Products Categories
| CAS No.: | 21462-39-5 |
|---|---|
| Name: | Clindamycin hydrochloride |
| Article Data: | 3 |
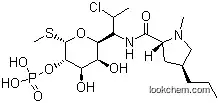
| Molecular Structure: | |
|
|
|
| Formula: | C18H33ClN2O5S.HCl |
| Molecular Weight: | 461.45 |
| Synonyms: | 7(S)-Chloro-7-deoxylincomycin hyddrochloride;L-threo-a-D-galacto-Octopyranoside, methyl7-chloro-6,7,8-trideoxy-6-[[[(2S,4R)-1-methyl-4-propyl-2-pyrrolidinyl]carbonyl]amino]-1-thio-,hydrochloride (1:1);7-Chloro-7-deoxylincomycin hydrochloride;Antirobe vet.; |
| EINECS: | 244-398-6 |
| Density: | == |
| Melting Point: | 141 °C |
| Boiling Point: | 647 °C at 760 mmHg |
| Flash Point: | 345.1 °C |
| Solubility: | water: 50 mg/mL, clear, colorless |
| Appearance: | white crystalline powder |
| Hazard Symbols: |
 Xi Xi
|
| Risk Codes: | 36/37/38 |
| Safety: | 26-36-37/39 |
| PSA: | 127.56000 |
| LogP: | 1.52030 |
- 483366-12-7(2S,4R)-1-Boc-2-cyano-4-hydroxypyrrolidine
- 361456-36-2METHYL (R)-(+)-ISOCYANATO-3-PHENYLPROPI&
- 5156-58-1N-(1-Benzyl-4-pipperidinyl)-N-phenylpropanamide HCl
- 81281-59-67-Benzylideneaminotheophylline
- 50288-62-5threo-Phenyl-2-piperidyl acetamide
- 82993-81-5D-threo-Ritalinic acid hydrochloride
- 47087-37-6Z-D-Glu-OMe
- 73441-42-6METHYL-5-CHLORO-2,2-DIMETHYLVALERATE
- 240444-70-62,3,5,6-Tetrafluor-4-(methoxymethyl)benzyl-2,2-dimethyl-3-(prop-1-en-1-yl)cyclopropancarboxylat
- 68439-39-4Poly(oxy-1,2-ethanediyl), alpha-(2-ethylhexyl)-omega-hydroxy-,
- 16684-06-3, 18323-44-9, 51064-35-8
clindamycin

- 21462-39-5
clindamycin hydrochloride

| Conditions | Yield |
|---|---|
| With hydrogenchloride In ethanol at 60℃; for 1h; pH=1; pH-value; Temperature; Large scale; | 89.5% |
- 21462-39-5
clindamycin hydrochloride

| Conditions | Yield |
|---|---|
| With dihydrogen peroxide In water at 10℃; for 2h; | 97.4% |

| Conditions | Yield |
|---|---|
| Stage #1: clindamycin hydrochloride With sodium hydroxide In water Stage #2: vinyl palmitate In toluene at 20 - 40℃; for 12.5h; Reagent/catalyst; Solvent; Temperature; Enzymatic reaction; regioselective reaction; | 90% |
- 108-24-7
acetic anhydride

- 21462-39-5
clindamycin hydrochloride

- 2150-02-9
bis(2-mercaptoethyl)ether

- 1446499-11-1
C30H50N2O10S3

| Conditions | Yield |
|---|---|
| Stage #1: clindamycin hydrochloride; bis(2-mercaptoethyl)ether With caesium carbonate In N,N-dimethyl-formamide at 50℃; for 12h; Stage #2: acetic anhydride With pyridine; dmap | 87% |

- 7442-07-1
6-amino-2-mercaptobenzothiazole

- 21462-39-5
clindamycin hydrochloride

| Conditions | Yield |
|---|---|
| With potassium carbonate In N,N-dimethyl-formamide at 100℃; for 16h; | 41% |
| With potassium carbonate In N,N-dimethyl-formamide at 90℃; for 16h; | 40% |
- 21462-39-5
clindamycin hydrochloride

| Conditions | Yield |
|---|---|
| With sodium hydride In N,N-dimethyl-formamide at 100℃; for 2.5h; Dehydrochlorination; | 36.28% |
- 21462-39-5
clindamycin hydrochloride

| Conditions | Yield |
|---|---|
| With sodium perborate pH=~ 8 - 9; | |
| With (Dichloroiodo)benzene |
- 21462-39-5
clindamycin hydrochloride

| Conditions | Yield |
|---|---|
| With dihydrogen peroxide; sodium tungstate pH=< 1; |
- 21462-39-5
clindamycin hydrochloride

- 24729-96-2
clindamycin phosphate
- 21462-39-5
clindamycin hydrochloride

- 1446499-23-5
C33H54N4O11S3

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: caesium carbonate / N,N-dimethyl-formamide / 12 h / 50 °C 2: caesium carbonate; tetra-(n-butyl)ammonium iodide / tetrahydrofuran / 16 h / 40 °C View Scheme | |
| Multi-step reaction with 3 steps 1: caesium carbonate / N,N-dimethyl-formamide / 12 h / 50 °C 2: sodium methylate; methanol / 3 h 3: caesium carbonate; tetra-(n-butyl)ammonium iodide / tetrahydrofuran / 16 h / 40 °C View Scheme |
- 866-84-2Potassium citrate
- 94-62-2Piperine
- 141109-19-5D-(+)-Methyl-alpha-(2-thienylethamino)(2-chlorophenyl)acetate hydrochloride
- 7784-24-9Aluminium potassium sulfate dodecahydrate
- 108-30-52,5-Furandione,dihydro-
- 36687-82-8L-Carnitine-L-tartrate
- 98-16-83-Aminobenzotrifluoride
- 161735-79-1Rasagiline mesylate
- 104987-11-3Tacrolimus
- 141-53-7Sodium formate
- 8001-54-5Quaternary ammonium compounds, alkylbenzyldimethyl, chlorides
- 9003-39-8Povidone
- 10161-34-9Trenbolone acetate
- 402957-28-2Telaprevir
- 68-19-9Cyanocobalamin

What can I do for you?
Get Best Price
Specification
The IUPAC name of Clindamycin HCl is (2S,4R)-N-[2-chloro-1-[(2R,3R,4S,5R,6R)-3,4,5-trihydroxy-6-methylsulfanyloxan-2-yl]propyl]-1-methyl-4-propylpyrrolidine-2-carboxamide hydrochloride. With the CAS registry number 21462-39-5, it is also named as 7-Chloro-7-deoxylincomycin hydrochloride. The product's categories are Antibiotic Explorer; Peptide Synthesis / Antibiotics. It is white crystalline powder with bitter taste which is easily soluble in water, soluble in methanol, pyridine, slightly soluble in ethanol, practically insoluble in acetone or chloroform. Additionally, this chemical should be stored at the temperature of 2-8 °C.
The other characteristics of this product can be summarized as: (1)ACD/LogP: 1.83; (2)# of Rule of 5 Violations: 0; (3)ACD/LogD (pH 5.5): -1; (4)ACD/LogD (pH 7.4): 1; (5)ACD/BCF (pH 5.5): 1; (6)ACD/BCF (pH 7.4): 1; (7)ACD/KOC (pH 5.5): 1; (8)ACD/KOC (pH 7.4): 20; (9)#H bond acceptors: 7; (10)#H bond donors: 4; (11)#Freely Rotating Bonds: 10; (12)Enthalpy of Vaporization: 109.28 kJ/mol; (13)Vapour Pressure: 1.79E-19 mmHg at 25°C; (14)Rotatable Bond Count: 7; (15)Tautomer Count: 2; (16)Exact Mass: 460.156548; (17)MonoIsotopic Mass: 460.156548; (18)Topological Polar Surface Area: 128; (19)Heavy Atom Count: 28; (20)Complexity: 502; (21)Defined Atom StereoCenter Count: 7; (22)Undefined Atom StereoCenter Count: 2.
Preparation and Uses of Clindamycin HCl: It is a derivative of lincomycin. And it is semi-synthetic antibiotics obtained from the replacement of lincomycin 7 hydroxyl by chlorine atom. The bactericidal activity to staphylococcus aureus and pneumococcal diseaseis is many times stronger than Lincomycin. So Clindamycin HCl is an antibiotic class medicine. The antimicrobial spectrum is similar to lincomycin.
When you are using this chemical, please be cautious about it as the following:
It is irritating to eyes, respiratory system and skin. In case of contact with eyes, rinse immediately with plenty of water and seek medical advice. If you want to contact this product, you must wear suitable protective clothing, gloves and eye/face protection.
People can use the following data to convert to the molecule structure.
1. SMILES:Cl.O[C@@H]1[C@@H](O)[C@@H](O)[C@H](O[C@@H]1SC)[C@H](NC(=O)[C@@H]2C[C@@H](CCC)CN2C)[C@H](C)Cl
2. InChI:InChI=1/C18H33ClN2O5S.ClH/c1-5-6-10-7-11(21(3)8-10)17(25)20-12(9(2)19)16-14(23)13(22)15(24)18(26-16)27-4;/h9-16,18,22-24H,5-8H2,1-4H3,(H,20,25);1H/t9-,10+,11-,12+,13-,14+,15+,16+,18+;/m0./s1
The following are the toxicity data which has been tested.
| Organism | Test Type | Route | Reported Dose (Normalized Dose) | Effect | Source |
|---|---|---|---|---|---|
| mouse | LD50 | intramuscular | 1100mg/kg (1100mg/kg) | BEHAVIORAL: ALTERED SLEEP TIME (INCLUDING CHANGE IN RIGHTING REFLEX) BEHAVIORAL: SOMNOLENCE (GENERAL DEPRESSED ACTIVITY) BEHAVIORAL: ATAXIA | Toho Igakkai Zasshi. Journal of Medical Society of Toho University. Vol. 18, Pg. 354, 1971. |
| mouse | LD50 | intraperitoneal | 361mg/kg (361mg/kg) | "Compounds Available for Fundamental Research, Volume II-6, Antibiotics, A Program of Upjohn Company Research Laboratory." Vol. 2(6), Pg. -, 1971. | |
| mouse | LD50 | intravenous | 245mg/kg (245mg/kg) | BEHAVIORAL: SOMNOLENCE (GENERAL DEPRESSED ACTIVITY) BEHAVIORAL: CONVULSIONS OR EFFECT ON SEIZURE THRESHOLD | Toxicology and Applied Pharmacology. Vol. 21, Pg. 516, 1972. |
| mouse | LD50 | oral | 2539mg/kg (2539mg/kg) | BEHAVIORAL: SOMNOLENCE (GENERAL DEPRESSED ACTIVITY) | Toho Igakkai Zasshi. Journal of Medical Society of Toho University. Vol. 18, Pg. 354, 1971. |
| mouse | LD50 | subcutaneous | 1036mg/kg (1036mg/kg) | BEHAVIORAL: SOMNOLENCE (GENERAL DEPRESSED ACTIVITY) LUNGS, THORAX, OR RESPIRATION: DYSPNEA BEHAVIORAL: CONVULSIONS OR EFFECT ON SEIZURE THRESHOLD | Toho Igakkai Zasshi. Journal of Medical Society of Toho University. Vol. 18, Pg. 354, 1971. |
| rat | LD50 | intramuscular | 273mg/kg (273mg/kg) | Drugs in Japan Vol. -, Pg. 331, 1990. | |
| rat | LD50 | intraperitoneal | 745mg/kg (745mg/kg) | BEHAVIORAL: SOMNOLENCE (GENERAL DEPRESSED ACTIVITY) | Toho Igakkai Zasshi. Journal of Medical Society of Toho University. Vol. 18, Pg. 354, 1971. |
| rat | LD50 | oral | 2193mg/kg (2193mg/kg) | BEHAVIORAL: SOMNOLENCE (GENERAL DEPRESSED ACTIVITY) | Toho Igakkai Zasshi. Journal of Medical Society of Toho University. Vol. 18, Pg. 354, 1971. |
| rat | LD50 | subcutaneous | 2618mg/kg (2618mg/kg) | BEHAVIORAL: SOMNOLENCE (GENERAL DEPRESSED ACTIVITY) BEHAVIORAL: CONVULSIONS OR EFFECT ON SEIZURE THRESHOLD | Toxicology and Applied Pharmacology. Vol. 21, Pg. 516, 1972. |