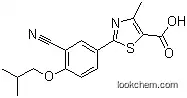
144060-53-7 Usage
Description
Febuxostat is a potent, non-purine compound that acts as an antihyperuricemic agent and a selective xanthine oxidase inhibitor. It is characterized by its ability to inhibit the expression of cytokines/chemokines and has been reported to inhibit LPS-induced TNF-α, VCAM-1, MMP9, and MCP-1 expression. Febuxostat is more potent and selective than allopurinol for inhibiting xanthine oxidase, making it a valuable pharmaceutical candidate for the treatment of hyperuricemia and chronic gout.
Uses
Used in Pharmaceutical Industry:
Febuxostat is used as an antihyperuricemic agent for the treatment of hyperuricemia and chronic gout. It effectively lowers serum urate levels when administered at a dosage of 40-120 mg/day, managing hyperuricemia in patients with gout.
Used in Gout Management:
Febuxostat is used as a xanthine oxidase/xanthine dehydrogenase inhibitor for the chronic management of hyperuricemia in patients with gout. It helps in reducing serum uric acid concentrations below saturation levels, leading to crystal dissolution and eventual disappearance.
Used in Hyperlipidemia Treatment:
Febuxostat is also used as an antihyperlipidemic agent, contributing to the management of hyperlipidemia in patients.
Chemical Properties:
Febuxostat is a crystalline solid with the brand names Uloric and Adenuric.
Indications and Usage
Febuxostat is a new generation xanthine oxidase inhibitor developed by Tejin Co. (Japan,) used clinically for for long-term treatment of hyperuicemia (gout,) a new and highly effective non-purine selective inhibitor of xanthine oxidase. It is not recommended for gout patients without hyperuricemia.
Mechanisms of Action
The production of uric acid in the body is related to purine metabolism. In the last step of the production, hypoxanthine produces xanthine through the action of xanthine oxidoreductase (XOR), finally producing uric acid. Inhibiting the activity of this enzyme can effectively reduce the production of uric acid. Xanthine oxidase is the main enzyme promoting uric acid production. Through highly selective inhibition of oxidized and reduced xanthine oxidase, Febuxostat can reduce synthesis of uric acid, decreasing its concentration and effectively treating gout . Through liver metabolism, Xanthine oxidase does not rely on renal excretion, so patients with moderate to severe liver and kidney dysfunction do not need to reduce dosages. Febuxostat is a non-purine XOR inhibitor, so it is very safe.
Originator
Teijin (Japan)
Biochem/physiol Actions
Febuxostat is a potent non-purine xanithine oxidase inhibitor. Febuxostat is used in urate lowering therapies (ULTs) for the treatment of gout.
Clinical Use
Fabuxostat was discovered by Teijin Pharmaceuticals and
licensed to TAP Pharmaceuticals (which is currently part of Takeda
Pharmaceuticals) and was approved in the U.S. for the treatment of
hyperuricemia in patients with gout. It is a once-daily non-purine
based agent with potent inhibitory activity against xanthine oxidase.
The safety profile of the drug also does not require dose
adjustment for patients with mild to moderate renal or hepatic
impairment. Febuxostat is the first new agent cleared for this indication
in 40 years.
Synthesis
There are a number of routes available to
prepare this agent as discussed in recent publications. The synthesis
shown in Scheme 10 is a short and concise route and does
not require the use of toxic reagents. Thus the commercially
available and easily prepared 4-hydroxythiobenzamide (52) was
reacted with ethyl bromoacetoacetate (53) in refluxing ethanol to
provide the thiazole ester 54 in ≈60% yield after crystallization.
The phenolic ester 54 was then treated with hexamethylenetetramine
(HMTA) in polyphosphoric acid at 80 °C to provide the crude aldehyde 55 (74% conversion by HPLC). Reaction of phenol 55 and
isobutyl bromide (56) in the presence of potassium carbonate with
catalytic potassium iodide in DMF gave isobutyl ether 57 (64%, two
steps). This ether was then converted in one pot to nitrile 58 in 93%
by reacting the aldehyde with hydroxylamine hydrochloride and
sodium formate in refluxing formic acid. Saponification of the ester
58 with aqueous sodium hydroxide provided fabuxostat (X).
Drug interactions
Potentially hazardous interactions with other drugs
Azathioprine: avoid concomitant use, increased risk
of neutropenia.
Cytotoxics: avoid concomitant use with
mercaptopurine.
Theophylline: use with caution
Metabolism
Extensively metabolised by conjugation via the uridine
diphosphate glucuronosyltransferase (UDPGT) enzyme
system, and by oxidation via the cytochrome P450
isoenzyme system to form active metabolites. About 49%
of a dose is excreted via the urine, and 45% via the faeces
(12% as unchanged drug)
Check Digit Verification of cas no
The CAS Registry Mumber 144060-53-7 includes 9 digits separated into 3 groups by hyphens. The first part of the number,starting from the left, has 6 digits, 1,4,4,0,6 and 0 respectively; the second part has 2 digits, 5 and 3 respectively.
Calculate Digit Verification of CAS Registry Number 144060-53:
(8*1)+(7*4)+(6*4)+(5*0)+(4*6)+(3*0)+(2*5)+(1*3)=97
97 % 10 = 7
So 144060-53-7 is a valid CAS Registry Number.
InChI:InChI=1/C16H16N2O3S/c1-9(2)8-21-13-5-4-11(6-12(13)7-17)15-18-10(3)14(22-15)16(19)20/h4-6,9H,8H2,1-3H3,(H,19,20)
144060-53-7Relevant articles and documents
A facile one-pot synthesis of 4-alkoxy-1,3-benzenedicarbonitrile
Hasegawa, Masaichi
, p. 857 - 864 (1998)
2-(3-Cyano-4-isobutoxyphenyl)-4-methylthiazole-5-carboxlic acid (TEI-6720) was prepared. The introduction of cyano group to 4-nitrobenzonitrile with KCN in dry DMSO followed by quenching with alkyl halide afforded the key intermediates, 4-alkoky-1,3-benzenedicarbonitriles, in good yield. The reaction was completed in dry DMSO, while no reaction occurred in dry DMF. This observation can be suggested by the participation of DMSO in the reaction.
New preparation method of febuxostat intermediate
-
, (2020/03/06)
The invention relates to a new preparation method of a febuxostat intermediate. The method includes: taking cheap 4-hydroxybenzaldehyde as an initial raw material, firstly preparing aldoxime from 4-hydroxybenzaldehyde and hydroxylamine hydrochloride, then adding a corresponding thio reagent, and preparing a compound 4-hydroxythiobenzamide (152A1-00) by Beckmann rearrangement reaction; utilizing one-pot process, adopting cheap 4-hydroxybenzaldehyde as an initial raw material, carrying out a series of reactions, and then performing cyclization with 2-halogenated ethyl acetoacetate to obtain ethyl 2-(4-hydroxyphenyl)-4-methyl-5-thiazolecarboxylate or different salt forms (152A2x) thereof; and using isobutyl sulfonate (152H1x) with more easily controllable quality to replace bromo-isobutane soas to prepare ethyl 2-(3-formyl-4-isobutoxyphenyl)-4-methyl-5-thiazolecarboxylate (152A4-00). In conclusion, the method provided by the invention is more beneficial to safe, simple and cost-efficientindustrial scale preparation of the febuxostat intermediate with higher purity.
Febuxostat and intermediates and synthesis thereof
-
, (2020/05/09)
The invention relates to febuxostat and intermediates and synthesis thereof, in particular to a method for synthesizing 2-(3-formyl-4-hydroxyphenyl)-4-methyl-5-thiazole ethyl formate, which comprisesthe following operation steps: (1) adding a reactant 2-(4-hydroxyphenyl)-4-methyl-5-thiazole ethyl formate into a mixture of polyphosphoric acid and methanesulfonic acid, and uniformly stirring the materials; (2) adding a Darf reaction reagent hexamethylenetetramine into the reaction mixture while stirring, continuously reacting, and cooling; and (3) adding saturated brine ice, separating out solid, filtering, cleaning the solid with water to-be-neutral, and drying to obtain the product. The invention also relates to 2-(3-formyl-4-hydroxyphenyl)-4-methyl-5-thiazole ethyl formate and to the usethereof for the preparation of febuxostat. The method has excellent performance.