl acetoacetate, with the CAS registry number 141-97-9, is also known as Acetoacetic ester; Ethyl 3-oxobutanoate; Ethyl beta-ketobutyrate; EAA. It belongs to the product categories of Carbonyl Compounds;Chemical Synthesis;E-H;Esters;Organic Building Blocks.Its EINECS number is 205-516-1.This chemical's molecular formula is C6H10O3 and molecular weight is 130.14. What's more,Its systematic name is Ethyl acetoacetate.It is a colorless liquid with a fruity odor.And it is stable,Incompatible with acids,bases,oxidizing agents,reducing agents,alkali metals,Combustible.The Ethyl acetoacetate may cause adverse health effects if ingested or inhaled, and it is irritate to skin, eyes and mucous membranes.When you use it ,you should avoid contact with skin and eyes. In case of contact with eyes, rinse immediately with plenty of water and seek medical advice.It should Store in a tightly closed container,in a cool,dry place.Keep away from sources of ignition.
Physical properties about Ethyl acetoacetate are:
(1)ACD/LogP: 0.716; (2)# of Rule of 5 Violations: 0; (3)ACD/LogD (pH 5.5): 0.72; (4)ACD/LogD (pH 7.4): 0.72; (5)ACD/BCF (pH 5.5): 2.06; (6)ACD/BCF (pH 7.4): 2.06; (7)ACD/KOC (pH 5.5): 58.42; (8)ACD/KOC (pH 7.4): 58.39; (9)#H bond acceptors: 3; (10)#H bond donors: 0; (11)#Freely Rotating Bonds: 3; (12)Index of Refraction: 1.408; (13)Molar Refractivity: 31.651 cm3; (14)Molar Volume: 128.178 cm3; (15)Surface Tension: 30.2719993591309 dyne/cm; (16)Density: 1.015 g/cm3; (17)Flash Point: 67.019 °C; (18)Enthalpy of Vaporization: 41.683 kJ/mol; (19)Boiling Point: 180.567 °C at 760 mmHg; (20)Vapour Pressure: 0.889999985694885 mmHg at 25°C.
Preparation of Ethyl acetoacetate:
Ethyl acetoacetate is produced industrially by treatment of diketene with ethanol which is a classic laboratory procedure. It is prepared via the Claisen condensation of ethyl acetate. Two moles of ethyl acetate condense to form one mole each of ethyl acetoacetate and ethanol.

Uses of Ethyl acetoacetate:
Ethyl acetoacetate is used as starting material for the syntheses of alpha-substituted acetoacetic esters and cyclic compounds, e.g. pyrazole, pyrimidine and coumarin derivatives as well as intermediate for vitamins and pharmaceuticals.
You can still convert the following datas into molecular structure:
(1)SMILES:CCOC(=O)CC(=O)C;
(2)Std. InChI:InChI=1S/C6H10O3/c1-3-9-6(8)4-5(2)7/h3-4H2,1-2H3;
(3)Std. InChIKey:XYIBRDXRRQCHLP-UHFFFAOYSA-N.
The toxicity data of Ethyl acetoacetate as follows:
| Organism | Test Type | Route | Reported Dose (Normalized Dose) | Effect | Source |
|---|---|---|---|---|---|
| mouse | LD50 | oral | 5105mg/kg (5105mg/kg) | Journal of Pharmaceutical Sciences. Vol. 60, Pg. 1810, 1971. | |
| rabbit | LD | skin | > 20mL/kg (20mL/kg) | Union Carbide Data Sheet. Vol. 3/12/1969, | |
| rat | LD50 | oral | 3980mg/kg (3980mg/kg) | Journal of Industrial Hygiene and Toxicology. Vol. 31, Pg. 60, 1949. |