M. Olivares, M. Albrecht / Journal of Organometallic Chemistry 920 (2020) 121290
5
ꢀ
DCB at 150 C for 5 min before adding the alcohol substrate decreased
(1.5 mL) and stirred according to the general procedure to obtain
complex 5 as a yellow solid. Yield 28 mg, 49%. H NMR (300 MHz,
1
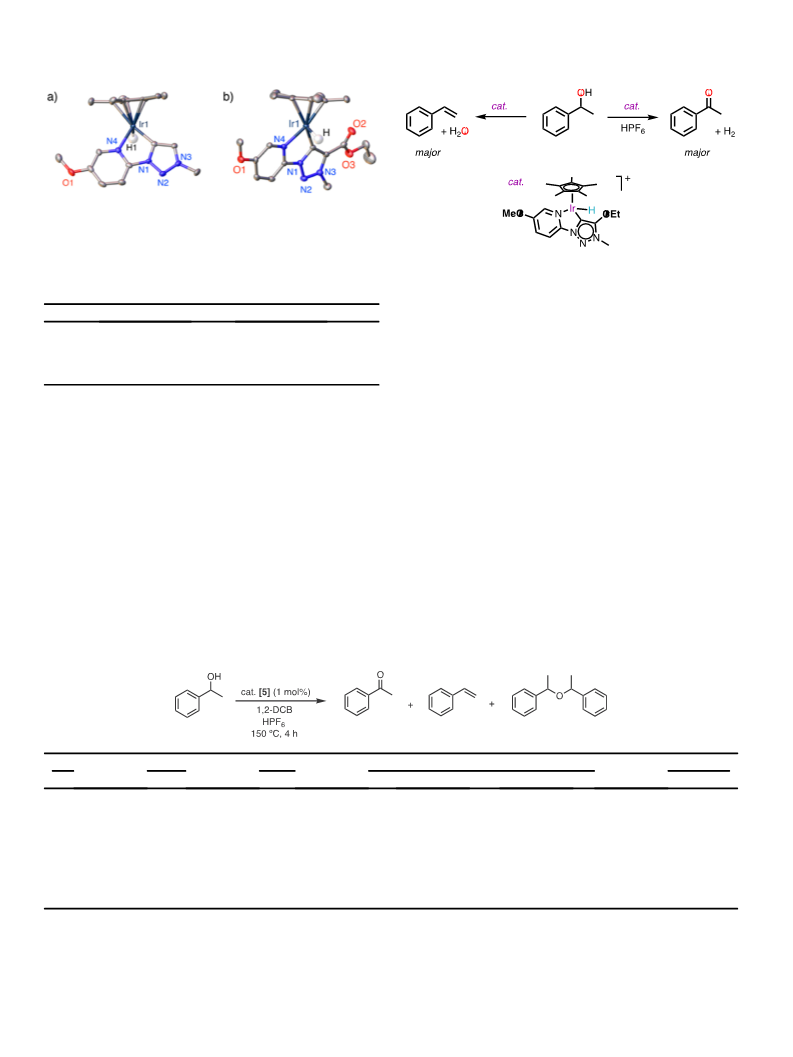
the formation of ketone, presumably because the formed cationic
species is not sufficiently stabilized (entries 6, 7). Instead, HPF
thermally decompose to HF and PF [40], which is supposed to pro-
duce the analogous IreF complex with selectivity towards styrene
production analogous to complex 1. A control experiment using the
3
6
may
CD
3
CN):
d
¼ 8.35 (s, 1H, CpyH), 7.99 (d, JHH ¼ 9.0 Hz, 1H, CpyH), 7.69
3
5
(d, JHH ¼ 9.0 Hz, 1H, CpyH), 4.45e4.13 (m, 2H, OCH
2
Me), 4.07 (s, 3H,
NCH
3
), 3.99 (s, 3H, OCH
HH ¼ 7.3 Hz, 3H, OCH CH
(75 MHz, CD CN):
(CpyeNtrz), 139.9 (CpyH), 138.9 (CtrzeOEt), 126.2 (CpyH), 114.5 (CpyH),
92.8 (Cp*), 73.6 (OCH Me), 57.6 (OCH ), 35.7 (NCH ), 15.2
(OCH CH ), 10.4 (CpeCH ). HR-MS (CH CN): m/z calculated for
Ir [MeOTf] ¼ 563.1993; found, 563.1999.
Complex 6: Complex 2 (60 mg, 0.085 mmol) and HSiEt
0.85 mmol) were stirred according to the general procedure to
3
), 1.99 (s, 15H, CpeCH ), 1.42 (t,
3
3
13
1
J
2
3
), ꢁ13.86 (s, 1H, IreH). C{ H} NMR
iridium chloride complex 1 in presence of HPF
6
afforded almost
3
d
¼ 157.7 (Cpy eOMe), 156.9 (CtrzeIr), 145.7
exclusively styrene with only traces of ketone (<2%, entry 8), indi-
cating that halide coordination is prohibiting alcohol dehydrogena-
tion. In addition, the distinct selectivity of complexes 1 and 5 under
acidic conditions reveals the relevance of the hydride and hydride
activation for shifting the selectivity towards dehydrogenation.
Addition of the acid in portions, viz. 2 eq at the beginning and 2
further eq after 1 h, slows down substrate conversion considerably
and produced only 61% conversion after 4 h, indicating substantial
catalyst deactivation even though selectivity towards dehydroge-
nation remained at 56% (entry 9), i.e. the same level as when adding
mol% acid at the reaction onset (cf entry 3). A blank reaction using
just HPF yet no complex 5 revealed the full consumption of the
substrate after 2 h and formation predominantly of styrene (entry
0). Dehydration of alcohols by strong acids via protonation fol-
lowed by elimination of H O is well known [41,42] and rationalizes
the dual role of HPF in this process: on one hand side, it is required
2
3
3
2
3
3
3
þ
21 30 4 2
C H N O
3
(100 mL,
1
obtain complex 6. Yield 25 mg, 44%. H NMR (300 MHz, CD
3
CN):
4
3
d
¼ 8.35 (d, JHH ¼ 2.6 Hz,1H, CpyH), 8.01 (d, JHH ¼ 9.2 Hz,1H, CpyH),
3
4
7.89 (s, 1H, CtrzH), 7.69 (dd, JHH ¼ 9.2 Hz, JHH ¼ 2.6 Hz, 1H, CpyH),
4.27 (s, 3H, NCH ), 3.99 (s, 3H, OCH ), 1.99 (s, 15H, CpeCH
), ꢁ14.18
(s, 1H, IreH). C{ H} NMR (75 MHz, CD CN):
¼ 157.5 (Cpy eOMe),
154.3 (CtrzeIr), 145.3 (CpyeNtrz), 140.0 (CpyH), 132.2 (CtrzH), 126.0
(CpyH), 114.9 (CpyH), 92.8 (Cp*), 57.6 (OCH ), 40.1 (NCH ), 10.2
(CpeCH ). Anal. Calcd for C20 IrN S (667.72): C, 35.98; H,
3
3
3
13
1
6
3
d
6
3
3
1
3
H
26
F
3
4 4
O
2
3.92; N, 8.39. Found: C, 35.66; H, 3.86; N, 8.16. HR-MS (CH
3
CN): m/z
þ
6
calculated for C19
H
26
N
4
OIr [MeOTf] ¼ 519.1736; found, 519.1713.
to abstract the iridium-bound hydride to form the dehydrogenation
catalyst, while on the other hand side it induces a non-metal
catalyzed dehydration which compromises selectivity. Hence,
there is an optimum iridium:acid ratio for promoting dehydroge-
nation with these iridium hydride complexes.
Complex 7a: Reaction of complex 3a (40 mg, 0.05 mmol) and
HSiEt (86 L, 0.54 mmol) were stirred according to the general
procedure to afford 7a as a yellow solid. Yield 17 mg, 48%. H NMR
3
m
1
3
(300 MHz, CD
(m, 2H, CpyH), 7.49 (ddd, JHH ¼ 7.4 Hz, JHH ¼ 5.8 Hz, JHH ¼ 1.8 Hz,
H, CpyH), 4.68e4.44 (m, 2H, OCH Me) 4.44 (s, 3H, NCH ), 1.94 (s,
), 1.44 (t, JHH ¼ 7.1 Hz, 3H, OCH CH ), ꢁ13.93 (s, 1H,
IreH). C{ H} NMR (75 MHz, CD CN):
¼ 159.8 (CtrzeIr),159.3 (C]
O), 153.6 (CpyH), 150.9 (CpyeNtrz), 140.8 (CpyH), 136.8 (CtrzeCOOEt),
126.6 (CpyH), 114.8 (CpyH), 93.7 (Cp*), 63.9 (OCH Me), 42.6 (NCH ),
14.4 (OCH CH ), 10.3 (CpeCH ). Anal. Calcd for C22 IrN
(709.76): C, 37.23; H, 3.98; N, 7.89. Found: C, 36.52; H, 3.69; N, 7.62.
HR-MS (CH CN): m/z calculated for Ir
[MeOTf] ¼ 561.1836; found, 561.1827.
Complex 7b: Complex 3b (60 mg, 0.077 mmol) and HSiEt
(125 L, 0.77 mmol) were stirred according to the general pro-
cedure to give 7b. Yield 21 mg, 37%. H NMR (300 MHz, CD
3
CN):
d
¼ 8.82 (d, JHH ¼ 5.8 Hz, 1H, CpyH), 8.13e7.95
3 3 4
1
2
3
3
3
. Conclusions
15H, CpeCH
3
2
3
13
1
3
d
Functionalized pyridyl-triazolylidene iridium chloride com-
plexes have been demonstrated to catalyze alcohol dehydration.
Tailoring of catalytic activity was accomplished by modification of
the donor properties of the triazolylidene ligand and revealed a
direct correlation between the ligand electronic properties and the
catalytic performance, with stronger donors increasing activity. The
corresponding iridium hydride complexes catalyze both the dehy-
dration as well as the dehydrogenation of alcohols. Their activity is
switched by the presence/absence of HPF
catalysis is attractive for the valorization of abundant feedstock
chemicals.
2
3
2
3
3
28
H F
3
4 5
O S
3
21 28 4 2
C H N O
þ
3
m
1
6
. Such multipurpose
3
CN):
¼ 8.41 (d, JHH ¼ 2.6 Hz,1H, CpyH), 8.08 (d, JHH ¼ 9.2 Hz,1H, CpyH),
4
3
d
3
4
7.72 (dd, JHH ¼ 9.2 Hz, JHH ¼ 2.6 Hz, 1H, CpyH), 4.64e4.44 (m, 2H,
OCH Me), 4.42 (s, 3H, NCH ), 4.01 (s, 3H, OCH ), 1.94 (s, 15H,
CpeCH CH ), ꢁ13.70 (s, 1H, IreH).
), 1.44 (t, JHH ¼ 7.1 Hz, 3H, OCH
C{ H} NMR (75 MHz, CD CN):
¼ 159.4 (C]O), 158.0 (CtrzeIr),
157.7 (CpyeOMe),144.7 (CpyeNtrz),139.6 (CpyH),136.7 (CtrzeCOOEt),
126.5 (CpyH), 115.1 (CpyH), 93.6 (Cp*), 63.8 (OCH Me), 57.6 (OCH ),
). HR-MS (CH CN): m/z
2
3
3
3
4
. Experimental section
3
2
3
1
3
1
3
d
General. The syntheses of the hydride compounds were carried
out in the glove box, and all the solvents were dried under active
molecular sieves and degassed using freeze-pump-thaw tech-
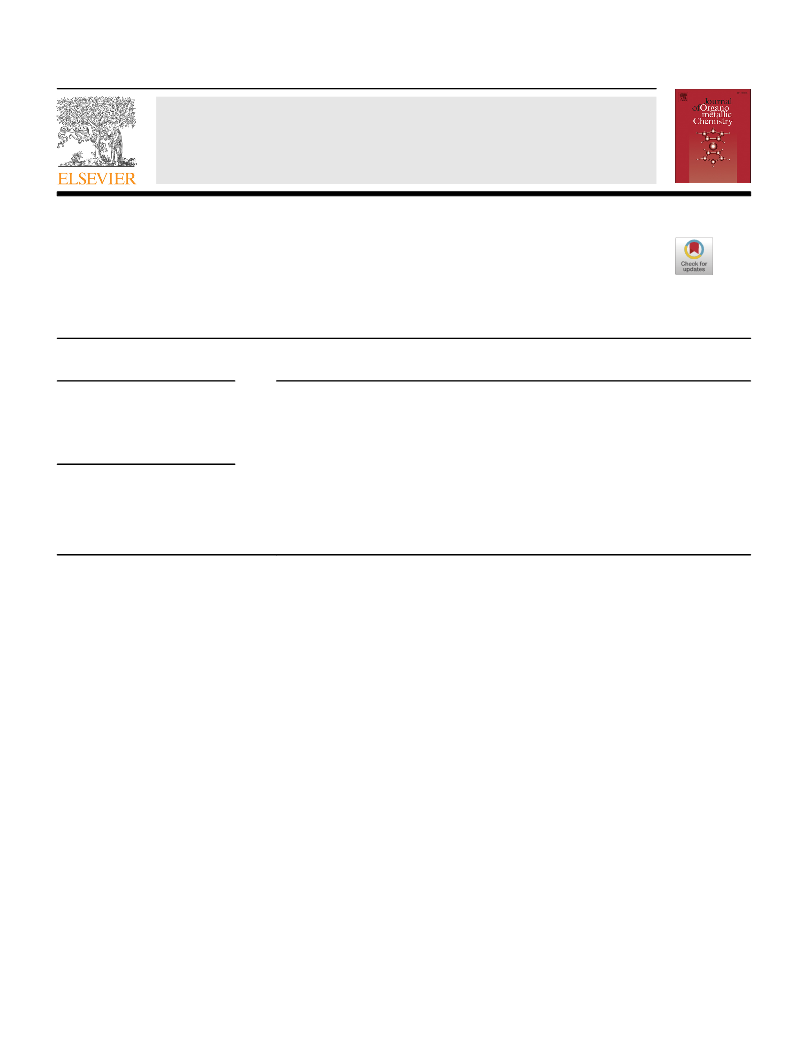
niques. Complexes 1e4 were prepared according to literature
procedure [33]. Unless specified, NMR spectra were recorded at
2
3
3
42.4 (NCH ), 14.5 (OCH
2
CH
3
), 10.3 (CpeCH
3
3
þ
calculated for C22
H
30
N
4
O
3
Ir [MeOTf] ¼ 591.1947; found, 591.1933.
Complex 7c: Reaction of complex 3c (40 mg, 0.052 mmol) and
HSiEt (83 L, 0.52 mmol) were suspended in 1,2-dichloroethane
ꢀ
1
2
7
5 C on Bruker spectrometers operating at 300 MHz ( H NMR) and
3
m
1
3
5 MHz ( C NMR), respectively. Chemical shifts (
d
in ppm, coupling
(1 mL) and stirred according to the general procedure to afford
1
1
constants J in Hz) were referenced to residual solvent signals ( H,
complex 7c as a yellow solid. Yield 14 mg, 36%. H NMR (300 MHz,
13
3
4
C). Assignments are based on homo- and heteronuclear shift
correlation spectroscopy. All complexes show a quartet around
CD
1H, CpyH), 7.08 (dd,
4.63e4.45 (m, 2H, OCH
1.93 (s, 15H, CpeCH
3
CN):
d
¼ 8.53 (d, JHH ¼ 6.7 Hz, 1H, CpyH), 7.67 (d, JHH ¼ 2.6 Hz,
3
4
J
HH ¼ 6.7 Hz,
J
HH ¼ 2.6 Hz, 1H, CpyH),
Me), 4.43 (s, 3H, NCH ), 4.03 (s, 3H, OCH ),
CH
), ꢁ13.80
¼ 169.3 (CpyeOMe),
160.3 (CtrzeIr), 159.2 (C]O), 153.5 (CpyH), 151.9 (CpyeNtrz), 136.8
(CtrzeCOOEt), 113.9 (CpyH), 100.0 (CpyH), 93.1 (Cp*), 63.7 (OCH Me),
57.9 (OCH ), 42.5 (NCH ), 14.3 (OCH CH ), 10.2 (CpeCH ). Anal.
Calcd for C23 IrN S (739.79): C, 37.34; H, 4.09; N, 7.57.
e
1
20 ppm in the 13C NMR spectrum due to the OTf counterion.
2
3
3
3
Purity of the complexes has been established by NMR spectroscopy.
3
), 1.44 (t, JHH ¼ 7.1 Hz, 3H, OCH
2
3
13
1
General procedure for the synthesis of the complexes 5e7: In
a Schlenk flask in the glove box, compound 1e3 (1 eq) and HSiEt
10 eq) were suspended in dry 1,2-dichloroethane (1.5 mL) and
stirred for 2 h at 100 C and for another 2 h at room temperature.
The reaction mixture was layered with dry Et O, which induced
precipitation of complexes 5e7 as yellow solids.
Complex 5: Reaction of complex 1 (60 mg, 0.08 mmol) and
HSiEt (100 L, 0.80 mmol) were suspended in dichloroethane
(s, 1H, IreH). C{ H} NMR (75 MHz, CD CN):
3
d
3
(
2
ꢀ
3
3
2
3
3
2
H F
30 3
4 6
O
Found: C, 37.15; H, 4.01; N, 7.36. HR-MS (CH
3
CN): m/z calculated for
þ
C
22
H
30
N
4
O
3
Ir [MeOTf] ¼ 591.1947; found, 591.1921.
3
m
Typical procedure for the dehydration of 1-phenylethanol

 Albrecht, Martin
Albrecht, Martin
 Olivares, Marta
Olivares, Marta