182431-12-5 Usage
Description
Lomitapide was approved by the US FDA in December 2012 for the treatment
of patients with familial hypercholesteremia (referred to as HoFH) in conjunction with a low-fat diet andother lipid-lowering treatments. Lomitapide was discovered from a high-through put
screen that identified several structurally distinct MTP inhibitors. Combination
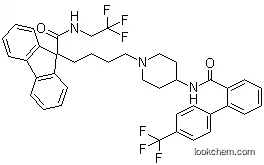
of key structural features from two structurally distinct HTS hits provided potent MTP inhibitors. Parallel analog synthesis led to lomitapide as an optimized structure. Lomitapide was synthesized via alkylation of 9-fluorenylcarboxylic acid with 1,4-dibromobutane which, after trifluoroethylamide formation, provided a bromide intermediate that was displaced by Boc-4-aminopiperidine. Introduction of the 4'-trifluoromethylbiphenylcarboxamide gave lomitapide, which
was found to inhibit MTP with an IC50 of 0.5 nM and to exhibit good
cholesterol-lowering efficacy in Sprague–Dawley rats (intravenous and oral
ED50~0.2 mg/kg).
Originator
Bristol-Myers Squibb (United States)
Uses
Lomitapide has been used as a microsomal triglyceride transfer protein (MTP) inhibitor to study its effects on very-low-density lipoproteins (VLDL) export in mouse hepatocytes.
Brand name
Juxtapid
Biochem/physiol Actions
Lomitapide is an inhibitor of microsomal triglyceride transfer protein (MTP). Lomitapide has been shown to be highly effective in reducing LDL-cholesterol and triglycerides, and has been aproved for treatment of homozygous familial hypercholesterolemia.
Check Digit Verification of cas no
The CAS Registry Mumber 182431-12-5 includes 9 digits separated into 3 groups by hyphens. The first part of the number,starting from the left, has 6 digits, 1,8,2,4,3 and 1 respectively; the second part has 2 digits, 1 and 2 respectively.
Calculate Digit Verification of CAS Registry Number 182431-12:
(8*1)+(7*8)+(6*2)+(5*4)+(4*3)+(3*1)+(2*1)+(1*2)=115
115 % 10 = 5
So 182431-12-5 is a valid CAS Registry Number.
182431-12-5Relevant articles and documents
CRYSTALLINE POLYMORPH OF N-(2,2,2-TRIFLUOROETHYL-9-[4-[R4-R[[[I4'- (TRIFLUOROMETHYL) [ 1,1 ' -BIPHENYL] -2-YL] CARBONYL] AMINO] -1 -PIPERIDINYL] BUTYL] -9H- FLUORENE-9-CARBOXAMIDE METHANESULFONATE AND PROCESS FOR PREPARATION THEREOF
-
, (2017/07/06)
The present invention relates to? crystalline polymorph of N-(2,2.2-trifluoroethyl)-9- [4- [4- [ [[4,-(trifluoromethyl) [1,1 '-biphenyl] -2-yl] carbonyl] amino] - 1 -piperidinyl] butyl] -9H- fluorene-9-carboxamide methanesulfonate salt represented by the following structural formula- la and process for preparation thereof.
AMORPHOUS FORM OF LOMITAPIDE MESYLATE
-
, (2016/05/02)
Provided is novel amorphous form of lomitapide mesylate salt and process for preparation thereof.
Method for synthesizing triglyceride transfer protease inhibitor
-
, (2016/12/26)
The invention discloses a method for synthesizing the triglyceride transfer protease inhibitor Lomitapide. Specifically, based on improvement of an existing technique, the compounds 9-carboxyfluorene and 1,4-dibromobutane are used as raw materials, five-step reaction is conducted, and the defects of the prior art are overcome. Compared with the prior art, the method has the main advantage that column chromatography purification is avoided, and product purification is conducted with a recrystallization method which is economical and environment-friendly.