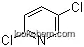
16110-09-1Relevant articles and documents
Continuous production process of 2, 3, 5-trichloropyridine
-
Paragraph 0015; 0018, (2021/10/30)
According to a continuous production process of 2, 3, 5-trichloropyridine, a continuous tubular chlorinator is adopted, pyridine is taken as a starting raw material, chlorination reaction is continuously carried out step by step by virtue of a catalyst 1 and a catalyst 2 which are independently researched and developed, and 2, 5-dichloropyridine and 2, 3, 5-trichloropyridine are prepared in sequence. In the preparation process of a catalyst 1 and a catalyst 2, the mass ratio of 1-chloropropane to imidazole to 2-chloroethyl diphenylphosphine to nickel tetracarbonyl is 1.2: 1: 3.7: 2.5, and the mass ratio of 1-chloropropane to imidazole to 2-chloroethyl diphenylphosphine to sodium hexachlororhodate hydrate is 1.2: 1: 3.7: 15. By means of the continuous tubular chlorinator, cheap and easily available pyridine is used as a starting raw material, chlorine atoms are introduced step by step through catalytic chlorination, the atom economy is good, the reaction selectivity is good, the yield of 2, 3, 5-trichloropyridine can be stabilized at 80% or above, the production efficiency is high, the process is easy to control, and the method is environmentally friendly and suitable for industrial production.
Method for preparing 3,6-dichloro-pyridine-2-carboxylic acid through hydrogenation reduction of 3,4,5,6-tetrachloropyridine-2-carboxylic acid
-
Paragraph 0054; 0055; 0056; 0057, (2016/12/01)
The invention discloses a method for preparing 3,6-dichloro-pyridine-2-carboxylic acid through hydrogenation reduction of 3,4,5,6-tetrachloropyridine-2-carboxylic acid and belongs to the technical field of production of chemical products. Under specific conditions of temperature, pressure, PH value and the like, chlorine on a pyridine ring is removed through catalytic hydrogenation and the 3,6-dichloro-pyridine-2-carboxylic acid is prepared. According to the method, a clean and environment-friendly catalytic hydrogenation process is used, the 3,4,5,6-tetrachloropyridine-2-carboxylic acid is subjected to hydrogenation reduction high-selectively to prepare the 3,6-dichloro-pyridine-2-carboxylic acid, highly toxic dangerous goods such as reducing agent hydrazine and dichloromethane are not used in the production process, the reaction conditions are mild, the device is simple and easy and convenient to operate, the cost is low, production zero release of the 3,6-dichloro-pyridine-2-carboxylic acid is realized, popularization and application of the green chemical technology are accelerated, and the method has remarkable economic advantages and environment-friendly advantages.
Copper-catalyzed conversion of aryl and heteroaryl bromides into the corresponding chlorides
Feng, Xiujuan,Qu, Yiping,Han, Yanlei,Yu, Xiaoqiang,Bao, Ming,Yamamoto, Yoshinori
supporting information, p. 9468 - 9470 (2012/10/29)
An efficient method for the synthesis of aryl and heteroaryl chlorides is described. The reactions of aryl and heteroaryl bromides with tetramethylammonium chloride proceeded smoothly in the presence of a copper catalyst under mild reaction conditions to produce the corresponding chlorides in satisfactory to excellent yields.