Organic Letters
Letter
Lett. 1992, 21, 1983. (d) T. Teunissen, H. Chem. Commun. 1998, 1367.
(e) van Engelen, M. C.; Teunissen, H. T.; de Vries, J. G.; Elsevier, C. J. J.
Mol. Catal. A: Chem. 2003, 206, 185.
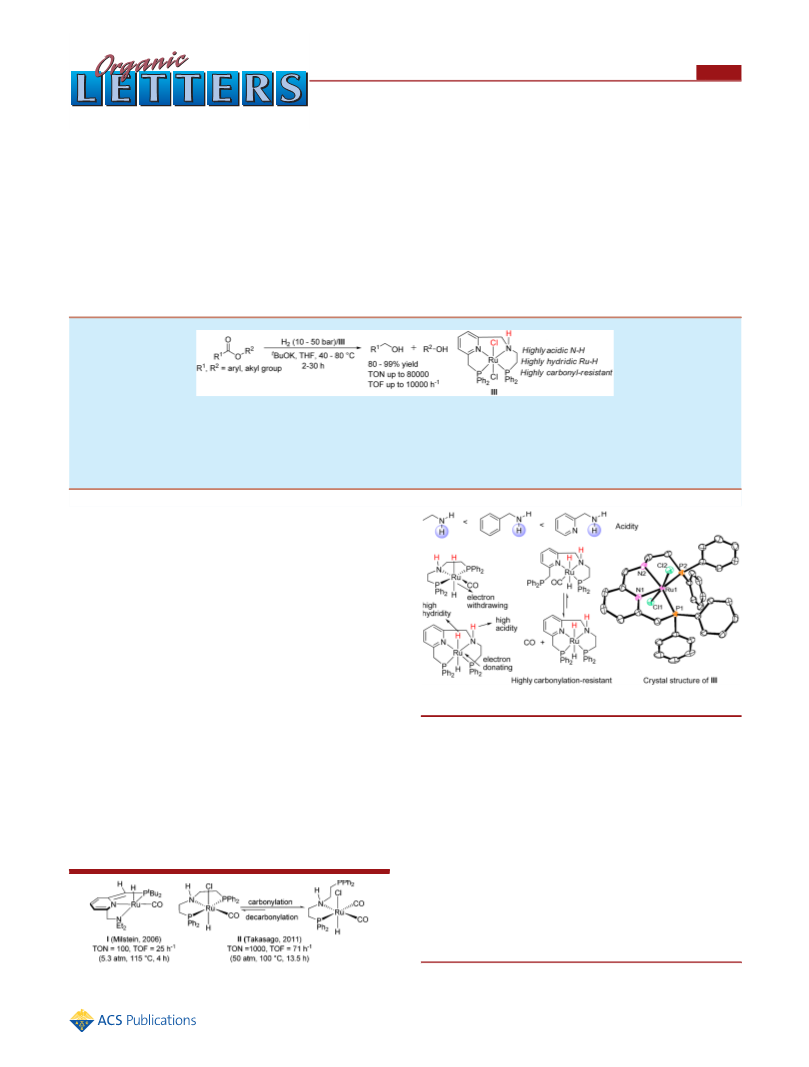
hydride transfer, the H2 complexation in the first part of the
catalytic cycle is the rate-determining step (ca. formation of
TS2_Na_L), with an overall free energy barrier of 21.9 kcal/mol
calculated for the model catalyst 0_Na_CO, 16.3 kcal/mol for
the model catalyst 0_Na_Py, and 20.4 kcal/mol for the model
catalyst 0_Na. Replacement of the carbonyl ligand in 0_Na_L
with a pyridine ligand results in a significant decrease in the
overall barrier by 5.6 kcal/mol, suggesting that a pyridine ligand
has great promoting effect relative to CO. A carbonyl ligand
weakens the complexation ability of the metal center for H2 and
therefore gives rise to a higher overall barrier. The model catalyst
0_Na gives an overall barrier of 20.4 kcal/mol, 1.5 kcal/mol
lower than the model catalyst 0_Na_CO does. A difference of 1.5
kcal/mol in the overall barrier represents about 10 times
difference in the reaction rate when we apply the Arrhenius rate
equation. Besides, in our calculations, when the sodium cation in
0_Na is replaced by a potassium cation (giving the model catalyst
0_K), an overall barrier was calculated to be 18.5 kcal/mol, which
is 1.9 kcal/mol lower when compared with that using the model
catalyst 0_Na, consistent with the experimental observation of
rate enhancement.15
(5) (a) Zhang, J.; Leitus, G.; Ben-David, Y.; Milstein, D. Angew. Chem.,
Int. Ed. 2006, 45, 1113. (b) Balaraman, E.; Gunanathan, C.; Zhang, J.;
Shimon, L. J. W.; Milstein, D. Nat. Chem. 2011, 3, 609. (c) Fogler, E.;
Balaraman, E.; Ben-David, Y.; Leitus, G.; Shimon, L. J. W.; Milstein, D.
Organometallics 2011, 30, 3826. (d) Gunanathan, C.; Milstein, D. Acc.
Chem. Res. 2011, 44, 588. (e) Fogler, E.; Garg, J. A.; Hu, P.; Leitus, G.;
Shimon, L. J. W.; Milstein, D. Chem.Eur. J. 2014, 20, 15727.
(6) (a) Saudan, L. A.; Saudan, C. M.; Debieux, C.; Wyss, P. Angew.
Chem., Int. Ed. 2007, 46, 7473. (b) Saudan, L.; Dupau, P.; Riedhauser, J.-
J.; Wyss, P.; Firmenich S. A. Swiss Patent WO2006106483A1, 2006; p
40. (c) Saudan, L.; Dupau, P.; Riedhauser, J.-J.; Wyss, P.; Firmenich S. A.
Swiss Patent WO2006106484A1, 2006; p 29. (d) Saudan, C.; Saudan,
L.; Firmenich S. A. Swiss Patent WO2010061350A1, 2010; p 55.
(7) (a) Kuriyama, W.; Ino, Y.; Ogata, O.; Sayo, N.; Saito, T. Adv. Synth.
Catal. 2010, 352, 92. (b) Ino, Y.; Kuriyama, W.; Ogata, O.; Matsumoto,
T. Top. Catal. 2010, 53, 1019. (c) Kuriyama, W.; Matsumoto, T.; Ino, Y.;
Ogata, O. Takasago International Corporation, Japan. Patent
WO2011048727A1, 2011; p 62. (d) Kuriyama, W.; Matsumoto, T.;
Ogata, O.; Ino, Y.; Aoki, K.; Tanaka, S.; Ishida, K.; Kobayashi, T.; Sayo,
N.; Saito, T. Org. Process Res. Dev. 2012, 16, 166.
(8) (a) Acosta-Ramirez, A.; Bertoli, M.; Gusev, D. G.; Schlaf, M. Green
Chem. 2012, 14, 1178. (b) Spasyuk, D.; Smith, S.; Gusev, D. G. Angew.
Chem., Int. Ed. 2012, 51, 2772. (c) Spasyuk, D.; Gusev, D. G.
Organometallics 2012, 31, 5239. (d) Spasyuk, D.; Smith, S.; Gusev, D. G.
Angew. Chem., Int. Ed. 2013, 52, 2538.
(9) (a) W. W. N, O.; Lough, A. J.; Morris, R. H. Chem. Commun. 2010,
46, 8240. (b) Ito, M.; Ootsuka, T.; Watari, R.; Shiibashi, A.; Himizu, A.;
Ikariya, T. J. Am. Chem. Soc. 2011, 133, 4240. (c) Touge, T.; Hakamata,
T.; Nara, H.; Kobayashi, T.; Sayo, N.; Saito, T.; Kayaki, Y.; Ikariya, T. J.
Am. Chem. Soc. 2011, 133, 14960. (d) Junge, K.; Wendt, B.; Westerhaus,
F. A.; Spannenberg, A.; Jiao, H.; Beller, M. Chem.Eur. J. 2012, 18,
9011. (e) Otsuka, T.; Ishii, A.; Dub, P. A.; Ikariya, T. J. Am. Chem. Soc.
2013, 135, 9600. (f) Chakraborty, S.; Dai, H.; Bhattacharya, P.;
Fairweather, N. T.; Gibson, M. S.; Krause, J. A.; Guan, H. J. Am. Chem.
Soc. 2014, 136, 7869.
In conclusion, we have developed a highly efficient (TON up
to 80000, TOF up to 10000 h−1) tetradentate ruthenium
complex for ester reduction. In particular, the hydrogenations of
fatty acid esters to fatty alcohols showed excellent results, which
hold potential to replace the heterogeneous hydrogenation
process. Deep insight into the bifunctional mechanism helped to
develop the highly efficient catalyst, and DFT studies explained a
possible reason for the high efficiency of catalyst III. This insight
will help us develop more efficient catalysts for hydrogenation of
esters to alcohols, a fundamental process in chemical trans-
formations.
ASSOCIATED CONTENT
* Supporting Information
■
S
(10) (a) Clarke, M. L. Catal. Sci. Technol. 2012, 2, 2418. (b) Dub, P. A.;
Ikariya, T. ACS Catal. 2012, 2, 1718. (c) Werkmeister, S.; Junge, K.;
Beller, M. Org. Process Res. Dev. 2014, 18, 289.
Experimental details and characterization data. This material is
(11) (a) Abdur-Rashid, K.; Clapham, S. E.; Hadzovic, A.; Harvey, J. N.;
Lough, A. J.; Morris, R. H. J. Am. Chem. Soc. 2002, 124, 15104.
AUTHOR INFORMATION
Corresponding Authors
■
(b) Sandoval, C. A.; Ohkuma, T.; Muniz, K.; Noyori, R. J. Am. Chem. Soc.
̃
2003, 125, 13490. (c) Abbel, R.; Abdur-Rashid, K.; Faatz, M.; Hadzovic,
A.; Lough, A. J.; Morris, R. H. J. Am. Chem. Soc. 2005, 127, 1870.
(d) Takebayashi, S.; Bergens, S. H. Organometallics 2009, 28, 2349.
(e) John, J. M.; Takebayashi, S.; Dabral, N.; Miskolzie, M.; Bergens, S. H.
J. Am. Chem. Soc. 2013, 135, 8578. (f) Dub, P. A.; Henson, N. J.; Martin,
R. L.; Gordon, J. C. J. Am. Chem. Soc. 2014, 136, 3505.
Notes
The authors declare no competing financial interest.
(12) (a) Ohkuma, T.; Sandoval, C. A.; Srinivasan, R.; Lin, Q.; Wei, Y.;
Muniz, K.; Noyori, R. J. Am. Chem. Soc. 2005, 127, 8288. (b) Arai, N.;
Suzuki, K.; Sugizaki, S.; Sorimachi, H.; Ohkuma, T. Angew. Chem., Int.
Ed. 2008, 47, 1770.
(13) The X-ray crystal data of III have been deposited with the
Cambridge Crystallographic Data Centre as supplementary publication
no. CCDC 989213. Copies of the data can be obtained, free of charge,
on application to the CCDC, 12 Union Road, Cambridge CB2 1EZ, UK
(14) Labet, M.; Thielemans, W. Chem. Soc. Rev. 2009, 38, 348.
(15) Hartmann, R.; Chen, P. Angew. Chem., Int. Ed. 2001, 40, 3581.
̃
ACKNOWLEDGMENTS
■
We are grateful for financial support by grants from Wuhan
University (203273463), the Natural Science Foundation of
Hubei Province (2014CFB181), and the “111” Project of the
Ministry of Education of China and the National Natural Science
Foundation of China (Grant Nos. 21372179 and 21432007).
REFERENCES
■
(1) Seyden-Penne, J. Reductions by the Allumino- and Borohydride in
Organic Synthesis, 2nd ed.; Wiley-VCH: New York, 1997.
(2) Takebayashi, S.; Bergens, S. H. Organometallics 2009, 28, 2349.
(3) (a) Rieke, R.; Thakur, D.; Roberts, B.; White, G. J. Am. Oil Chem.
1997, 74, 333. (b) Pouilloux, Y.; Autin, F.; Barrault, J. Catal. Today 2000,
63, 87.
(4) (a) Grey, R. A.; Pez, G. P.; Wallo, A. J. Am. Chem. Soc. 1981, 103,
7536. (b) Matteoli, U.; Menchi, G.; Bianchi, M.; Piacenti, F. J. Mol. Catal.
1988, 44, 347. (c) Hara, Y.; Inagaki, H.; Nishimura, S.; Wada, K. Chem.
D
Org. Lett. XXXX, XXX, XXX−XXX

 Tan, Xuefeng
Tan, Xuefeng
 Wang, Yan
Wang, Yan
 Liu, Yuanhua
Liu, Yuanhua
 Wang, Fangyuan
Wang, Fangyuan
 Shi, Liyang
Shi, Liyang
 Lee, Ka-Ho
Lee, Ka-Ho
 Lin, Zhenyang
Lin, Zhenyang
 Lv, Hui
Lv, Hui
 Zhang, Xumu
Zhang, Xumu