
Tetrahedron Letters (2020)
Update date:2022-08-11
Topics:
 Sun, Wanwan
Sun, Wanwan
 Chen, Xiaobei
Chen, Xiaobei
 Hu, Ying
Hu, Ying
 Geng, Huihui
Geng, Huihui
 Jiang, Yuanrui
Jiang, Yuanrui
 Zhou, Yuxin
Zhou, Yuxin
 Zhu, Wenjing
Zhu, Wenjing
 Hu, Min
Hu, Min
 Hu, Haohua
Hu, Haohua
 Wang, Xingyi
Wang, Xingyi
 Wang, Xinli
Wang, Xinli
 Zhang, Shilei
Zhang, Shilei
 Hu, Yanwei
Hu, Yanwei
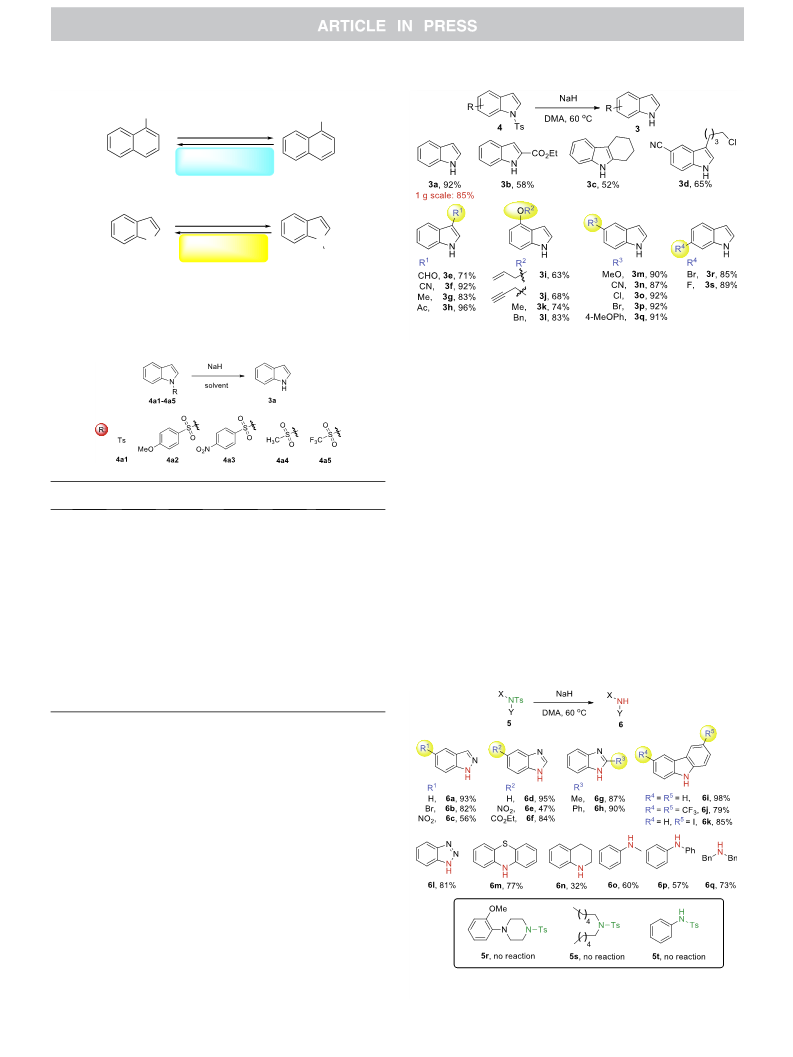
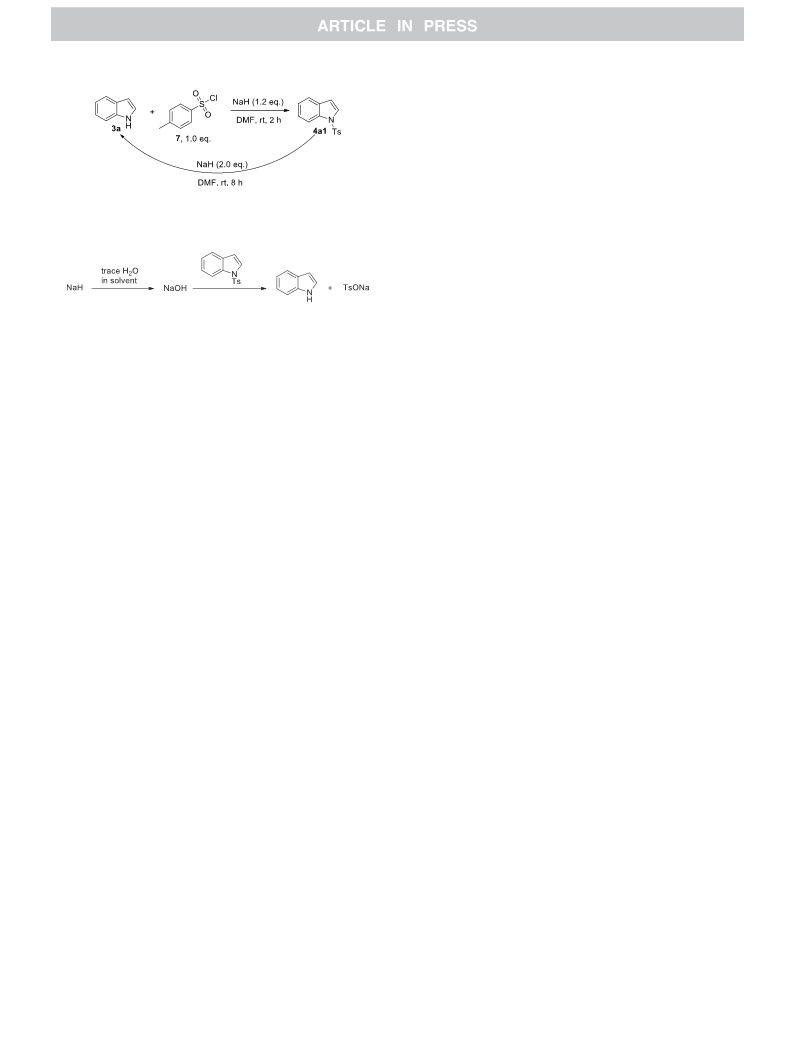
A NaH-mediated detosylation reaction of various Ts-protected indoles, azaheterocycles, anilines and dibenzylamine was reported. The method features cheap reagent, convenient operations, mild reaction conditions and broad substrate scope. Moreover, this study revealed that the loading of NaH in tosylation reactions of nitrogen-containing compounds with NaH as a base in DMA or DMF should be controlled due to the possibility of adverse detosylation.
View More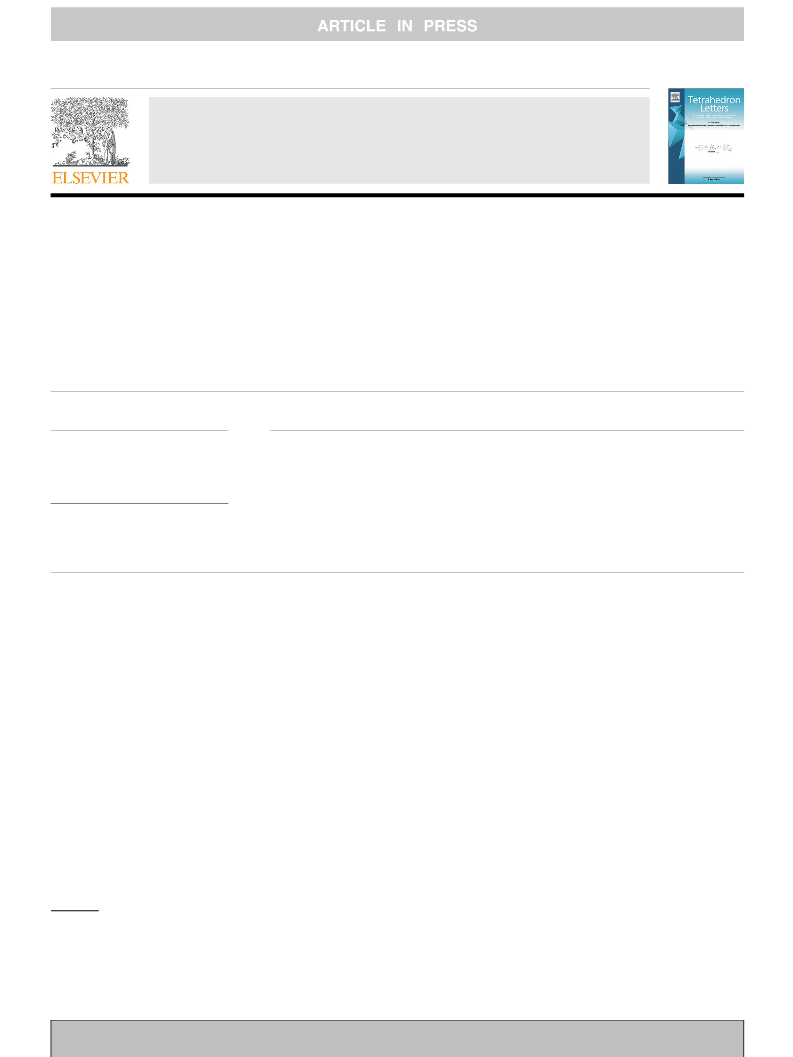

Chongqing Shuangfeng Chemical Co.,Ltd
Contact:+86-23-49850156
Address:No.663,xuanhua Rd,yongchuan,chongqing,China
Wuhan Chemchemical Co., Ltd.(expird)
Contact:15973022782
Address:7-5-6218,Incubation Centre,Guandong Industry Park, East Lake High-Tech Development Zone,Wuhan City.
Chengdu NoVi Biotechnology Co., Ltd.(expird)
Contact:13551243286/028-81458053
Address:NO.168-1-224 JULONG ROAD WUHOU DISTRICT, CHENGDU CITY,SICHUAN PROVINCE
Zhengzhou Xinlian Chemical Tech Co. ,Ltd
Contact:0371-65771781 021-52042910
Address:H Part, Building I, No. 700 Gonglu Road, Pudong New Area, Shanghai
Jiangsu Chiatai Qingjiang Pharmaceutical Co.,Ltd
Contact:+86-517-86283327
Address:9 North Hantai Road, Huaian, China
Doi:10.1021/acs.jmedchem.8b01287
(2018)Doi:10.1016/S0957-4166(00)80170-6
(1993)Doi:10.1080/10286020.2012.762764
(2013)Doi:10.1021/acs.joc.6b00191
(2016)Doi:10.1002/jhet.2036
(2015)Doi:10.1016/S0008-6215(00)82373-3
(1974)